according to standard literature and latest studies and reporting what is the pathogenesis of PXE
Reading File
Finding Sources
Searching PubMed
pseudoxanthoma elasticum pathogenesis ABCC6
Searching the Web
pseudoxanthoma elasticum PXE pathogenesis 2024 2025 ABCC6 ectopic mineralization mechanism
Reading File
Reading File
Reading Article
Reading Article
Reading File
Reading File
Reading File
Reading Web Page
PXE pathogenesis ABCC6 pyrophosphate mineralization mechanism elastic fiber
Finding Sources
Loading Image
Loading Image
I now have comprehensive information from multiple authoritative sources. Let me compile the complete answer.
Pathogenesis of Pseudoxanthoma Elasticum (PXE)
PXE (OMIM #264800) is an autosomal recessive multisystem connective tissue disease characterized by progressive ectopic mineralization and fragmentation of elastic fibers in the skin, Bruch's membrane of the eye, and the walls of midsized arteries. Understanding its pathogenesis requires integrating genetics, metabolic biochemistry, and tissue-level consequences.
1. Genetic Basis: ABCC6 Mutations
PXE is caused by biallelic loss-of-function mutations in the ABCC6 gene (chromosome 16p13.1), which encodes ATP-binding cassette sub-family C member 6 (ABCC6), also called multidrug resistance-associated protein 6 (MRP6). More than 300 pathogenic variants have been described (missense, nonsense, frameshift, large deletions). The most common in European populations is the p.Arg1141X nonsense mutation, accounting for ~30% of mutant alleles.
ABCC6 is an ATP-dependent transmembrane organic anion transporter expressed predominantly in the basolateral (sinusoidal) membrane of hepatocytes and to a lesser degree in renal proximal tubular cells. It is virtually absent from the affected target tissues — skin, eyes, and arteries — which is the key to understanding PXE as a metabolic (hepatic) disease rather than a primary connective tissue disorder.
(Andrews' Diseases of the Skin; Dermatology 2-Volume Set 5e, Fuster & Hurst's The Heart 15e)
2. The Central Metabolic Cascade: ABCC6 → ATP → PPi
This is the core molecular mechanism, now well-established from transplantation studies and parabiotic mouse models (Jiang et al., 2009–2010):
Step 1 — Hepatic ATP efflux
ABCC6 mediates the efflux of ATP across the basolateral membrane of hepatocytes into the portal sinusoidal circulation. This is the liver's primary contribution to systemic nucleotide pools.
Step 2 — ENPP1-mediated hydrolysis
The membrane-bound ectonucleotidase ENPP1 (ectonucleotide pyrophosphatase/phosphodiesterase 1) hydrolyzes extracellular ATP into AMP + inorganic pyrophosphate (PPi).
Step 3 — CD73 further metabolizes AMP → adenosine
CD73 (encoded by NT5E) converts AMP to adenosine, another anti-calcification molecule. This complete pathway (ABCC6 → ENPP1 → CD73 → TNAP) constitutes a purinergic signaling cascade that regulates mineralization inhibition.
Step 4 — PPi deficiency leads to unchecked mineralization
PPi is a potent inhibitor of hydroxyapatite crystal formation and precipitation onto organic matrices. In PXE patients, plasma PPi levels are significantly reduced (~50% of normal). Without adequate PPi, calcium phosphate deposits form preferentially on elastic fibers in connective tissues.
This explains the tissue selectivity: elastic fibers in skin dermis, Bruch's membrane, and arterial media have a particular propensity for calcium binding in the absence of PPi suppression.
(Kauffenstein G et al., Biology 2024, PMID 38392293; Jansen et al., PNAS 2013; Pfau K et al., Prog Retin Eye Res 2024, PMID 38815804)
3. The Purinergic Disease Concept (2024 Update)
The 2024 review by Kauffenstein, Martin & Le Saux reframes PXE as part of a "purinergic disease continuum" alongside:
| Disease | Gene | Pathway step affected |
|---|---|---|
| PXE | ABCC6 | ATP efflux from liver |
| GACI (Generalized Arterial Calcification of Infancy) | ENPP1 | ATP → PPi conversion |
| CALJA (Calcification of Joints and Arteries) | NT5E/CD73 | AMP → adenosine |
All three disorders share overlapping phenotypes and disrupt the same purinergic pathway at different points. PPi deficiency is the final common mechanism of ectopic mineralization in all three.
Beyond PPi, there are broad alterations in purinergic receptor signaling (P1 and P2 receptors), which may contribute to vascular tone dysregulation, inflammation, and platelet dysfunction in PXE.
4. Role of Other Mineralization Inhibitors
Matrix Gla Protein (MGP)
MGP is a potent local inhibitor of soft tissue calcification. It requires vitamin K-dependent γ-glutamyl carboxylation (mediated by GGCX) to become active. In PXE, there is secondary undercarboxylation of MGP in target tissues, contributing to reduced local mineralization inhibition. This is why warfarin (VKORC1 inhibitor → blocks vitamin K recycling → impairs MGP carboxylation) markedly accelerates ectopic mineralization in Abcc6−/− mice.
Clinical implication: Vitamin K antagonists should be avoided in PXE patients.
Fetuin-A
Fetuin-A (α2-Heremans-Schmid glycoprotein) is a serum glycoprotein that acts as a systemic "crystal inhibitor" by forming calciprotein particles. Reduced fetuin-A has been reported in PXE patients, potentially amplifying the pro-mineralization state.
Tissue-nonspecific alkaline phosphatase (TNAP)
TNAP hydrolyzes PPi → Pi (phosphate), so it promotes calcification by consuming PPi. Inhibition of TNAP in Abcc6−/− mice attenuates ectopic mineralization, making it a therapeutic target.
5. Role of the Bone Marrow and Immune System (Emerging Evidence, 2024)
A landmark 2024 study (PMC11260544) demonstrated that bone marrow–derived cells (particularly lymphocytes) also express ABCC6, and that restoring wild-type bone marrow in Abcc6−/− mice significantly reduces calcification. This suggests:
- The liver is not the sole source of circulating PPi-enabling activity
- The adaptive immune system and local inflammation (lymphangiogenesis was found in PXE skin) contribute substantially to the calcification process
- This opens a new mechanistic dimension beyond the "pure hepatic metabolic disease" model
6. Tissue-Level Pathology: Elastic Fiber Mineralization
Once plasma PPi is deficient and local inhibitors are insufficient, hydroxyapatite crystals preferentially nucleate on the mid-laminar elastic fibers in:
- Dermis (mid-portion) → yellow papules, "plucked chicken skin"
- Bruch's membrane (between RPE and choroid) → angioid streaks, peau d'orange, macular neovascularization
- Medial layer of midsized arteries → premature atherosclerosis, peripheral artery disease, renal hypertension
Histopathology: Elastic fibers appear fragmented, twisted, and curled ("raveled wool") on H&E; calcium stains (von Kossa, Alizarin red) highlight early deposits. The fibers stain gray-blue on H&E. Calcification is most severe in the mid-dermis.
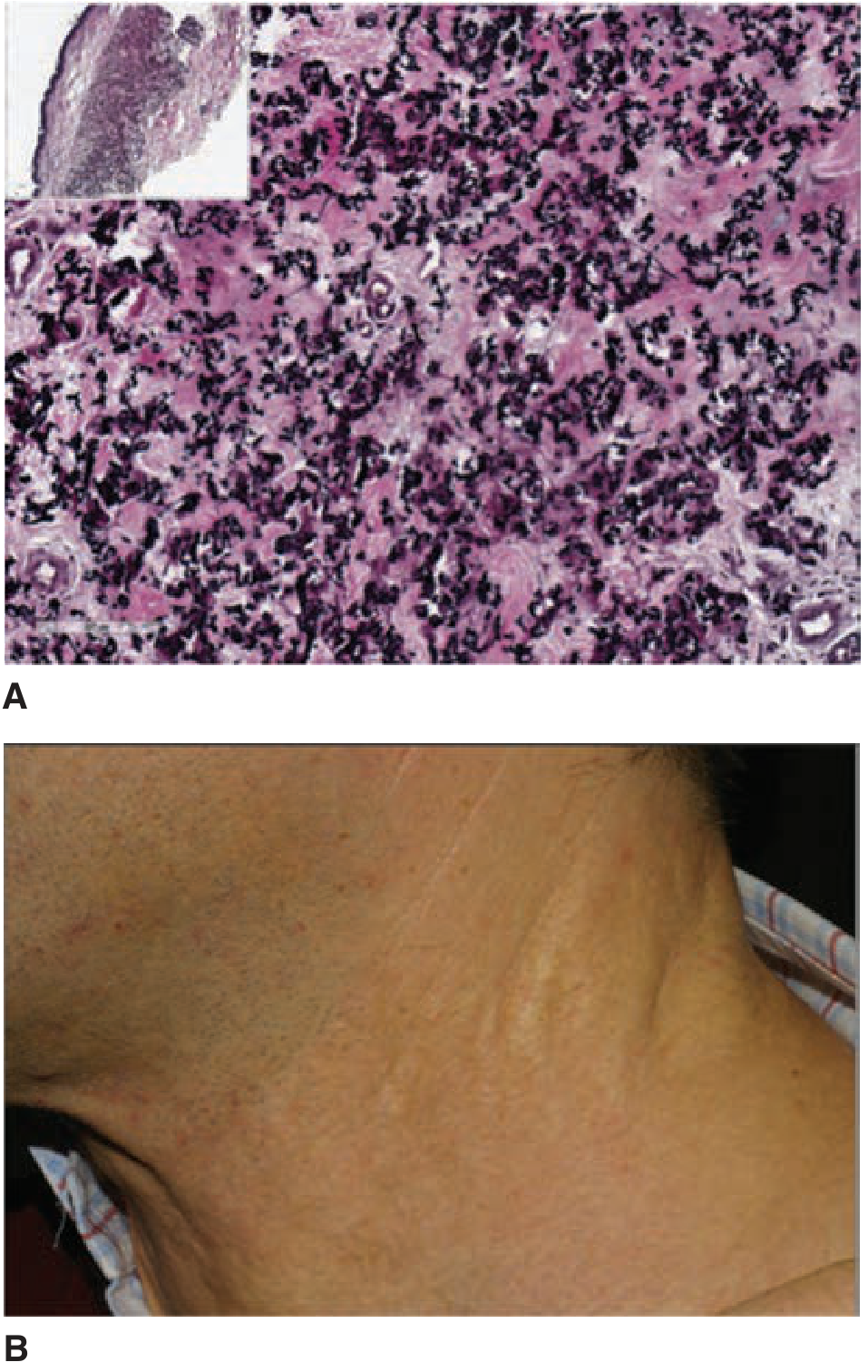
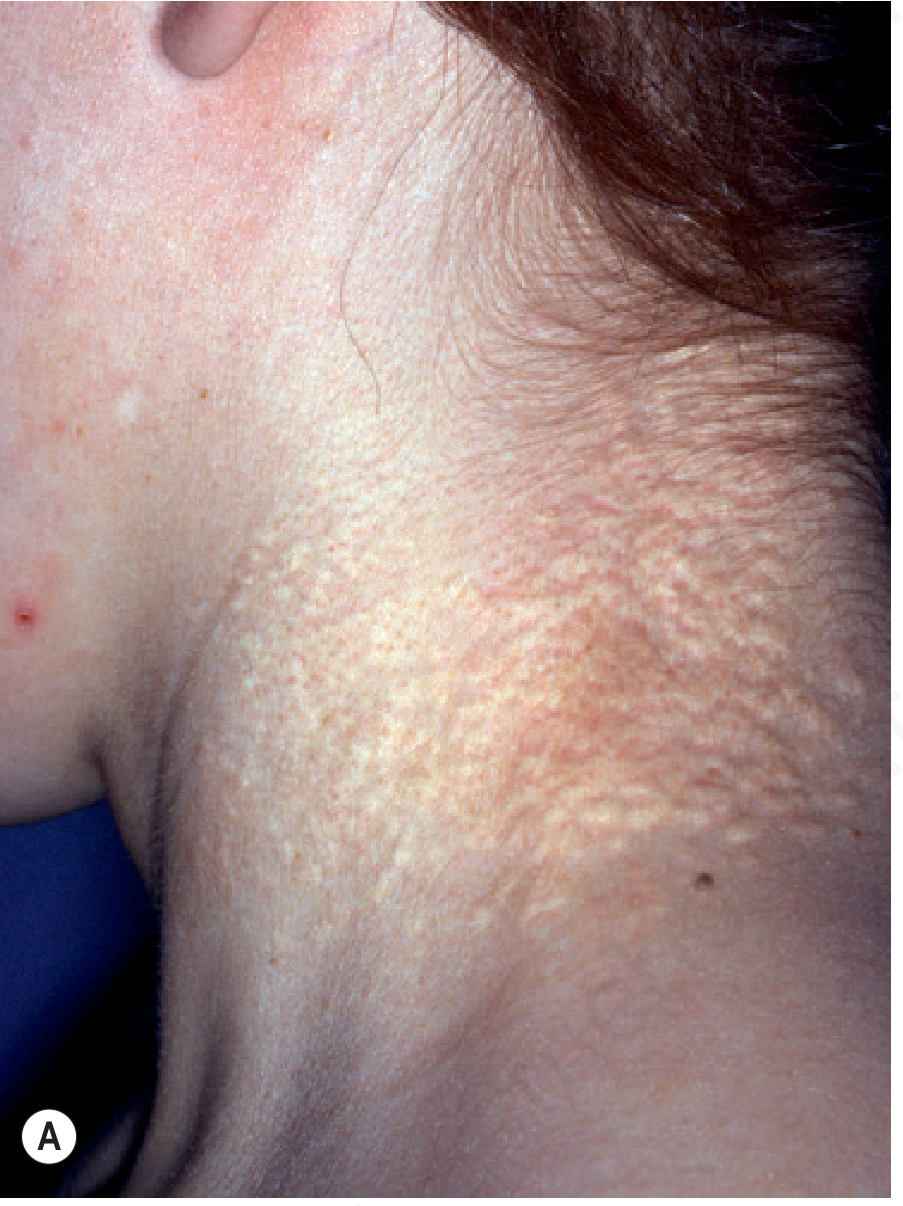
7. PXE-Like Phenocopies
Several conditions mimic or share mechanisms with PXE:
- β-thalassemia / sickle cell disease: Secondary hemolysis promotes ABCC6 downregulation through iron overload and oxidative stress; PXE-like skin, vascular, and ocular lesions develop
- GGCX mutations: Loss of γ-glutamyl carboxylase → impaired MGP activation → PXE-like phenotype + coagulopathy
- Penicillamine use (Wilson disease): Drug-induced elastin cross-linking disruption → clinicohistologic PXE mimicry
Summary Schematic
ABCC6 mutations (liver)
↓
↓ Hepatic ATP efflux
↓
ENPP1 cannot generate adequate PPi
↓
↓ Plasma PPi + ↓ Carboxylated MGP + ↓ Fetuin-A
↓
Hydroxyapatite nucleation on elastic fibers
↓
Skin (dermis) → yellow papules, skin laxity
Eye (Bruch's membrane) → angioid streaks, CNV, blindness
Arteries (media) → premature atherosclerosis, hypertension, claudication
Key Recent Literature
| PMID | Study | Key Finding |
|---|---|---|
| 38815804 | Pfau et al., Prog Retin Eye Res 2024 | Comprehensive genetics–pathophysiology–clinical review; ocular focus |
| 38392293 | Kauffenstein et al., Biology 2024 | PXE as a "purinergic disease"; ABCC6→ENPP1→CD73→TNAP axis |
| 41519013 | Martin et al., Ann Dermatol Venereol 2026 | REACT-PXE consensus on diagnosis and future research |
| PMC11260544 | Bone marrow transplant study 2024 | Bone marrow ABCC6 contributes to PPi; adaptive immune system involvement |
Textbook sources: Andrews' Diseases of the Skin (9780323547536); Dermatology 2-Volume Set 5e (9780702082252); Fuster & Hurst's The Heart 15e (9781264257560); Emery's Elements of Medical Genetics and Genomics (9780702079665)
This is a shared conversation. Sign in to Orris to start your own chat.