Cool sculpting for fatloss
Reading File
Finding Sources
Searching PubMed
cryolipolysis fat reduction efficacy safety
Searching the Web
CoolSculpting cryolipolysis fat loss mechanism efficacy safety 2025
Reading File
Reading File
Searching Images
cryolipolysis CoolSculpting fat freezing procedure applicator
Here is a comprehensive overview of CoolSculpting (cryolipolysis) for fat reduction:
CoolSculpting (Cryolipolysis) for Fat Reduction
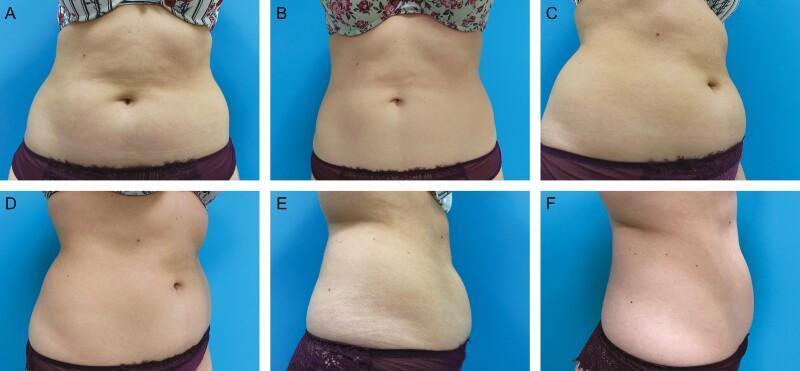
What It Is
CoolSculpting is the brand name for cryolipolysis, an FDA-cleared, non-invasive body contouring procedure that uses controlled cooling to selectively destroy subcutaneous fat. It was FDA-approved starting in 2010 (initially for flanks), with subsequent approvals for the abdomen, thighs, back, submental (chin), submandibular, bra fat, and buttock regions.
Mechanism of Action
The science rests on a key biological principle: adipocytes (fat cells) are more sensitive to cold than surrounding skin, nerves, blood vessels, and muscle.
- Lipid crystallization — Fat cells contain lipids that crystallize at temperatures well above the freezing point of water (~4°C). Skin cells are largely unaffected at these temperatures.
- Controlled cooling — A vacuum-powered applicator suctions the target tissue between two cooling panels, chilling it to a precise sub-zero temperature for 35–60 minutes.
- Apoptosis — Stressed fat cells undergo programmed cell death (apoptosis), not necrosis, which limits damage to surrounding tissue.
- Inflammatory clearance — Over the following 2–3 months, the body's immune system (macrophages) removes the dead adipocytes via an induced inflammatory response. This results in a permanent reduction of fat cells in the treated area.
"Following apoptosis, an induced inflammatory response clears the cells over the proceeding 3 months." — Andrews' Diseases of the Skin
Efficacy
- A single treatment session reduces the fat layer by ~20–25% in the treated area.
- Full results are visible at 2–3 months post-treatment.
- Many patients require a repeat session at 3 months for optimal results.
- Post-treatment massage of the treated area has been shown to increase fat reduction by more than 50% and is now a standard part of the procedure.
- A 2025 systematic review and meta-analysis (PMID 40473257) confirmed efficacy specifically for submental (double chin) fat reduction.
The Procedure
| Feature | Detail |
|---|---|
| Setting | Office, no anesthesia or sedation needed |
| Duration | 35–60 min per area |
| Downtime | None — return to work/exercise immediately |
| Applicators | Multiple shapes/sizes for different body regions |
| Sessions | 1–2 treatments per area, spaced 3 months apart |
Side Effects & Risks
Short-term (common, self-resolving):
- Erythema (redness), edema, bruising at treatment site
- Temporary numbness/altered sensation — may last up to 2 months
- "Cold panniculitis" reaction (inflammatory response to cold in the fat)
Prolonged:
- Hypoesthesia (reduced sensation) from cold damage to lipid-rich myelin sheaths of sensory nerves
- Delayed pain — managed with compression garments, lidocaine patches, or gabapentin
Rare but serious:
- Paradoxical adipose hyperplasia (PAH) — ~1 in 20,000 cases. Instead of shrinking, the treated fat paradoxically enlarges, forming a firm, painless mass in the shape of the applicator. The mechanism is unclear. Treatment: surgical liposuction.
What It Is NOT
- Not a weight-loss treatment — it targets localized fat bulges, not overall obesity. It does not affect blood lipid levels or body weight significantly.
- Not equivalent to liposuction — it is less aggressive; liposuction removes more fat in one session but requires downtime and anesthesia.
- Not suitable for everyone — contraindicated in cryoglobulinemia, cold agglutinin disease, paroxysmal cold hemoglobinuria, and Raynaud's phenomenon.
Ideal Candidates
- Adults near their ideal body weight with pinchable, localized fat deposits that don't respond to diet and exercise
- Common treatment areas: abdomen, flanks ("love handles"), inner/outer thighs, submental region, upper arms, back fat, bra fat
Comparison with Other Non-Invasive Fat Reduction Methods
| Method | Mechanism | Fat Reduction |
|---|---|---|
| CoolSculpting (cryolipolysis) | Cold-induced apoptosis | ~20–25% per session |
| SculpSure | Laser heat destruction | ~24% per session |
| Kybella (deoxycholate) | Chemical lysis of adipocytes | Submental only, 2–4 sessions |
| HIFU/Ultrasound (e.g., Liposonix) | Ultrasonic thermal disruption | Variable |
Sources: Andrews' Diseases of the Skin, Clinical Dermatology; Dermatology 2-Volume Set 5e (Elsevier); FDA Non-Invasive Body Contouring; PMID 40473257 (2025 systematic review).
Is this a fat loss treatment in clinical guidelines
Searching PubMed
obesity management clinical guidelines
Searching the Web
CoolSculpting cryolipolysis obesity fat loss clinical guidelines AHA ACC recommended treatment
Finding Sources
Reading File
No — CoolSculpting/cryolipolysis is not a fat loss treatment in any major clinical guidelines for obesity.
Here's the clear distinction:
Clinical Guidelines for Obesity: What They Recommend
Major guidelines (NHLBI, ACC/AHA/TOS, AACE, ESC) define a stepwise treatment approach for obesity based on BMI and complications:
| Stage | Interventions Recommended |
|---|---|
| All patients | Lifestyle modification: caloric restriction, physical activity, behavioral support |
| BMI ≥27–30 + comorbidities | Add pharmacotherapy (e.g., orlistat, GLP-1 agonists, naltrexone/bupropion) |
| BMI ≥35–40 + complications | Bariatric surgery (sleeve gastrectomy, RYGB) |
Cryolipolysis does not appear in any of these guideline frameworks. None of the major bodies — NHLBI, ACC/AHA, AACE, ESC, AGA — include it as a recommended treatment for obesity or meaningful fat loss.
Why CoolSculpting Is Excluded from Obesity Guidelines
-
It targets cosmetic fat, not metabolic fat. It only reaches subcutaneous fat (the fat under the skin you can pinch). It has no effect on visceral fat — the metabolically dangerous fat around organs that drives cardiovascular disease, diabetes, and metabolic syndrome.
-
It does not produce clinically meaningful weight loss. Guidelines define clinically meaningful weight loss as ≥5% of body weight, associated with measurable improvements in BP, lipids, and blood glucose. CoolSculpting achieves ~20–25% reduction in a small, localized area — this translates to grams, not kilograms.
-
It does not treat obesity-related complications. The complications-centric AACE model requires therapies that reduce morbidity (e.g., reducing HbA1c, blood pressure, cardiovascular risk). CoolSculpting addresses none of these.
-
It is explicitly contraindicated in obesity. WebMD and clinical references state it should be avoided in patients with obesity — it is designed for people near their ideal body weight with isolated fat deposits.
What CoolSculpting Actually Is, Clinically
It is classified as a cosmetic/aesthetic body contouring procedure — found in dermatology and plastic surgery references, not in internal medicine or endocrinology treatment guidelines. It is appropriate for:
- Adults at or near normal BMI with stubborn, localized subcutaneous fat
- Cosmetic improvement of contour (abdomen, flanks, chin, thighs)
- Situations where diet/exercise have already achieved overall weight goals but focal deposits persist
Bottom line: CoolSculpting is an FDA-cleared cosmetic device, not a medical treatment for obesity or fat loss as defined in clinical guidelines. If a patient's goal is medically significant weight reduction, clinical guidelines direct them toward lifestyle intervention, pharmacotherapy (e.g., semaglutide, tirzepatide), and/or bariatric surgery.
— Fuster and Hurst's The Heart, 15th Edition; ACC/AHA/TOS Guidelines (2014); AACE Obesity Guidelines
This is a shared conversation. Sign in to Orris to start your own chat.