Explain the mechanism of diabetic nephropathy vs membranous nephropathy vs minimal change disease vs other nephrotic syndromes
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
I have comprehensive content from multiple authoritative textbooks. Now let me compile the full answer with all the mechanisms, comparing each nephrotic syndrome in depth.
Nephrotic Syndromes: Mechanisms Compared
Overview: The Common Final Pathway
All nephrotic syndromes converge on podocyte injury as the central event causing proteinuria (>3.5 g/day), hypoalbuminemia, generalized edema, and hyperlipidemia/lipiduria. The mechanisms diverge sharply in how podocytes are damaged.
1. Diabetic Nephropathy
Category: Metabolic/hemodynamic — non-immune-mediated
Core Mechanism
Sustained hyperglycemia drives injury through four converging pathways:
| Pathway | Mechanism | Consequence |
|---|---|---|
| Advanced glycation end-products (AGEs) | Glucose non-enzymatically glycates GBM collagen and mesangial matrix proteins → cross-linking, reduced turnover | GBM thickening, mesangial expansion |
| Polyol pathway | Aldose reductase converts glucose → sorbitol → fructose; depletes NADPH and glutathione | Oxidative stress, endothelial dysfunction |
| PKC activation | Hyperglycemia activates protein kinase C → upregulates TGF-β, VEGF, plasminogen activator inhibitor-1 | Pro-fibrotic signaling, GBM thickening |
| RAAS overactivation | Intraglomerular hypertension → angiotensin II → mesangial expansion, TGF-β → fibrosis | Glomerulosclerosis; RAAS blockers are now standard therapy |
Hemodynamic Component
Hyperglycemia causes afferent arteriolar vasodilation (via tubuloglomerular feedback resetting, nitric oxide, and prostaglandins) → glomerular hyperfiltration → increased transcapillary pressure → mechanical stress on podocytes and GBM. This is the earliest detectable abnormality (microalbuminuria stage).
Genetic Susceptibility
The ACE gene insertion/deletion polymorphism (D allele → high ACE activity) and polymorphisms in IL-6, IL-10, ICAM1, and VCAM1 modulate susceptibility and progression. Only ~30–40% of type 2 diabetics develop clinically significant nephropathy, underscoring the importance of these modifiers. — Brenner and Rector's The Kidney
Morphology
- Light microscopy: Diffuse and nodular mesangial expansion; Kimmelstiel-Wilson nodules (pathognomonic); GBM thickening; hyaline arteriolosclerosis of both afferent and efferent arterioles (afferent-only involvement is typical of hypertension)
- Immunofluorescence: Linear IgG and albumin trapping in GBM (non-immune; due to increased permeability and protein insudation — not antibody-mediated)
- EM: GBM thickening, diffuse foot process effacement in advanced disease
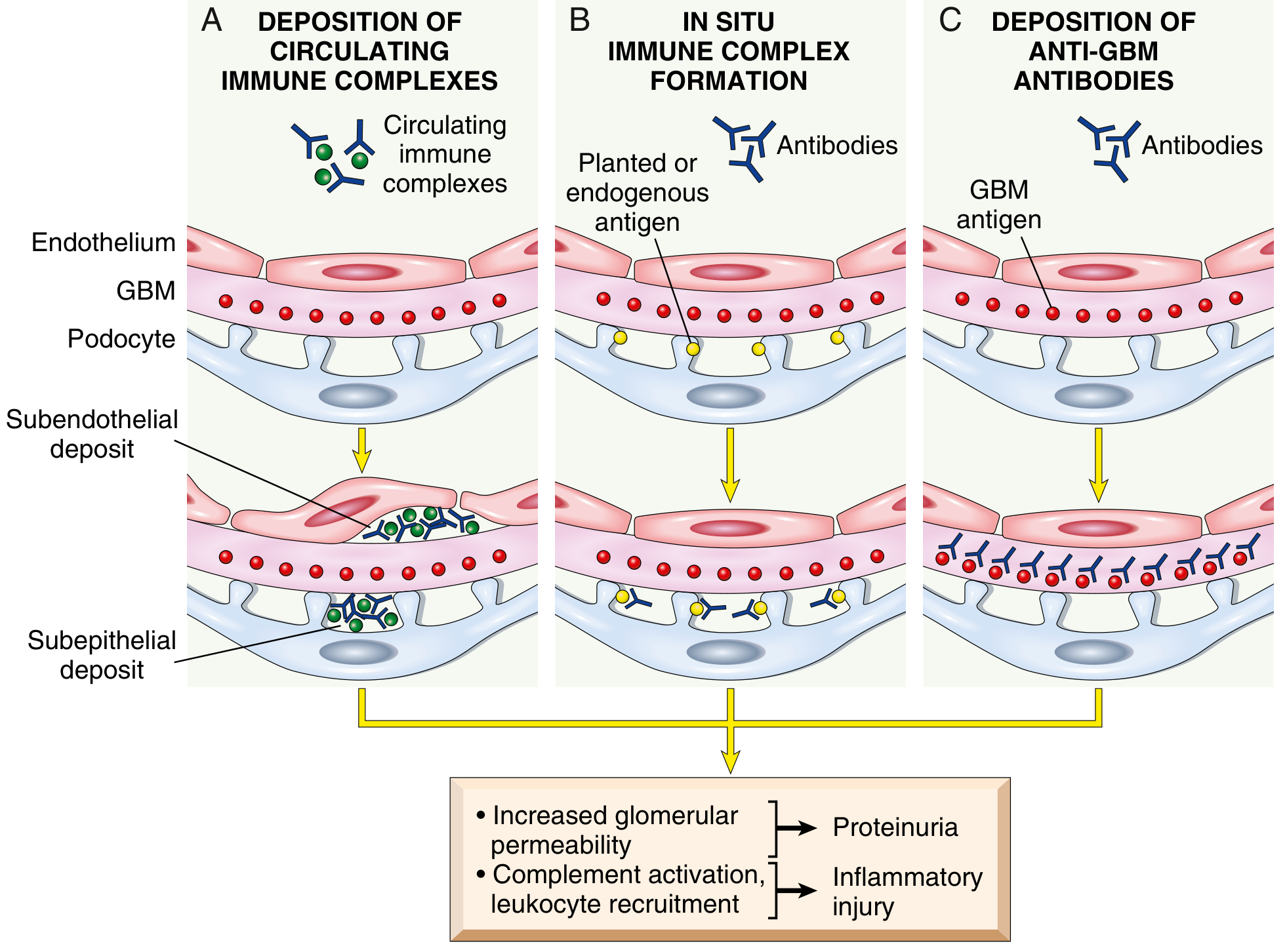
2. Membranous Nephropathy (MN)
Category: Autoimmune — in situ immune complex formation, subepithelial
Core Mechanism
The primary target in ~70% of idiopathic cases is M-type phospholipase A2 receptor (PLA2R), a transmembrane glycoprotein expressed on the outer surface of podocytes. IgG4 autoantibodies bind PLA2R in situ, forming immune complexes on the subepithelial aspect of the GBM — directly underneath the podocyte foot processes. — Comprehensive Clinical Nephrology, 7th Edition
Key features of the IgG4–PLA2R complex:
- IgG4 does not activate the classical complement pathway
- Instead, activates the lectin (MBL) complement pathway → generates membrane attack complex (C5b-9)
- C5b-9 inserts into the podocyte membrane → podocyte activation and injury → disruption of the slit diaphragm and cytoskeleton → proteinuria
- Importantly: little to no inflammation (no leukocyte infiltration), because IgG4 is non-inflammatory
Other antigens (less common): THSD7A (thrombospondin type-1 domain-containing 7A), EXT1/EXT2 (often associated with lupus/autoimmune disease), neutral endopeptidase (rare neonatal MN), bovine serum albumin.
Secondary Causes
Tumor cells can shed antigens that:
- Form circulating immune complexes → trapped in capillary wall → reform subepithelially
- Deposit directly in the subepithelial space where circulating antibody binds them
- Trigger oncogenic virus-related immune dysregulation
Malignancy-associated MN is distinguished by absence of anti-PLA2R antibodies, IgG1/IgG2 predominance (vs. IgG4 in primary), and >8 inflammatory cells/glomerulus on biopsy. — Brenner and Rector's The Kidney
Morphology
- Light microscopy: Diffuse GBM thickening; "spike and dome" pattern on silver stain (spikes = projections of new GBM matrix between deposits)
- Immunofluorescence: Granular IgG deposits along GBM in a subepithelial pattern ("lumpy-bumpy")
- EM: Electron-dense subepithelial deposits; foot process effacement
3. Minimal Change Disease (MCD)
Category: Podocytopathy — circulating factor-mediated (incompletely understood)
Core Mechanism
The exact trigger is unknown, but the leading hypothesis involves circulating molecules that injure podocytes and cause proteinuria through foot process effacement. Several candidates have been proposed:
| Candidate | Evidence |
|---|---|
| T cell-derived permeability factor | T cells are activated in MCD; T cell hybridomas secrete a factor causing heavy proteinuria in rats |
| IL-13 | Overexpressed by T cells in MCD; overexpression in rats causes histologic MCD |
| Angiopoietin-like 4 (ANGPTL4) | Overexpressed by podocytes in MCD; causes proteinuric response; reducible by steroids and N-acetyl-D-mannosamine |
| CD80 (B7.1) | Elevated in urine and podocytes; correlates with disease activity; normally expressed only on dendritic cells/B cells |
| Anti-nephrin antibodies | A subset of MCD patients have IgG antibodies against nephrin (slit diaphragm protein); recently recognized — Robbins, Cotran & Kumar |
Recent evidence also reveals mild glomerular endothelial injury with circulating endothelial biomarkers in most MCD patients, suggesting it is not a strict "pure podocytopathy" as historically considered. — Comprehensive Clinical Nephrology, 7th Edition
Why steroids work: Corticosteroids reduce ANGPTL4 overexpression and reverse foot process effacement; remission of proteinuria follows. This responsiveness is the clinical hallmark distinguishing MCD from most other nephrotic syndromes.
Morphology
- Light microscopy: Completely normal — hence "minimal change"
- Immunofluorescence: Negative — no immune deposits
- EM: Diffuse effacement (fusion) of podocyte foot processes ± vacuolization, microvillus formation — the only diagnostic abnormality
- Proximal tubular cells laden with protein droplets and lipid (reabsorption artifacts)
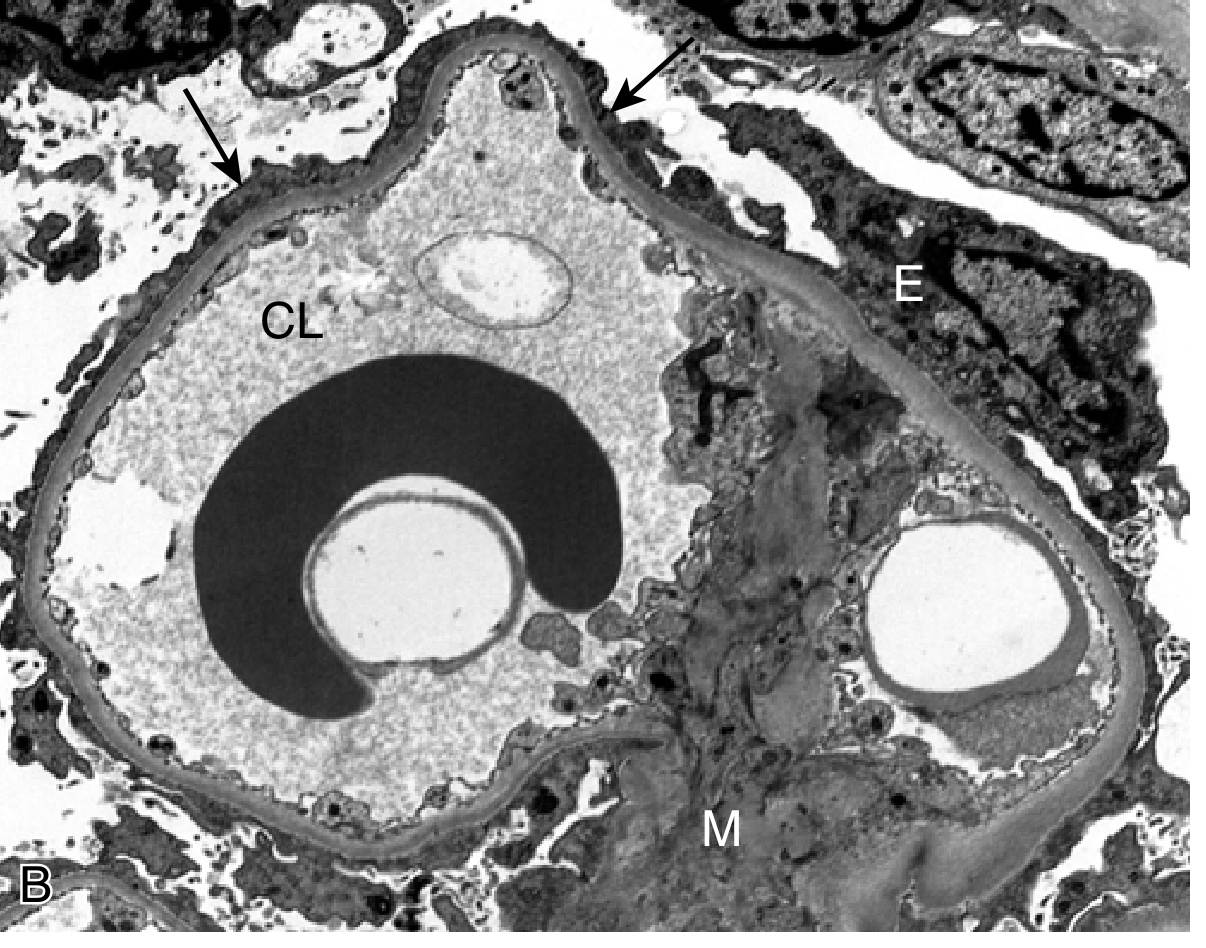
Clinical correlate: Proteinuria is selective (mainly albumin — smaller proteins lost preferentially), unlike FSGS where proteinuria is non-selective. >90% of children respond to a short corticosteroid course. — Robbins & Kumar Basic Pathology
4. Focal Segmental Glomerulosclerosis (FSGS)
Category: Podocytopathy — primary (circulating factor) or secondary
Core Mechanism
Podocyte injury is the initiating event. Podocytes cannot divide; once lost, adjacent podocytes must cover greater GBM surface area → foot process effacement → further loss → denuded GBM → protein and lipid entrapment → ECM deposition → glomerulosclerosis.
Primary FSGS: A circulating permeability factor causes podocyte injury. Cardiotrophin-like cytokine 1 (CLC-1) is one candidate, but others are under investigation. Rapid recurrence of proteinuria after renal transplantation (sometimes within hours) provides strong evidence for a circulating mediator. — Comprehensive Clinical Nephrology, 7th Edition
Genetic FSGS: Mutations in >60 podocyte-related genes including nephrin (NPHS1), podocin (NPHS2), and cytoskeletal proteins. APOL1 polymorphisms (high-risk alleles G1/G2) markedly increase susceptibility in people of African descent.
Secondary FSGS causes:
- Nephron loss → glomerular hypertension (maladaptive hyperfiltration)
- Obesity (same mechanism — hyperfiltration)
- HIV (collapsing variant — HIVAN) and COVID-19 (COVAN) — especially in APOL1 high-risk individuals
- Heroin and some therapeutic drugs
Collapsing FSGS Variant
Podocyte loss is so rapid that the glomerular tuft collapses entirely; parietal epithelial cells migrate onto the denuded tuft. Associated with APOL1 high-risk alleles, HIVAN, COVAN, and pamidronate use. Carries a particularly poor prognosis.
Morphology
- Light microscopy: Focal (some glomeruli) and segmental (part of tuft) obliteration of capillary loops; mesangial matrix increase; hyalinosis (IgM and protein insudation); foamy macrophages
- Immunofluorescence: Non-specific IgM and C3 trapping in sclerotic areas
- EM: Diffuse foot process effacement (in primary, acute-phase FSGS); focal podocyte detachment with GBM denudation
vs. MCD: Both show foot process effacement on EM with negative IF, but FSGS has segmental sclerosis on light microscopy; FSGS is non-selective proteinuria; steroid-resistant; >50% progress to ESKD within 10 years. — Robbins & Kumar Basic Pathology
5. Membranoproliferative GN (MPGN)
Category: Immune complex deposition — both mesangial and subendothelial
Mechanism
Immune complexes deposit in both the mesangium and the subendothelial space → local complement activation → C5a generation → leukocyte (neutrophil, monocyte, platelet) recruitment → oxidant release, protease secretion (elastase, cathepsin G, metalloproteinases) → GBM degradation and cellular injury. Upregulation of E/P-selectin, ICAM-1, and proinflammatory cytokines (IL-1, TNF-α) amplify the response.
Common associations: HCV with cryoglobulins (IgM rheumatoid factor binding IgG in the complex), lupus nephritis (nucleosome binding facilitates complex deposition), SBE, other chronic infections. — Comprehensive Clinical Nephrology, 7th Edition
Morphology: Mesangial and endothelial cell proliferation; "tram-track" GBM duplication (new GBM laid around subendothelial deposits); granular IF with C3, IgG, IgM.
6. C3 Glomerulopathy (Dense Deposit Disease / C3GN)
Category: Alternative complement pathway dysregulation — no immune complexes
Mechanism
Spontaneous intraglomerular activation of the alternative complement pathway due to:
- C3 nephritic factor (C3NeF): autoantibody that binds and stabilizes C3bBb (the alternative pathway C3 convertase) → persistent C3 activation → hypocomplementemia (low C3; normal C1, C4)
- Factor H mutations: loss of the main inhibitor of the alternative pathway
Result: C3 deposition without IgG; in Dense Deposit Disease, the lamina densa of the GBM is permeated by a ribbonlike, homogeneous, extremely electron-dense material. — Robbins, Cotran & Kumar
7. Amyloidosis
Category: Protein deposition — non-immune, metabolic/dysproteinemia
Mechanism
Misfolded proteins (AL amyloid from immunoglobulin light chains in plasma cell dyscrasias; AA amyloid from serum amyloid A in chronic inflammation) deposit in the mesangium and along capillary walls as β-pleated sheet fibrils. These fibrils disrupt the filtration barrier physically. No immune activation or podocyte signaling is primarily involved.
Diagnosis: Congo red staining with apple-green birefringence under polarized light; EM shows 8–10 nm fibrils in a random arrangement.
Summary Comparison Table
| Feature | Diabetic Nephropathy | Membranous Nephropathy | Minimal Change Disease | FSGS |
|---|---|---|---|---|
| Primary mechanism | Metabolic/hemodynamic (AGEs, PKC, RAAS) | Anti-PLA2R IgG4 → subepithelial immune deposits → C5b-9 | Circulating factor (IL-13, ANGPTL4, anti-nephrin?) | Circulating permeability factor / genetic podocyte mutation |
| Podocyte injury | Indirect (metabolic stress, glomerular hypertension) | C5b-9 insertion into podocyte | Direct (unknown factor) | Direct loss → denuded GBM |
| Light microscopy | Kimmelstiel-Wilson nodules, GBM thickening | GBM thickening, "spikes" | Normal | Focal, segmental sclerosis |
| Immunofluorescence | Linear IgG/albumin (non-specific trapping) | Granular subepithelial IgG | Negative | Non-specific IgM/C3 in sclerotic areas |
| EM | GBM thickening, foot process effacement (late) | Subepithelial electron-dense deposits | Diffuse foot process effacement only | Foot process effacement + podocyte detachment |
| Proteinuria type | Non-selective (advanced disease) | Non-selective | Selective (albuminuria) | Non-selective |
| Steroid response | None | Variable/resistant (anti-CD20 preferred for primary MN) | Excellent (>90% children) | Generally poor |
| Complement | Normal | Normal | Normal | Normal |
| Key biomarker | HbA1c; GFR trajectory | Anti-PLA2R antibody titer | — | APOL1 genotype (risk) |
Sources: Robbins & Kumar Basic Pathology (9780323790185); Robbins, Cotran & Kumar Pathologic Basis of Disease (9780443264528); Comprehensive Clinical Nephrology, 7th Edition (9780323825924); Brenner and Rector's The Kidney (9780323532655)
Convert each and everything to diagram and sketch
Generating Image
Generating Image
Generating Image
Generating Image
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.