HARRISOM S AUREUS LESSPM NOTES
Reading File
Finding Sources
Searching the Web
Staphylococcus aureus Harrison's Principles microbiology key points
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive Harrison's 22E content on S. aureus. Let me compile the full lesson notes.
📚 Staphylococcus aureus — Lesson Notes
Source: Harrison's Principles of Internal Medicine, 22nd Edition (2025), Chapter 152 — Franklin D. Lowry & Anne-Catrin Uhlemann
1. INTRODUCTION
S. aureus is the most virulent of ≥40 staphylococcal species. It is a pluripotent pathogen — causes disease through both toxin-mediated and non-toxin-mediated mechanisms. Despite effective antibiotics, it remains a leading cause of global morbidity and mortality.
2. MICROBIOLOGY & TAXONOMY
| Feature | Detail |
|---|---|
| Gram stain | Gram-positive cocci in grape-like clusters |
| Size | ~1 μm diameter |
| Catalase | Positive (distinguishes from Streptococci) |
| Motility | Non-motile |
| Oxygen | Aerobic & facultatively anaerobic |
| Key marker | Coagulase-positive (converts fibrinogen → fibrin) |
| Other markers | Protein A positive, DNAse positive, ferments mannitol |
| Colonies | Golden, β-hemolytic on blood agar |
| Survival | Prolonged survival on environmental surfaces |
📌 Note: S. pseudintermedius and S. argenteus are also coagulase-positive but are non-S. aureus staphylococci (NSaS). The older term "coagulase-negative staphylococci" is being replaced by NSaS (non-S. aureus staphylococci).
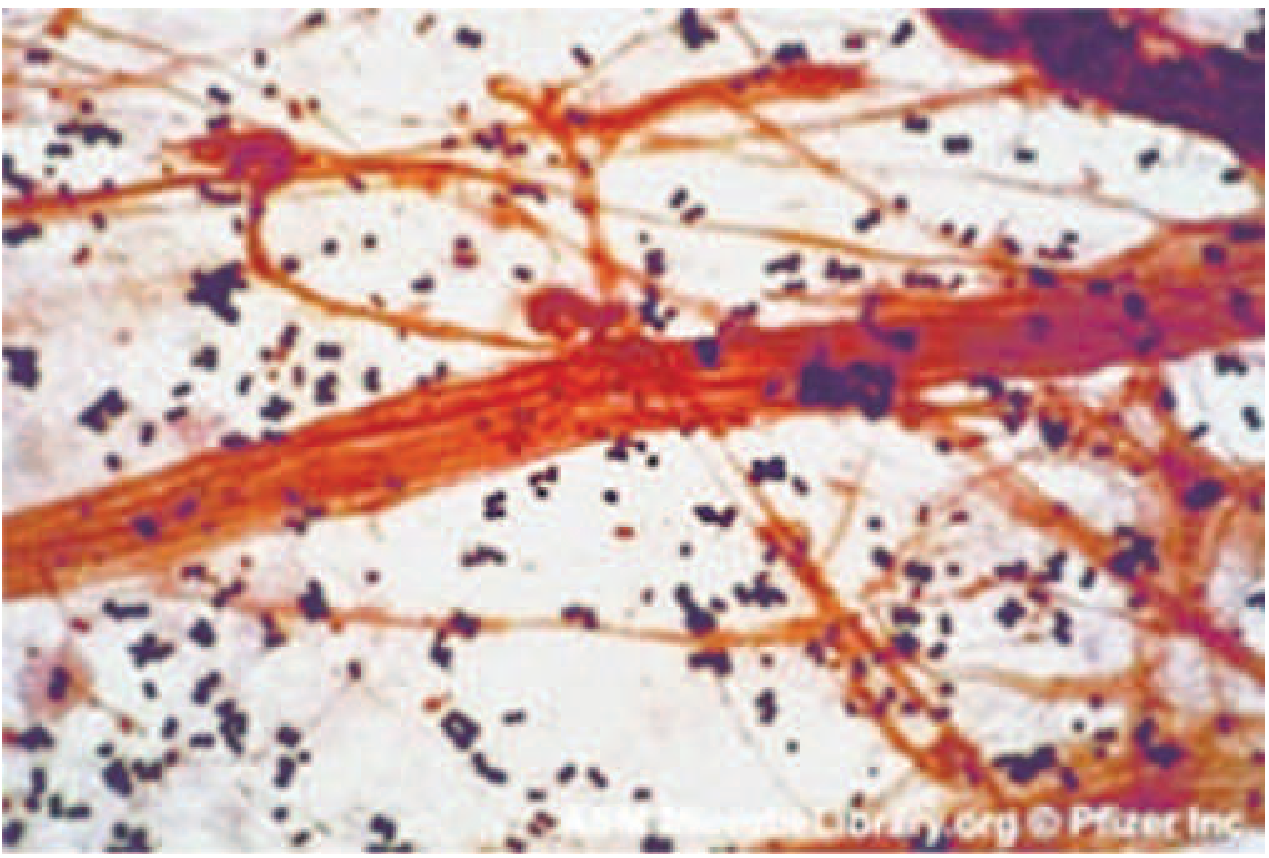
Identification Methods
- Latex kits detecting protein A + clumping factor
- MALDI-TOF mass spectrometry (increasingly used for speciation)
- Molecular typing: pulsed-field gel electrophoresis (PFGE), SpA typing
- Whole-genome sequencing = gold standard for outbreak discrimination
3. EPIDEMIOLOGY
- Normal flora on skin, anterior nares, oropharynx, and vagina
- ~30% of individuals are colonized at any one time
- Most infections arise from the patient's own commensal flora (autoinfection)
- Transmission: mainly direct personal contact with infected site; rarely aerosols
High-Risk Groups
- Diabetes mellitus (↑ colonization + insulin injections + impaired leukocyte function)
- Neutropenia (chemotherapy)
- Chronic granulomatous disease (CGD)
- Hyper-IgE (Job syndrome), Chédiak-Higashi syndrome
- End-stage renal disease, HIV infection
- Patients with prosthetic devices, intravascular catheters, surgical wounds
- Injection drug users (IDU) — especially infective endocarditis
MRSA Epidemiology
- Hospital-acquired MRSA (HA-MRSA): Nosocomial, often multidrug resistant
- Community-acquired MRSA (CA-MRSA): Established community pathogen since 1990s
- Most common cause of community-acquired SSTIs
- Risk factors: poor hygiene, close contact, contaminated material, broken skin
- Groups: children, prisoners, athletes, Native Americans, IDU
- Dominant US clone: ST8 / USA300
- 5–10% of CA-MRSA infections are invasive; can affect immunocompetent individuals
4. PATHOGENESIS
General Concept
S. aureus is a pyogenic pathogen par excellence — known for causing abscess formation at local and distant (metastatic) sites. Response: intense PMN infiltration → macrophage/fibroblast infiltration → fibrinous capsule OR spread to bloodstream.
Virulence Factors
A. Structural/Surface Components
| Factor | Function |
|---|---|
| Peptidoglycan | Thick cell wall — allows survival on dry surfaces; triggers cytokine release |
| Teichoic acids | Adherence to nasal epithelium; activates complement |
| Capsular polysaccharide | Antiphagocytic |
| Protein A (SpA) | Binds IgG Fc region → blocks opsonophagocytosis; key surface marker |
| Clumping factor | Fibrinogen binding → clumping in blood |
| MSCRAMMs (e.g., fibronectin-binding proteins) | Adhesion to host tissues and prosthetic devices |
| Biofilm | Critical for device-related infections |
B. Secreted Toxins
| Toxin | Disease | Mechanism |
|---|---|---|
| α-Toxin (alpha-hemolysin) | Tissue destruction, hemolysis | Pore-forming; lyses RBCs, platelets, leukocytes |
| β, γ, δ toxins | Hemolysis, tissue damage | Cytolytic |
| Panton-Valentine Leukocidin (PVL) | Necrotizing skin infections, severe pneumonia | Two-component pore-forming toxin; destroys PMNs. Strongly associated with CA-MRSA |
| Toxic Shock Syndrome Toxin-1 (TSST-1) | TSS | Superantigen — activates up to 20% of T cells nonspecifically → massive cytokine storm |
| Enterotoxins A–Q | Food poisoning, TSS | Heat-stable superantigens |
| Exfoliative toxins A & B (ET-A, ET-B) | Scalded skin syndrome (SSSS) | Serine proteases that cleave desmoglein-1 in superficial epidermis |
C. Immune Evasion Mechanisms
- Protein A — blocks opsonization
- Catalase — neutralizes H₂O₂ from PMNs
- Coagulase / clumping factor — fibrin coat protects against phagocytosis
- Leukocidins (PVL, γ-hemolysin) — kill phagocytes
- Intracellular persistence within osteoblasts, endothelial cells
5. CLINICAL DISEASES
A. Skin & Soft Tissue Infections (SSTIs) — Most Common
| Infection | Features |
|---|---|
| Impetigo | Superficial; honey-colored crusted lesions; children |
| Folliculitis | Hair follicle inflammation |
| Furuncle (boil) | Deep hair follicle infection with central necrosis |
| Carbuncle | Coalescence of multiple furuncles; systemic symptoms |
| Cellulitis | Spreading skin/subcutaneous infection |
| Wound infections | Post-surgical; most common cause of surgical site infections |
| Necrotizing fasciitis | Life-threatening; deep fascia involvement; often CA-MRSA/PVL |
B. Bacteremia & Sepsis
- S. aureus is the #1 cause of surgical wound infections and #2 cause of primary bacteremia (after NSaS)
- Bacteremia seeds distant sites → metastatic infections (endocarditis, osteomyelitis, epidural abscess, septic arthritis)
- Mortality ~20–40%
- Always conduct echocardiogram when bacteremia identified
C. Infective Endocarditis (IE)
- Native-valve IE: especially in IDU (right-sided → tricuspid valve)
- Prosthetic-valve IE: S. aureus + NSaS are leading causes
- Can present as acute endocarditis with rapid valve destruction
- Complications: emboli, abscesses, metastatic foci
D. Pneumonia
- Hematogenous (bacteremia) or direct inhalation (post-influenza)
- Cavitating/necrotizing pneumonia especially with PVL-producing CA-MRSA
- Classic: follows influenza infection — "secondary bacterial pneumonia"
E. Osteomyelitis & Septic Arthritis
- #1 cause of both hematogenous osteomyelitis and septic arthritis
- Children: long bones (metaphysis)
- Adults: vertebral osteomyelitis (check MRI spine in prolonged bacteremia)
F. Central Nervous System Infections
- Brain abscess, meningitis (especially with shunts/catheters)
- Epidural abscess (presents with back pain + fever → emergency)
G. Toxin-Mediated Diseases
1. Food Poisoning
- Preformed, heat-stable enterotoxin — disease occurs without viable bacteria
- Onset: 1–6 hours after ingestion
- Symptoms: vomiting, diarrhea, cramping — no fever
- Self-limited (~24–48 hours)
- Sources: improperly stored/cooked food (meats, dairy, salads)
2. Toxic Shock Syndrome (TSS)
- Agent: TSST-1 (menstrual) or enterotoxins (non-menstrual)
- Mechanism: Superantigen binds MHC-II directly → massive T-cell activation → cytokine storm
CDC Clinical Criteria for TSS (must meet all 5 for Confirmed; 4 of 5 for Probable):
| Criterion | Detail |
|---|---|
| Fever | ≥38.9°C (102°F) |
| Rash | Diffuse macular erythroderma (sunburn-like) |
| Desquamation | 1–2 weeks after onset, especially palms & soles |
| Hypotension | SBP ≤90 mmHg in adults; < 5th percentile in children |
| Multisystem involvement | ≥3 of: GI, muscular, mucous membrane, renal, hepatic, hematologic, CNS |
Lab: Negative blood/CSF cultures for other pathogens; negative serology for RMSF, leptospirosis, measles. Blood cultures may be positive for S. aureus.
- Menstrual TSS: associated with superabsorbent tampons
- Non-menstrual: wound infections, nasal packing, post-partum
- Recurrences possible if antibody to TSST-1 fails to develop
- Illness only in persons lacking anti-TSST-1 antibody
3. Staphylococcal Scalded Skin Syndrome (SSSS)
- Caused by exfoliative toxins A & B (serine proteases cleaving desmoglein-1)
- Primarily affects newborns and children (adults rare — have neutralizing antibodies + better renal clearance)
- Spectrum: localized blisters → generalized exfoliation
- Nikolsky's sign positive: gentle lateral pressure → shear of superficial epidermis
- Mucous membranes spared (distinguishes from Stevens-Johnson syndrome)
- Thin-walled, fluid-filled bullae → denuded underlying skin
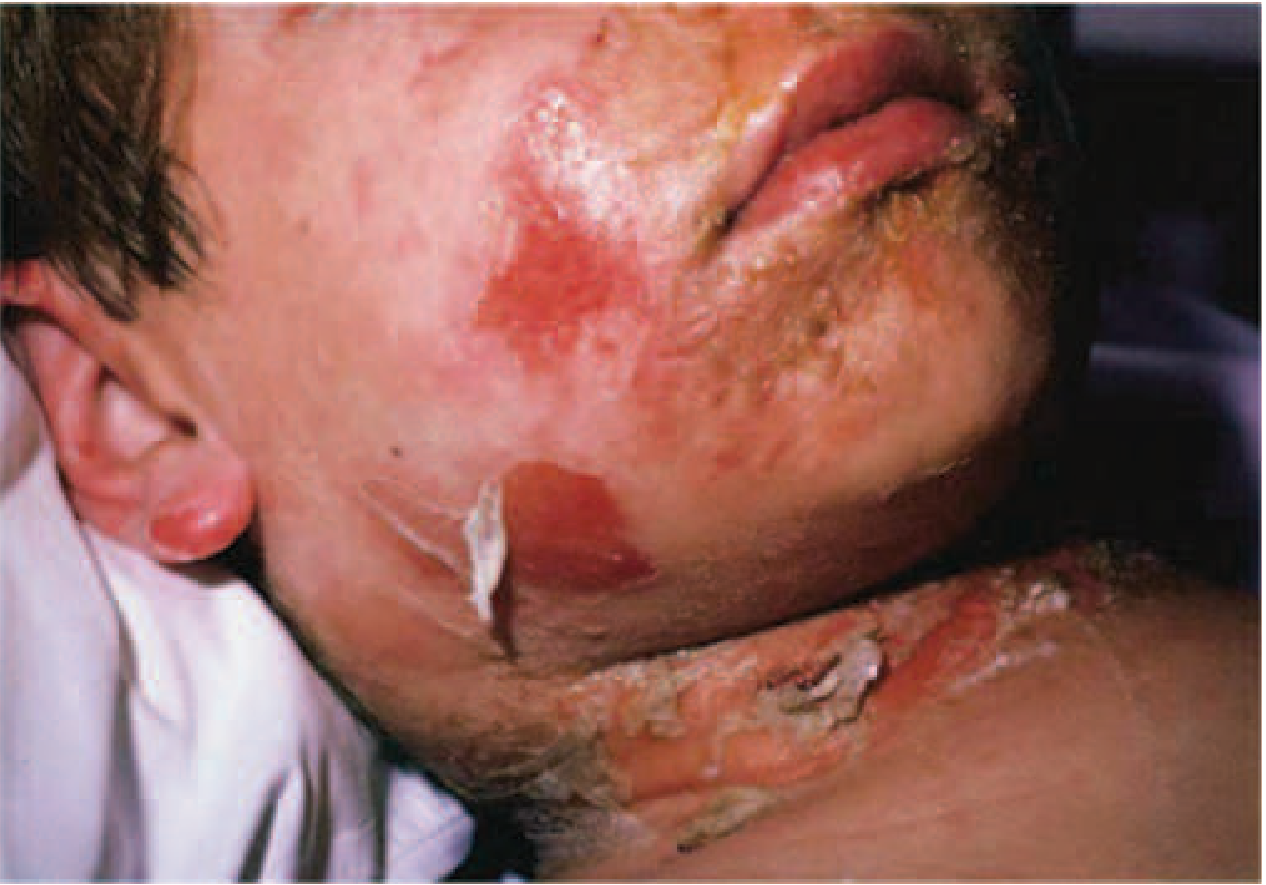
6. DIAGNOSIS
| Method | Use |
|---|---|
| Gram stain | Gram-positive cocci in clusters; useful for pyogenic infections |
| Blood culture | Essential for bacteremia; less useful for toxin-mediated disease |
| Culture on selective media | Mannitol-salt agar, chromogenic agar for contaminated specimens |
| Coagulase test | Tube (free) + slide (bound/clumping factor) — identifies S. aureus |
| Latex agglutination | Detects Protein A + clumping factor |
| NAAT / PCR | Screening for MSSA and MRSA carriage (nares swab) |
| MALDI-TOF | Rapid speciation from colonies |
| WGS | Outbreak investigations — gold standard strain typing |
7. TREATMENT
Antibiotic Selection
| Strain | Drug of Choice | Alternatives |
|---|---|---|
| MSSA (methicillin-sensitive) | Nafcillin / Oxacillin (IV) or Dicloxacillin (oral) | Cefazolin (preferred for surgical prophylaxis) |
| MRSA (oral) | TMP-SMX, Doxycycline / Minocycline, Clindamycin, Linezolid | — |
| MRSA (IV / serious) | Vancomycin (drug of choice) | Daptomycin, Tigecycline, Linezolid, Ceftaroline |
| VRSA / Difficult MRSA | Daptomycin, Linezolid, Ceftaroline | — |
⚠️ Empirical therapy should always cover MRSA until susceptibilities are known.
Management Principles
- Localized infections (furuncles, abscesses): Incision & drainage (I&D) is primary — antibiotics often not needed unless systemic signs
- Bacteremia: Minimum 2 weeks IV antibiotics; 4–6 weeks if complicated (endocarditis, osteomyelitis)
- Food poisoning: Supportive only (replace fluids); identify source for public health control
- TSS: Remove source of toxin; aggressive IV fluids; vancomycin ± IVIG
- SSSS: Antistaphylococcal antibiotics; wound care; fluids
Resistance Mechanism of MRSA
- mecA gene (on SCCmec element) encodes PBP2a — a modified penicillin-binding protein with low affinity for all β-lactams → resistance to all penicillins, cephalosporins, and carbapenems
- CA-MRSA (USA300) typically carries SCCmec type IV (smaller, more mobile)
- HA-MRSA typically carries SCCmec types I–III (larger; more resistance genes)
8. PREVENTION & CONTROL
| Setting | Measure |
|---|---|
| Healthcare | Thorough hand hygiene; contact precautions; active surveillance screening (MRSA nares PCR); decolonization protocols |
| Surgical | Preoperative decolonization with mupirocin ± chlorhexidine in MRSA carriers |
| Community | Avoid sharing personal items; proper wound care; hygiene education |
| Decolonization | Mupirocin nasal ointment (2% BID × 5 days) + chlorhexidine body wash |
| Vaccine | No licensed vaccine currently available |
9. QUICK-RECALL HIGH-YIELD SUMMARY
| Point | Fact |
|---|---|
| Gram stain | Gram+ cocci in grape-like clusters |
| Key marker | Coagulase +, Protein A +, catalase + |
| Colonies | Golden, β-hemolytic on blood agar |
| Most common community SSTI cause | MRSA (CA-MRSA) |
| TSS toxin | TSST-1 → superantigen |
| SSSS toxin | Exfoliative toxin A/B → cleaves desmoglein-1 |
| Food poisoning onset | 1–6 hours (preformed, heat-stable enterotoxin) |
| MRSA resistance gene | mecA → encodes PBP2a |
| IV drug of choice for MRSA | Vancomycin |
| MSSA IV drug of choice | Nafcillin / Oxacillin |
| Abscess management | I&D first |
| Nikolsky's sign | SSSS (mucous membranes spared) |
| Necrotizing pneumonia | PVL-producing CA-MRSA |
| USA300 | Dominant CA-MRSA clone in USA |
| Job syndrome risk | Hyperimmunoglobulin E → ↑ S. aureus infections |
Sources: Harrison's Principles of Internal Medicine, 22nd Ed. (2025), Chapter 152, pp. 1239–1245 · Medical Microbiology 9e · Goldman-Cecil Medicine
This is a shared conversation. Sign in to Orris to start your own chat.