Answer all the questions in accordance to MBBS 1st year professional university exam
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Searching Images
eosinophil stained microscopic slide bilobed nucleus
Searching Images
neutrophil phagocytosis steps bacteria
Reading File
Reading File
Searching Images
eosinophil blood smear bilobed nucleus eosinophilic granules
Searching Images
ABO blood group typing transfusion reaction immunology
Searching Images
hemostasis three steps vascular spasm platelet plug coagulation
I now have comprehensive textbook material to answer all questions. Let me compile a thorough, exam-ready answer.
MBBS 1st Year Professional Exam — Model Answers
LONG ESSAY (10 Marks)
Q1. Neutrophils in the Immune System & Steps of Phagocytosis (4 marks)
Introduction
Neutrophils (polymorphonuclear leukocytes / PMNs) are the most abundant circulating white blood cells (55–70% of total WBCs). They are the first responders of the innate immune system, arriving at sites of infection within minutes.
Main Functions of Neutrophils
- Phagocytosis — engulf and destroy bacteria and fungi
- Degranulation — release antimicrobial enzymes (elastase, myeloperoxidase, defensins) from primary (azurophilic) and secondary (specific) granules
- Respiratory burst — generate reactive oxygen intermediates (ROIs) via NADPH oxidase to kill ingested pathogens
- NET formation — release Neutrophil Extracellular Traps (webs of DNA + antimicrobial proteins) to trap pathogens extracellularly
- Chemokine secretion — amplify and coordinate the inflammatory response
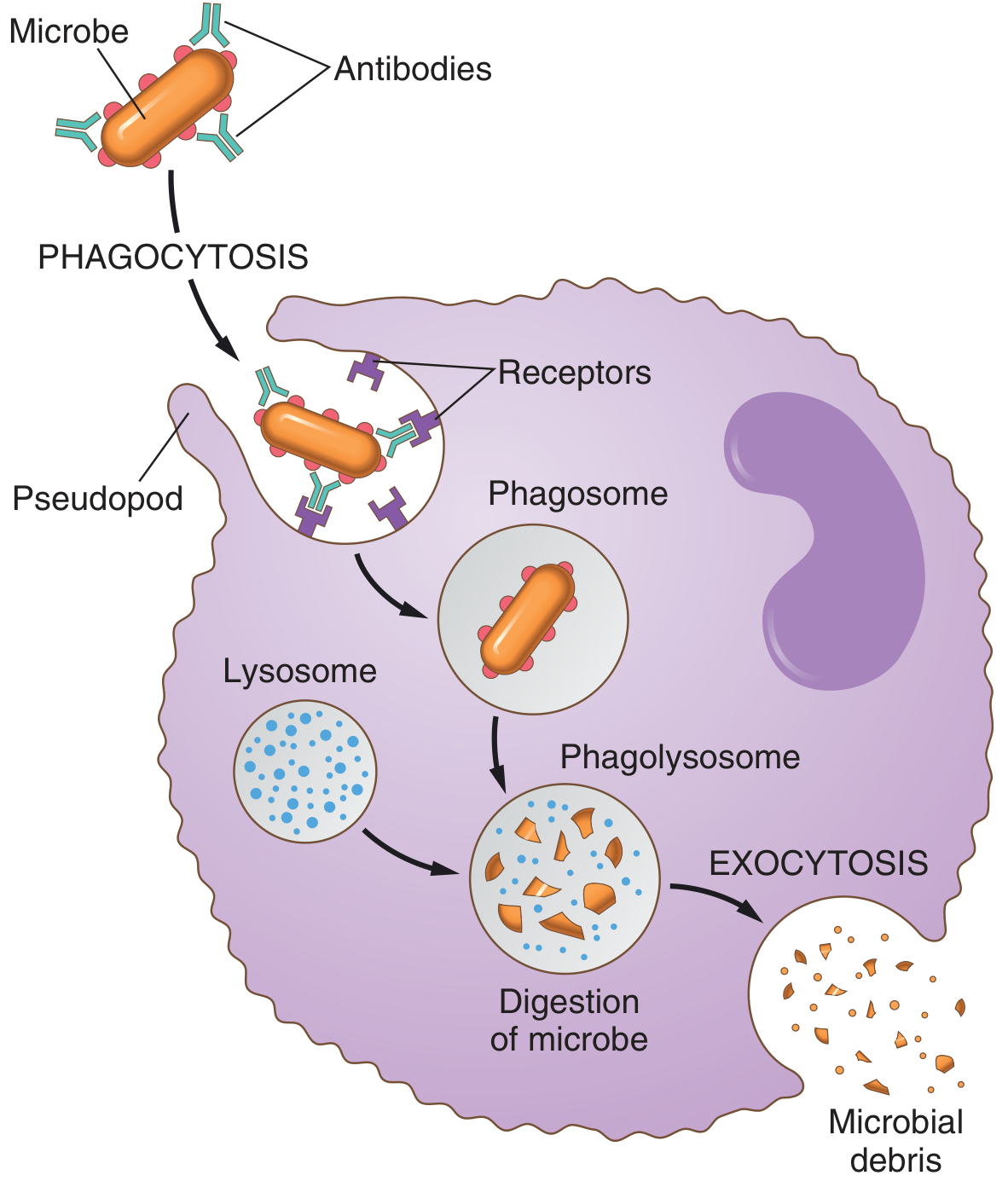
Steps Involved in Phagocytosis of Bacteria/Fungi
Step 1 — Margination & Diapedesis
In response to inflammatory signals, neutrophils marginate (roll along the endothelium via selectins), firmly adhere (via integrins), then squeeze through endothelial junctions into tissue — a process called diapedesis/extravasation. Both paracellular (through intercellular gaps) and transcellular (through the endothelial cell body) pathways are used.
Step 2 — Chemotaxis
Neutrophils migrate toward the infection site along a chemical gradient of chemoattractants: bacterial products, complement fragments (C3a, C5a), and cytokines (IL-8). This directional movement is called chemotaxis.
Step 3 — Recognition & Opsonisation
Neutrophils express surface receptors that recognize pathogens:
- Fc receptors — bind IgG antibodies coating the bacteria
- Complement receptors (CR3) — bind C3b opsonin on the bacterial surface
- Toll-like receptors (TLRs) — directly recognize pathogen-associated molecular patterns (PAMPs)
Opsonisation (coating bacteria with IgG and C3b) markedly enhances phagocytic efficiency.
Step 4 — Engulfment (Phagosome Formation)
The neutrophil attaches to the opsonised bacterium and extends pseudopodia that surround it on all sides. The pseudopodia fuse, enclosing the organism in a phagosome (phagocytic vesicle). A single neutrophil can phagocytose 3–20 bacteria before it dies.
Step 5 — Phagolysosome Formation & Killing
Primary granules (lysosomes) fuse with the phagosome to form a phagolysosome. Killing occurs via:
- Oxygen-dependent mechanisms:
- NADPH oxidase / phox system → generates superoxide anions (O₂⁻) → converted to H₂O₂ and hydroxyl radicals
- Myeloperoxidase (MPO) system → converts H₂O₂ + Cl⁻ → hypochlorous acid (HOCl, ~1000× more bactericidal than H₂O₂)
- Oxygen-independent mechanisms:
- Lysozyme (digests bacterial cell wall)
- Defensins (membrane-disrupting peptides)
- Lactoferrin (iron chelator, bacteriostatic)
- Elastase and cathepsin G (proteolytic enzymes)
Step 6 — Exocytosis
Digested products are expelled from the cell by exocytosis. Spent neutrophils undergo apoptosis and are cleared by macrophages.
Q1(a). Five Main Types of Leukocytes — Granulocytes vs Agranulocytes (3 marks)
Leukocytes are classified into two groups based on the presence or absence of cytoplasmic granules visible on light microscopy:
| Category | Cell Type | % of WBC | Key Features |
|---|---|---|---|
| Granulocytes | Neutrophils | 55–70% | Multi-lobed nucleus (2–5 lobes), pale granules; phagocytosis |
| Eosinophils | 1–4% | Bi-lobed nucleus, large bright pink/red granules; anti-parasitic, allergic reactions | |
| Basophils | 0.5–1% | Bi-lobed (often obscured), large blue-purple granules; IgE-mediated allergy, contain histamine | |
| Agranulocytes / Mononuclear | Lymphocytes | 20–35% | Large round nucleus, scant cytoplasm; T cells, B cells, NK cells — adaptive immunity |
| Monocytes | 2–8% | Kidney-shaped nucleus, grey cytoplasm; become tissue macrophages; phagocytosis, antigen presentation |
Memory aid: "Never Let Monkeys Eat Bananas" → Neutrophils, Lymphocytes, Monocytes, Eosinophils, Basophils
Q1(b). Diagram of Eosinophil & Role Against Parasites / Allergic Reactions (3 marks)
Diagram of Eosinophil on Stained Microscopic Slide
EOSINOPHIL ON H&E STAIN
╔═══════════════════════════╗
║ ●●●●●●●●●●●●●●●●●●●●● ║ ← Large bright pink/red
║ ●●●●●●●●●●●●●●●●●●●●● ║ (eosinophilic) granules
║ ●● ┌─────┐ ●●●● ┌────┐ ●║
║ ●● │ N │ ●●●● │ N │ ●║ ← Bi-lobed nucleus
║ ●● │lobe1│ ●●●● │lob2│ ●║ (connected by thin strand)
║ ●● └──┬──┘ ●●●● └─┬──┘ ●║
║ ●●●●●│●●●●●●●●●●│●●●●●●║
║ ●●●●●╰────────────╯●●●●●║ ← Thin chromatin bridge
╚═══════════════════════════╝
Key: N = nucleus lobe, ● = eosinophilic (pink) granules
Size: ~12–17 µm diameter
Microscopic features:
- Nucleus: Characteristically bi-lobed (two lobes connected by a thin chromatin bridge)
- Cytoplasm: Filled with large, coarse, bright pink-orange (eosinophilic) granules on H&E stain
- Granule contents: Major basic protein (MBP), eosinophil cationic protein (ECP), eosinophil-derived neurotoxin (EDN), eosinophil peroxidase
- Size: 12–17 µm
Role Against Multicellular Parasites
Multicellular helminths (e.g., Schistosoma, Ascaris, Trichinella) are too large to be phagocytosed. Eosinophils are uniquely equipped to destroy them via:
- ADCC (Antibody-Dependent Cellular Cytotoxicity): IgE and IgG antibodies on the parasite surface bind to eosinophil Fc receptors → activates degranulation
- Degranulation releases:
- Major Basic Protein (MBP) — toxic to larval surface membranes
- Eosinophil Cationic Protein (ECP) — ribonuclease activity, disrupts parasite tegument
- Eosinophil Peroxidase — generates reactive oxygen species → oxidative killing
- Signal of infection: peripheral blood eosinophilia is a hallmark of helminth infestation
Role in Allergic Reactions
In IgE-mediated (Type I) hypersensitivity:
- Mast cells and basophils degranulate → release histamine, leukotrienes, and eosinophil chemotactic factor of anaphylaxis (ECF-A)
- ECF-A recruits eosinophils to the site (airways in asthma, nasal mucosa in rhinitis, skin in eczema)
- Eosinophils release MBP and ECP → amplify the inflammatory response → epithelial damage
- They also produce Platelet Activating Factor (PAF) and Leukotriene C4 (LTC4) → bronchoconstriction
Eosinophilia (>0.5 × 10⁹/L) is the hallmark of both allergic diseases and helminthic infections.
SHORT ESSAYS (4 marks each)
Short Essay 1. Define Homeostasis & Describe Feedback Mechanisms
Definition
Homeostasis is the maintenance of a relatively stable internal environment (milieu intérieur) within physiological limits, despite continuous changes in the external environment. The term was coined by Walter Cannon (1926), building on Claude Bernard's concept of milieu intérieur.
Examples of controlled parameters: blood glucose (70–100 mg/dL), body temperature (37°C ± 0.5°C), arterial pH (7.35–7.45), serum K⁺, plasma osmolality.
Components of a Homeostatic Control System
Every homeostatic mechanism has four components:
- Stimulus — deviation of the variable from the set point
- Receptor (sensor) — detects the deviation and sends information to the control centre
- Control centre (integrating centre) — compares input with the set point and initiates an appropriate response
- Effector — produces the corrective response
Types of Feedback Mechanisms
1. Negative Feedback (most common)
The response opposes or reverses the original stimulus, restoring the variable toward the set point.
Example: Thermoregulation
- Body temp rises → detected by hypothalamic thermoreceptors → effectors (sweat glands, vasodilation) activated → heat loss → temperature returns to 37°C
Example: Blood glucose regulation
- Blood glucose rises → pancreatic β-cells secrete insulin → glucose uptake by cells → blood glucose falls back to normal
The system is characterized by a feedback loop that continuously monitors and corrects deviations.
2. Positive Feedback (less common, amplifies the response)
The response reinforces or amplifies the original stimulus — moves the variable further from the set point until a definitive endpoint is reached.
Example: Parturition (childbirth)
- Uterine stretching → oxytocin release → stronger contractions → more stretching → more oxytocin → cycle ends at delivery
Example: Blood clotting (coagulation cascade)
- Platelet adhesion → ADP release → recruits more platelets → plug grows → culminates in clot formation
Example: Action potential (all-or-nothing)
- Na⁺ influx → depolarization → more Na⁺ channels open → more influx → completed in milliseconds
3. Feed-Forward Mechanism (anticipatory)
The body prepares for a change before it occurs, based on prediction.
Example: Increase in heart rate and breathing rate before exercise begins (conditioned anticipatory response)
Short Essay 2. Primary & Secondary Active Transport with Examples
Active Transport — Definition
Active transport is the movement of molecules against their electrochemical gradient (from lower to higher concentration), requiring energy expenditure in the form of ATP.
Primary Active Transport
Definition: Uses ATP hydrolysis directly to drive the transport protein (pump) to move ions or molecules across the membrane.
Mechanism:
- Transport protein (ATPase pump) binds ATP → undergoes conformational change → translocates ion across the membrane against its gradient → ATP is hydrolyzed to ADP + Pᵢ
Key Examples:
| Pump | Location | Action |
|---|---|---|
| Na⁺/K⁺-ATPase | All cell membranes | Pumps 3 Na⁺ out and 2 K⁺ in per cycle; maintains resting membrane potential |
| Ca²⁺-ATPase (SERCA) | Sarcoplasmic reticulum of muscle | Pumps Ca²⁺ back into SR after muscle contraction |
| H⁺/K⁺-ATPase (Proton pump) | Gastric parietal cells | Pumps H⁺ out to acidify gastric lumen (target of PPIs) |
| H⁺-ATPase | Renal tubules, lysosomes | Acidifies urine and lysosome lumen |
Most important: Na⁺/K⁺-ATPase — uses ~30% of all cellular ATP; critical for excitability of neurons and muscle cells.
Secondary Active Transport
Definition: Uses the electrochemical gradient established by primary active transport (especially the Na⁺ gradient) as the energy source. ATP is not directly consumed — it is indirectly used (via the Na⁺ gradient created by Na⁺/K⁺-ATPase).
Mechanism: Transport protein (co-transporter or exchanger) couples the movement of a favored ion (Na⁺, moving down its gradient) to the simultaneous movement of another molecule against its gradient.
Two subtypes:
- Symport (co-transport): Both substances move in the same direction
- Antiport (counter-transport/exchange): Substances move in opposite directions
Key Examples:
| Transporter | Type | Location | Action |
|---|---|---|---|
| SGLT1 (Na⁺-glucose cotransporter) | Symport | Small intestinal epithelium & renal tubule | Na⁺ entry drives glucose absorption against gradient |
| Na⁺-amino acid cotransporter | Symport | Intestinal epithelium | Absorbs amino acids |
| Na⁺/Ca²⁺ exchanger (NCX) | Antiport | Cardiac muscle | 3 Na⁺ in → 1 Ca²⁺ out; reduces intracellular Ca²⁺ |
| Na⁺/H⁺ exchanger (NHE) | Antiport | Renal proximal tubule | Na⁺ in → H⁺ out; regulates acid-base balance |
| Na⁺-K⁺-2Cl⁻ cotransporter (NKCC2) | Symport | Loop of Henle (thick ascending limb) | Target of loop diuretics (furosemide) |
Summary Comparison
| Feature | Primary Active Transport | Secondary Active Transport |
|---|---|---|
| Energy source | ATP (direct) | Ion gradient (indirect ATP) |
| Protein type | ATPase pump | Cotransporter / exchanger |
| Coupled to | ATP hydrolysis | Na⁺ (usually) gradient |
| Example | Na⁺/K⁺-ATPase | SGLT1, Na⁺/Ca²⁺ exchanger |
Short Essay 3. Three Main Steps of Hemostasis (Clotting Disorder)
In a patient suspected of having a clotting disorder, understanding normal hemostasis is essential. Hemostasis has three main sequential steps:
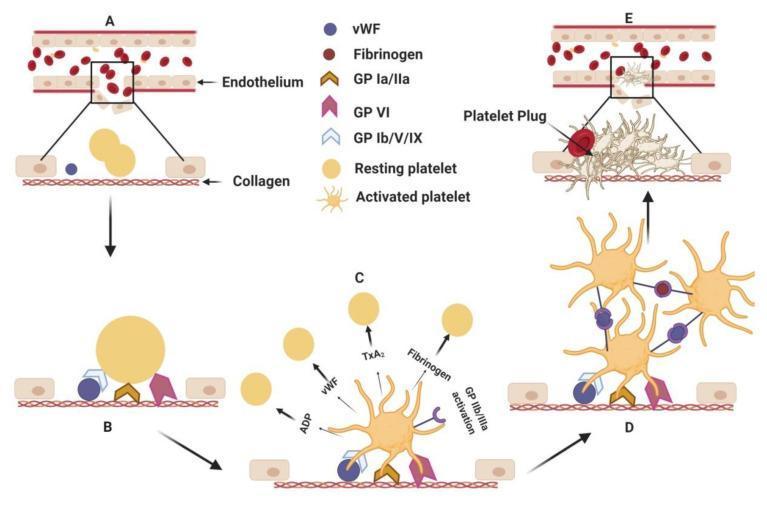
Step 1 — Vascular Spasm (Primary Response)
- Immediate response (seconds) to vessel injury
- Smooth muscle of the vessel wall contracts → vasoconstriction → reduces blood flow through the damaged vessel
- Triggered by: direct smooth muscle damage, nervous reflexes, local chemical mediators (endothelin, thromboxane A₂)
- Components: vascular smooth muscle
Step 2 — Primary Hemostasis — Platelet Plug Formation
- Occurs over 1–3 minutes
- Platelet Adhesion: Exposed subendothelial collagen and von Willebrand Factor (vWF) bind platelet receptors (GP Ib-IX-V for vWF; GP Ia/IIa and GP VI for collagen)
- Platelet Activation: Binding activates platelets → shape change (disc → spiky sphere) → release of granule contents:
- Dense granules: ADP, serotonin, Ca²⁺
- Alpha granules: fibrinogen, vWF, platelet factor 4, P-selectin
- ADP and TXA₂ (thromboxane A₂) recruit additional platelets
- Platelet Aggregation: GP IIb/IIIa receptors on activated platelets bind fibrinogen (and vWF) as bridges between adjacent platelets → formation of the primary platelet plug (loose, temporary)
- Components: platelets, vWF, fibrinogen, collagen
Step 3 — Secondary Hemostasis — Coagulation Cascade → Clot Formation
- Stabilises the platelet plug with a fibrin mesh (over 3–6 minutes)
- The coagulation cascade is triggered by:
- Extrinsic pathway: Tissue factor (TF / thromboplastin) released from damaged cells + Factor VII → TF-VIIa complex → activates Factor X
- Intrinsic pathway: Exposed collagen activates Factor XII → cascade through XI → IX → VIII → X
- Common pathway: Factor Xa + Va (prothrombinase complex) → converts prothrombin → thrombin → converts fibrinogen → fibrin monomers → fibrin polymers cross-linked by Factor XIII → stable clot
- Thrombin is the key enzyme: activates more clotting factors (positive feedback), activates platelets, and activates fibrinogen
| Step | Timing | Key Components | Endpoint |
|---|---|---|---|
| 1. Vascular spasm | Seconds | Smooth muscle, endothelin, TXA₂ | Vasoconstriction |
| 2. Platelet plug | 1–3 min | Platelets, vWF, collagen, GP IIb/IIIa, ADP, TXA₂ | Primary platelet plug |
| 3. Coagulation cascade | 3–6 min | Clotting factors, TF, Ca²⁺, thrombin, fibrin | Stable fibrin clot |
Clinical note: Defects in Step 2 (e.g., von Willebrand disease, thrombocytopenia) prolong the bleeding time. Defects in Step 3 (e.g., haemophilia A/B — Factor VIII/IX deficiency) prolong the aPTT/PT.
Short Essay 4. Blood Typing — ABO & Rh Systems in Transfusion
Relevance of Blood Typing in Transfusion
Blood typing is essential before any transfusion to prevent haemolytic transfusion reactions (HTRs) — potentially fatal immunological events triggered by incompatible blood.
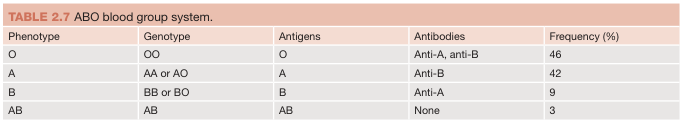
ABO Blood Group System
The ABO system is based on the presence or absence of A and B antigens (glycoproteins / glycolipids) on the red blood cell surface.
| Blood Group | Antigens on RBC | Antibodies in Plasma | Can donate to | Can receive from |
|---|---|---|---|---|
| A | A antigen | Anti-B (IgM) | A, AB | A, O |
| B | B antigen | Anti-A (IgM) | B, AB | B, O |
| AB | A + B antigens | Neither | AB only | A, B, AB, O (universal recipient) |
| O | Neither | Anti-A + Anti-B | A, B, AB, O (universal donor) | O only |
Key feature: ABO antibodies are natural (pre-formed/innate) — they develop within 6 months of birth without prior exposure to foreign RBCs, due to cross-reactivity with gut bacteria antigens.
Rh (Rhesus) Blood Group System
- Based on the D antigen (most immunogenic): individuals are Rh-positive (Rh⁺) if D antigen is present, Rh-negative (Rh⁻) if absent
- ~85% of the population is Rh⁺
- Unlike ABO: Rh antibodies are immune (acquired) — only produced after exposure to Rh-positive blood (via transfusion or pregnancy)
Immunological Reaction with Incompatible Blood
Mechanism of Acute Haemolytic Transfusion Reaction (AHTR):
- Recipient has pre-formed antibodies against donor RBC antigens (e.g., type A recipient receiving type B blood → anti-B IgM attacks donor B cells)
- Complement activation (classical pathway): Antigen-antibody complexes activate C1 → C3 → C5 → Membrane Attack Complex (MAC)
- Intravascular haemolysis: MAC punches holes in RBC membranes → immediate haemolysis → release of haemoglobin → haemoglobinaemia, haemoglobinuria
- Cytokine storm: Anaphylatoxins C3a and C5a → mast cell degranulation → histamine → hypotension, bronchospasm
- DIC (disseminated intravascular coagulation) may ensue
Clinical features of AHTR:
- Fever, chills, back/flank pain, hypotension
- Haemoglobinuria (red/cola-coloured urine)
- Acute kidney injury (haemoglobin tubular toxicity)
- DIC, shock — can be fatal
Rh Incompatibility — Haemolytic Disease of the Newborn (HDN):
- Rh⁻ mother carrying Rh⁺ fetus → at delivery, fetal RBCs enter maternal circulation → mother forms anti-D IgG
- In subsequent Rh⁺ pregnancies: maternal anti-D IgG crosses the placenta → destroys fetal RBCs → erythroblastosis fetalis (hydrops fetalis in severe cases)
- Prevented by anti-D immunoglobulin (Rhogam) given to Rh⁻ mothers at 28 weeks and within 72 hours of delivery
Prevention
- Pre-transfusion testing: ABO grouping, Rh typing, antibody screening, cross-matching
- Universal donor for emergency: O-negative packed RBCs
- Universal plasma donor: AB plasma
Summary of Mark Allocation
| Question | Topic | Marks |
|---|---|---|
| Long Essay Q1 | Neutrophil functions & phagocytosis steps | 4 |
| Long Essay Q1(a) | 5 leukocytes — granulocytes vs agranulocytes | 3 |
| Long Essay Q1(b) | Eosinophil diagram + parasite/allergy role | 3 |
| Short Essay 1 | Homeostasis + feedback mechanisms | 4 |
| Short Essay 2 | Primary & secondary active transport | 4 |
| Short Essay 3 | Three steps of hemostasis | 4 |
| Short Essay 4 | Blood typing ABO/Rh + transfusion reactions | 4 |
| Total | 26/30 (Short Essay 5 not provided in paper) |
Sources: Guyton & Hall Textbook of Medical Physiology; Histology: A Text and Atlas (Ross & Pawlina); Medical Physiology (Boron & Boulpaep); Harrison's Principles of Internal Medicine, 21st Ed; Bailey & Love's Surgery, 28th Ed.
This is a shared conversation. Sign in to Orris to start your own chat.