enteric fever
Reading File
Finding Sources
Finding Sources
Searching PubMed
"enteric fever" OR "typhoid fever"
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive review of enteric fever:
Enteric Fever (Typhoid Fever)
Definition & Etiology
Enteric fever is a systemic febrile illness caused by dissemination of Salmonella Typhi or S. Paratyphi (serotypes A, B, and C). The term "typhoid fever" persists from its early confusion with typhus; "enteric fever" was proposed in 1869 to reflect the anatomic site (small intestine) and is now used interchangeably. Unlike nontyphoidal Salmonella, these serovars have no animal reservoir—humans are the sole hosts.
— Harrison's Principles of Internal Medicine 22E, p. 1359
Epidemiology
- ~9.2–21 million cases of typhoid fever and ~5 million cases of paratyphoid fever occur annually, with 110,000–280,000 deaths/year
- Highest incidence: Indian subcontinent (India, Pakistan, Bangladesh, Nepal), Eastern Mediterranean, sub-Saharan Africa — exceeding 1000 cases/100,000 children in some urban areas
- Transmission: fecal-oral route via contaminated food/water; chronic carriers are a key reservoir
- In the US: ~5700 cases/year; 78% travel-associated, predominantly from the Indian subcontinent; only 3% of affected travelers had been vaccinated
Risk factors: contaminated drinking water/ice, street food, raw produce fertilized with sewage, household contacts with illness, poor hand hygiene, prior H. pylori infection (reduced gastric acidity)
— Harrison's, p. 1359–1360
Pathophysiology
- Ingested organisms penetrate the mucosa of the terminal ileum (via M cells overlying Peyer's patches)
- Pass to mesenteric lymph nodes → primary bacteremia → dissemination to reticuloendothelial system (liver, spleen, bone marrow)
- Secondary bacteremia occurs after intracellular replication → symptomatic disease
- Necrosis of Peyer's patches underlies the complications of hemorrhage and perforation
Clinical Course
Incubation period: 5–21 days (mean 10–14 days); depends on inoculum size and host immune status.
Week 1
- Stepwise rising fever up to 38.8–40.5°C (101.8–104.9°F)
- Headache (80%), chills, malaise, dry cough (30%), myalgias
- Relative bradycardia (pulse-temperature dissociation) in up to 50%
- Coated tongue (51–56%)
Week 2
- Sustained high fever
- Rose spots (~30% of patients): faint, salmon-colored, blanching maculopapular lesions on trunk/chest — visible at end of week 1 or during week 2; resolve in 2–5 days; Salmonella can be cultured from punch biopsies
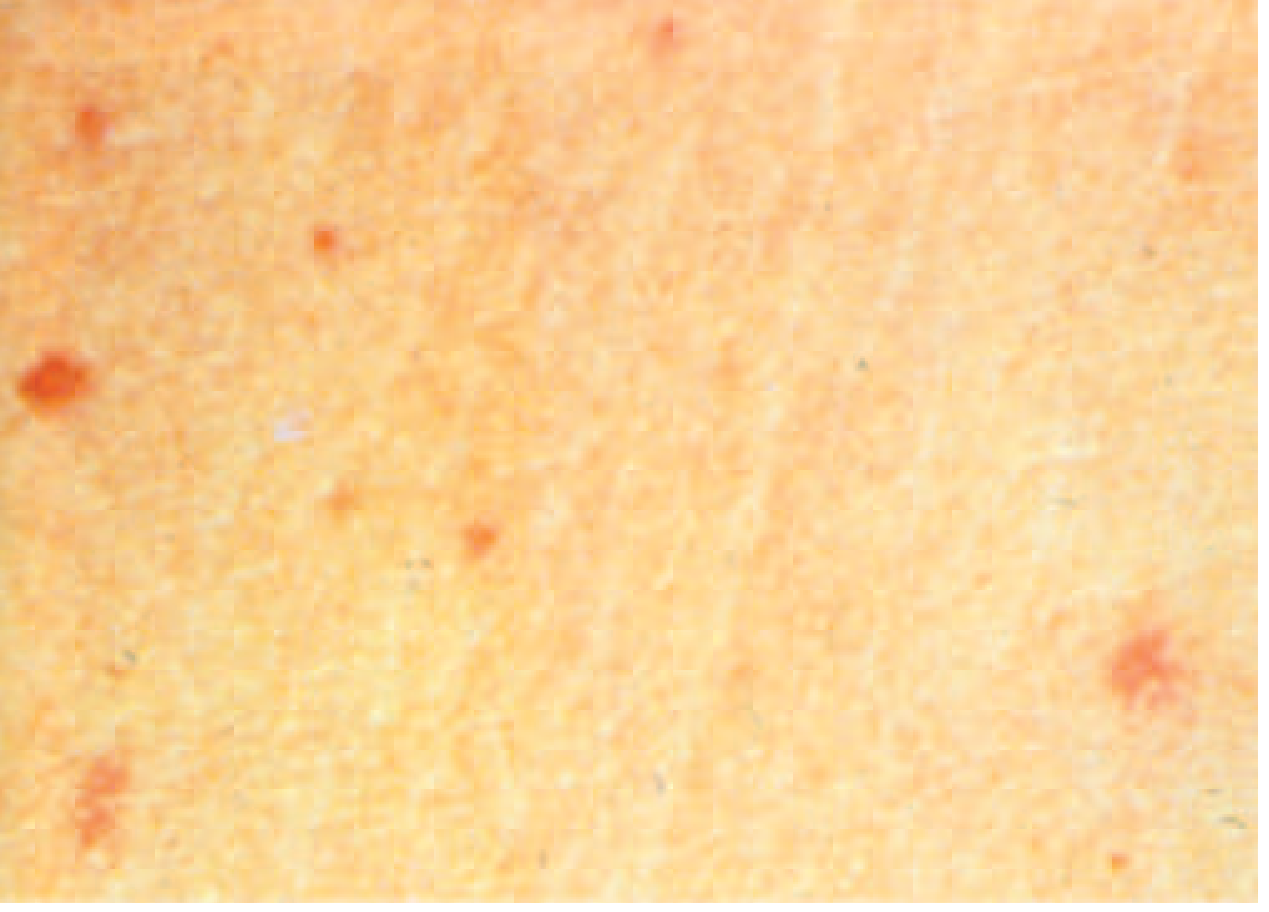
"Rose spots" of enteric fever due to S. Typhi or S. Paratyphi (Harrison's 22E, Fig. 171-2)
- Hepatosplenomegaly (~50%)
- Abdominal distension, pain, diarrhea or constipation
- CNS: apathy, confusion, delirium, rarely psychosis
Week 3–4
- Complications in ~5% of untreated patients: intestinal hemorrhage and perforation (from necrosis of Peyer's patches) — the most feared complications
- Rare: pancreatitis, cholecystitis, infective endocarditis, meningitis, pneumonia, hepatic/splenic abscess, orchitis
- Untreated illness typically resolves by end of week 4
— Harrison's, p. 1360; Goldman-Cecil Medicine, p. 3146
Diagnosis
| Test | Sensitivity | Notes |
|---|---|---|
| Blood culture | 40–80% (Week 1) | Gold standard; highest yield in week 1 |
| Bone marrow culture | ~80–95% | Remains positive even after antibiotics started |
| Stool culture | ~30–40% (Week 2–3) | Higher yield later in illness |
| Urine culture | ~25% | Lower yield |
| Widal test | Variable | Low specificity in endemic areas; not reliable |
| PCR | Emerging | Available in research settings |
- CBC: Leukopenia and neutropenia in ~20%; anemia; thrombocytopenia possible
- LFTs: Elevated transaminases are common
Antibiotic Therapy
Overall case-fatality rate is 2.5% (untreated: 10–30%); prompt appropriate treatment reduces mortality to <1%.
Drug Resistance — Critical Context
- MDR strains (resistant to chloramphenicol, ampicillin, TMP-SMX): emerged 1980s, widespread
- Decreased susceptibility to ciprofloxacin (DSC) / fluoroquinolone resistance: dominant on the Indian subcontinent (clone H58); 66% of US isolates were DSC in 2015
- XDR S. Typhi (since 2016, Pakistan): resistant to ceftriaxone + all of the above; susceptible only to azithromycin and carbapenems
Treatment Table (Adults)
| Indication | Agent | Dose | Duration |
|---|---|---|---|
| Empirical | Ceftriaxone | 2 g/d IV | 10–14 days |
| Ciprofloxacin* | 500 mg bid PO or 400 mg q12h IV | 5–7 days | |
| Azithromycin | 1 g/d PO | 10 days | |
| Fully susceptible | Ciprofloxacin or Ceftriaxone | As above | As above |
| Alt. | Amoxicillin / Chloramphenicol / TMP-SMX | Standard doses | 14–21 days |
| MDR | Ceftriaxone or Azithromycin | As above | As above |
| XDR (Pakistan) | Azithromycin | 1 g/d PO | 7 days |
| Meropenem | 1 g q8h IV | 10–14 days |
⚠ Fluoroquinolones should NOT be used empirically for travelers from South Asia due to high DSC/resistance rates.
— Harrison's 22E, p. 1361; Red Book 2021, p. 1023–1024
Special Situations
- Severe disease (delirium, shock, coma): add dexamethasone IV (3 mg/kg loading, then 1 mg/kg q6h × 8 doses = 48 hours)
- Relapse (up to 17% within 4 weeks): re-treat; azithromycin has lower relapse rates than fluoroquinolones or ceftriaxone
- Chronic carrier state (bile/gallbladder): 4 weeks oral ciprofloxacin or norfloxacin; cholecystectomy + antibiotics if medical therapy fails
Prevention
Vaccines
Two licensed vaccines are available:
| Vaccine | Type | Route | Efficacy | Duration |
|---|---|---|---|---|
| Ty21a (Vivotif) | Live attenuated | Oral, 4 capsules | ~50–80% | 5 years |
| Vi capsular polysaccharide (Typhim Vi) | Subunit | IM, single dose | ~50–70% | 2 years |
| Typhoid conjugate vaccine (TCV) | Vi-TT conjugate | IM, single dose | ~80–85% | Longer (preferred in children) |
- Neither vaccine protects against S. Paratyphi
- TCV (e.g., Typbar-TCV) is WHO-preferred for children; introduced widely in South Asia 2019–2022
- Vaccination recommended for travelers to endemic regions, household contacts of carriers, and lab personnel
Infection Control
- Standard + contact precautions; in children, continue until 3 negative stool cultures (≥48h after antibiotics)
- Exclusion from food handling until culture-negative
Key Points Summary
| Feature | Detail |
|---|---|
| Causative organism | S. Typhi, S. Paratyphi A/B/C |
| Incubation | 5–21 days (mean 10–14 days) |
| Classic triad | Prolonged fever + relative bradycardia + rose spots |
| Pathognomonic rash | Rose spots (salmon-colored, trunk/chest, 30%) |
| Feared complication | Intestinal perforation/hemorrhage (Week 3) |
| Best diagnostic test | Blood culture (week 1) / bone marrow culture |
| Drug of choice | Azithromycin or ceftriaxone (region-dependent) |
| XDR first-line | Azithromycin ± meropenem |
| Mortality (untreated) | 10–30%; treated <1% |
Recent evidence (2024–2025): A systematic review (PMID 39623850) found no significant difference in outcomes between ceftriaxone, fluoroquinolones, and azithromycin in RCTs — but most trials predate current resistance patterns. A 2024 systematic review (PMID 38579699) documented hemophagocytic lymphohistiocytosis as a rare but serious complication of enteric fever. Given XDR typhoid expansion from Pakistan, always check local resistance data before prescribing.
This is a shared conversation. Sign in to Orris to start your own chat.