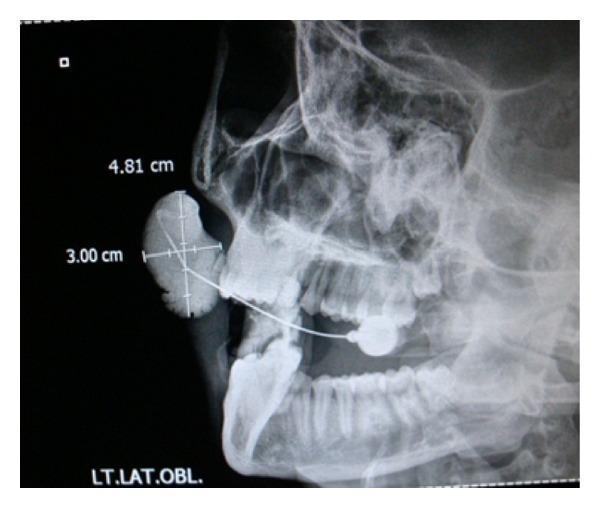
Sialogram in detail according to standard ENT textbook
sialography parotid duct contrast injection technique
Sialogram (Sialography)
Definition
Indications
| Indication | Notes |
|---|---|
| Chronic/recurrent sialadenitis | Evaluates ductal architecture |
| Sialolithiasis (salivary stones) | Detects radiolucent stones with 95–100% sensitivity using digital subtraction sialography |
| Ductal stenosis or stricture | Best modality to depict fine intraglandular branches |
| Sialectasis (ductal ectasia) | Classic "snowstorm" appearance in Sjögren syndrome |
| Sjögren syndrome staging | Sialography or MR sialography helps stage disease |
| Autoimmune salivary gland diseases | Evaluates duct anatomy in obstructive and inflammatory conditions |
| CT sialography | Supplemental in patients who cannot have MRI to evaluate a suspected mass in a dense gland |
"Although rarely needed, conventional sialography remains the best radiographic method for evaluating duct anatomy in obstructive, inflammatory, and autoimmune salivary gland diseases." — Cummings Otolaryngology, Diagnostic Imaging chapter
Contraindications
- Acute sialadenitis — the procedure may exacerbate symptoms or cause ascending infection
- Iodine contrast allergy — water-soluble contrast agents are iodine-based
- Stones in the oral portion of the Wharton duct — active inflammation
- Uncooperative patients (especially children) — requires duct cannulation
- Sublingual glands — rarely performed due to the multiple small ducts of Rivinus that open directly into the floor of the mouth, making overview difficult
Relevant Anatomy
Stensen Duct (Parotid)
- Orientation: Horizontally oriented
- Length: 6–7 cm
- Caliber: ~1–2 mm
- Opens opposite the upper second molar in the buccal mucosa
Wharton Duct (Submandibular)
- Orientation: Angles downward and laterally at ~45° relative to both sagittal and axial planes
- Length: ~5 cm
- Caliber: 1–3 mm
- Papilla is small, more difficult to cannulate
- More prone to perforation than the parotid duct
Equipment
- Blunt-tipped sialographic cannula with attached tubing
- Lacrimal dilators — may be necessary to enter the papilla
- Water-soluble contrast agent — diatrizoate meglumine (e.g., Gastrografin) provides excellent ductal opacification with no reported adverse effects following duct perforation
- Fluoroscopy unit
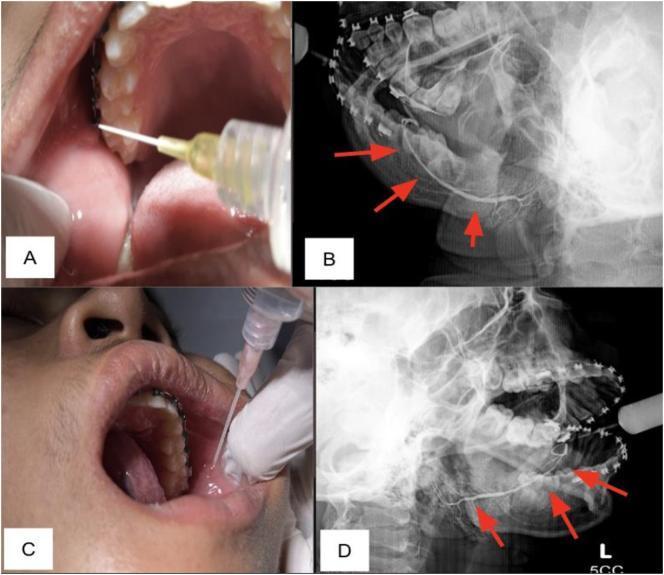
Technique (Step-by-Step)
-
Preparation: Dry the mucosa around the papilla; massage the gland to produce saliva (or use a secretagogue — e.g., lemon juice — placed under the tongue to stimulate salivary flow and identify the papilla)
-
Cannulation: A blunt-tipped sialographic cannula is introduced into the ductal orifice. Lacrimal dilators may be needed to widen the papilla
-
Contrast injection:
- Stensen duct: ~1 mL of water-soluble contrast
- Wharton duct: ~0.5 mL (smaller caliber)
- Injection should be slow and under fluoroscopic observation; stop when the patient feels slight pressure
-
Phases:
- Ductal (filling) phase: Main duct and its branches are opacified
- Acinar (parenchymal) phase: Contrast fills into terminal acini — maximizes parenchymal opacification and silhouettes mass lesions
-
Imaging projections: Anteroposterior (AP), lateral, and oblique views are obtained to optimize visualization of the ductal system
-
Post-sialographic CT: May be helpful in assisting localization of abnormalities within the gland following conventional sialography (CT-sialography). If CT is performed, the catheter is left in place and the gland is reinjected during scanning
Sialogram Technique Image
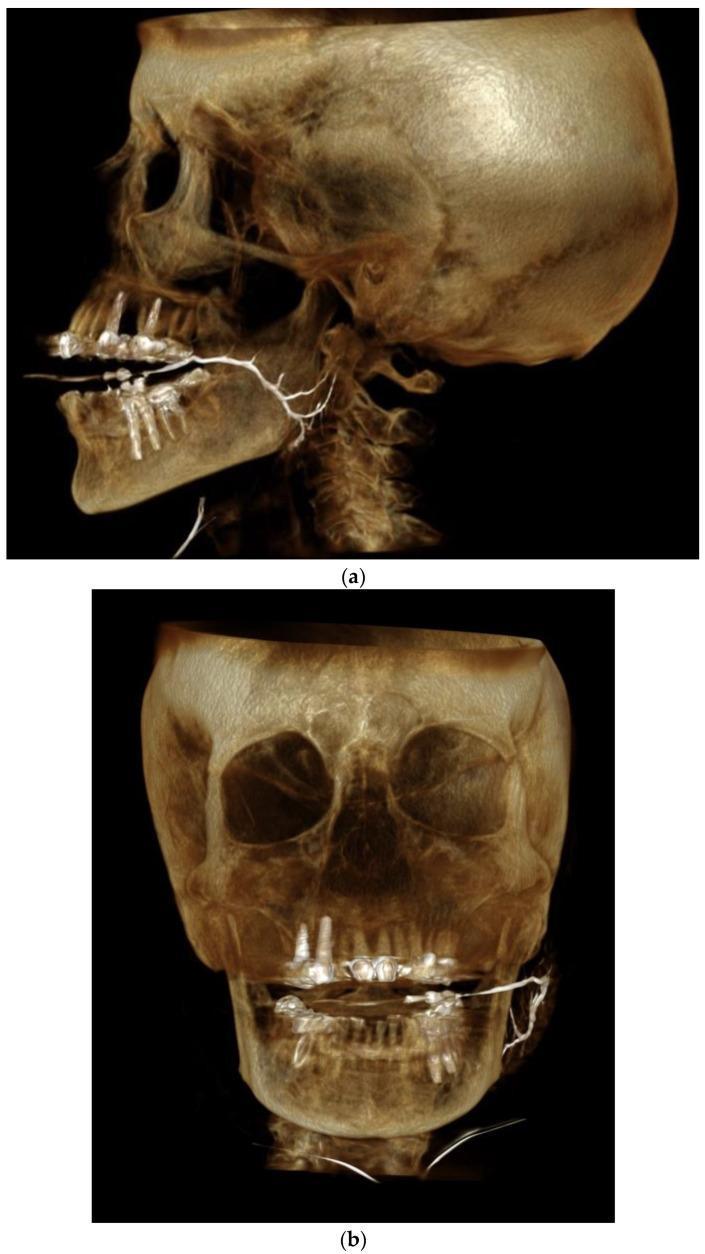
Normal Findings
- Stensen duct: Smooth, continuous radiopaque column from papilla to the gland hilum, with uniform tapering into intraglandular branches
- Branching pattern: Progressive branching into secondary and tertiary ducts within the gland parenchyma (arborization pattern)
- No filling defects, strictures, or areas of non-filling
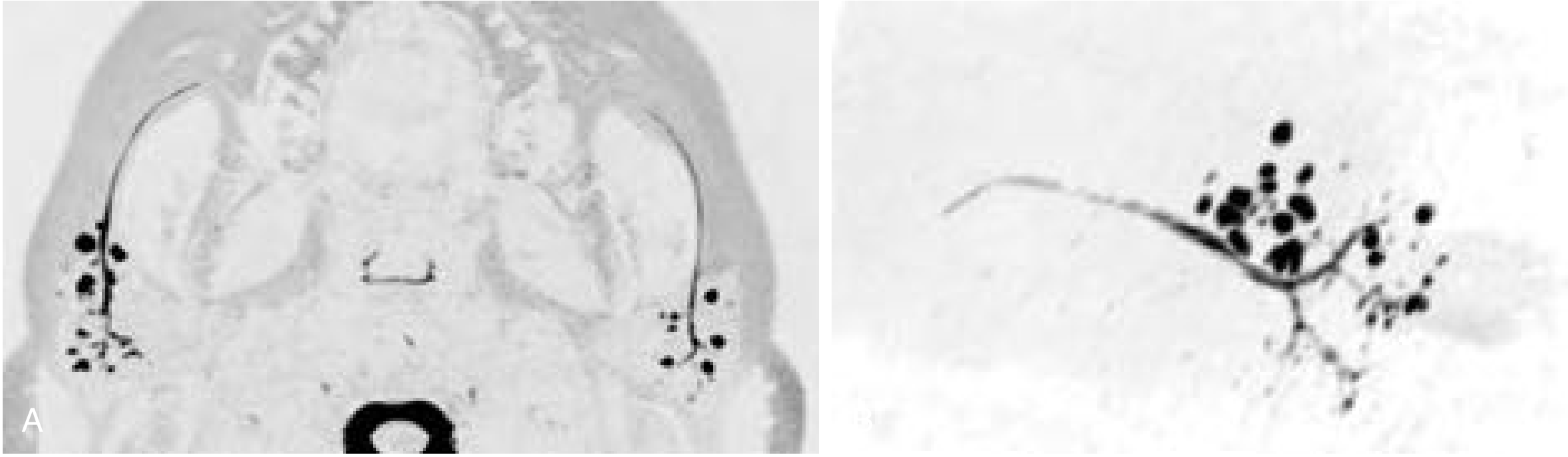
Pathological Findings
| Finding | Significance |
|---|---|
| Filling defect (radiolucent) | Sialolith (stone) |
| Abrupt cut-off of duct | Obstruction by stone or tumor |
| Stricture / stenosis | Chronic sialadenitis, trauma |
| Sialectasis (duct dilatation) | Distal obstruction or Sjögren syndrome |
| "Snowstorm" / punctate sialectasis | Classical finding in Sjögren syndrome — cystic dilation of terminal intraparenchymal ducts |
| Sialocele | Pooling of contrast in a cystic cavity — ductal disruption or dilatation |
| Extravasation | Ductal perforation |
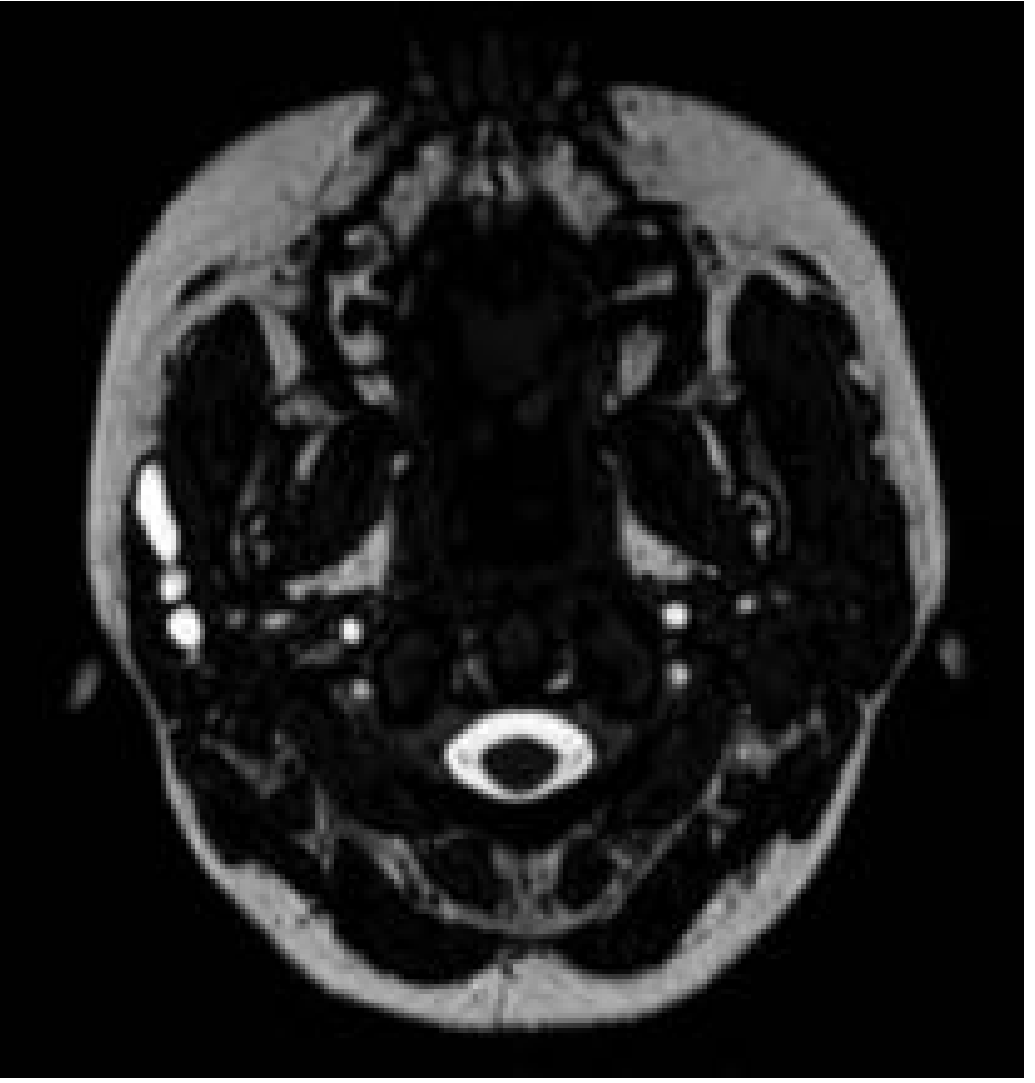
MR Sialogram — Sjögren Syndrome
MR Sialography — Parotid Duct Stenosis
Sialography Showing Sialocele / Obstruction
Complications
| Complication | Notes |
|---|---|
| Local pressure / pain | Very common; typically resolves within 24 hours |
| Post-sialographic sialadenitis | Ascending infection |
| Ductal perforation | Particularly with Wharton duct cannulation |
| Contrast extravasation | May cause local inflammatory reaction |
CT-Sialography
- Water-soluble or fat-soluble contrast is injected intraductally before CT acquisition
- The catheter is left in place and the gland is reinjected during CT
- Plane of study should be angled to avoid dental filling artifacts
- Caution: Concentrated sialographic contrast collecting in dilated ducts, acini, or large pools can cause significant streak artifacts that obscure small masses
- For optimal results, injection is extended into the acinar (parenchymal) phase to maximize parenchymal opacification and silhouette mass lesions
MR Sialography
| Feature | Details |
|---|---|
| No contrast injection needed | Non-invasive; no duct cannulation |
| No ionizing radiation | Safe even in acute inflammation |
| Sequences used | Heavily T2-weighted; RARE (Rapid Acquisition with Relaxation Enhancement), FSE, CISS, HASTE |
| First described | Lomas et al., 1996 |
| Performance | Well-validated for intraglandular + extraglandular ductal system in Sjögren syndrome, ductal stenosis, post-radiation xerostomia |
| vs. Conventional | Spatial resolution slightly inferior; however, eliminates need for conventional sialography in many settings |
| vs. Digital sialography (for sialolithiasis) | MR sialography with evoked salivation has similar accuracy to digital sialography and is superior to ultrasound |
| Limitation | Ductal metaplasia of sialolithiasis may be misinterpreted as mucoepidermoid carcinoma |
Digital Subtraction Sialography
- Lessens interference of surrounding bony structures
- Can detect radiolucent stones with reported sensitivity of 95–100%
- Provides better detail than plain film sialography for fine ductal pathology
Current Role and Comparison with Other Modalities
| Modality | Primary Use |
|---|---|
| Conventional sialography | Gold standard for ductal anatomy; obstructive/autoimmune disease |
| Ultrasound | First-line; detects >90% of stones >2 mm; operator-dependent |
| CT (non-contrast) | Excellent for sialoliths; 1–2 mm cuts highly accurate |
| MRI | Best for neoplastic lesions; perineural invasion; parapharyngeal extension |
| MR sialography | Best non-invasive replacement for conventional sialography |
| Nuclear (⁹⁹ᵐTc pertechnetate) | Functional assessment; Warthin tumor (hot nodule) |
"Conventional sialography is the gold standard for evaluating the ductal system in the salivary glands; magnetic resonance sialography has been shown to perform well in evaluating the intraglandular and extraglandular ductal system in the setting of Sjögren syndrome, ductal stenosis, and post-radiation xerostomia. Advances in CT and MRI make these cross-sectional modalities superior for the evaluation of most salivary lesions, both neoplastic and inflammatory." — Cummings Otolaryngology — Head and Neck Surgery
Sialoendoscopy
sialendoscopy wire basket stone extraction salivary duct
Sialoendoscopy (Sialendoscopy)
Definition
"This innovative method may become the gold standard for the investigation and treatment of many salivary gland pathologies." — Cummings Otolaryngology
Indications
| Category | Specific Conditions |
|---|---|
| Obstructive | Sialolithiasis (stones in Stensen or Wharton duct) |
| Inflammatory / Chronic | Chronic and acute sialadenitis unresponsive to medical therapy |
| Pediatric | Juvenile recurrent parotitis (JRP) — most frequent pediatric indication (68.9%) |
| Autoimmune | Sjögren syndrome — duct dilation and washout |
| Structural | Congenital and acquired ductal strictures |
| Post-treatment | Radioiodine-induced sialadenitis — washout, debris removal, stent placement |
| Diagnostic | Visualization of sialoliths, stenosis, polyps, sialodochitis, undetected stones |
In a systematic review of pediatric sialendoscopy: JRP was the most frequent indication (68.9%), followed by sialolithiasis (14.7%).
Contraindications
- Active infection / acute sialadenitis — risk of spreading infection
- Inability to cooperate (relative; can be managed with GA)
Instrumentation
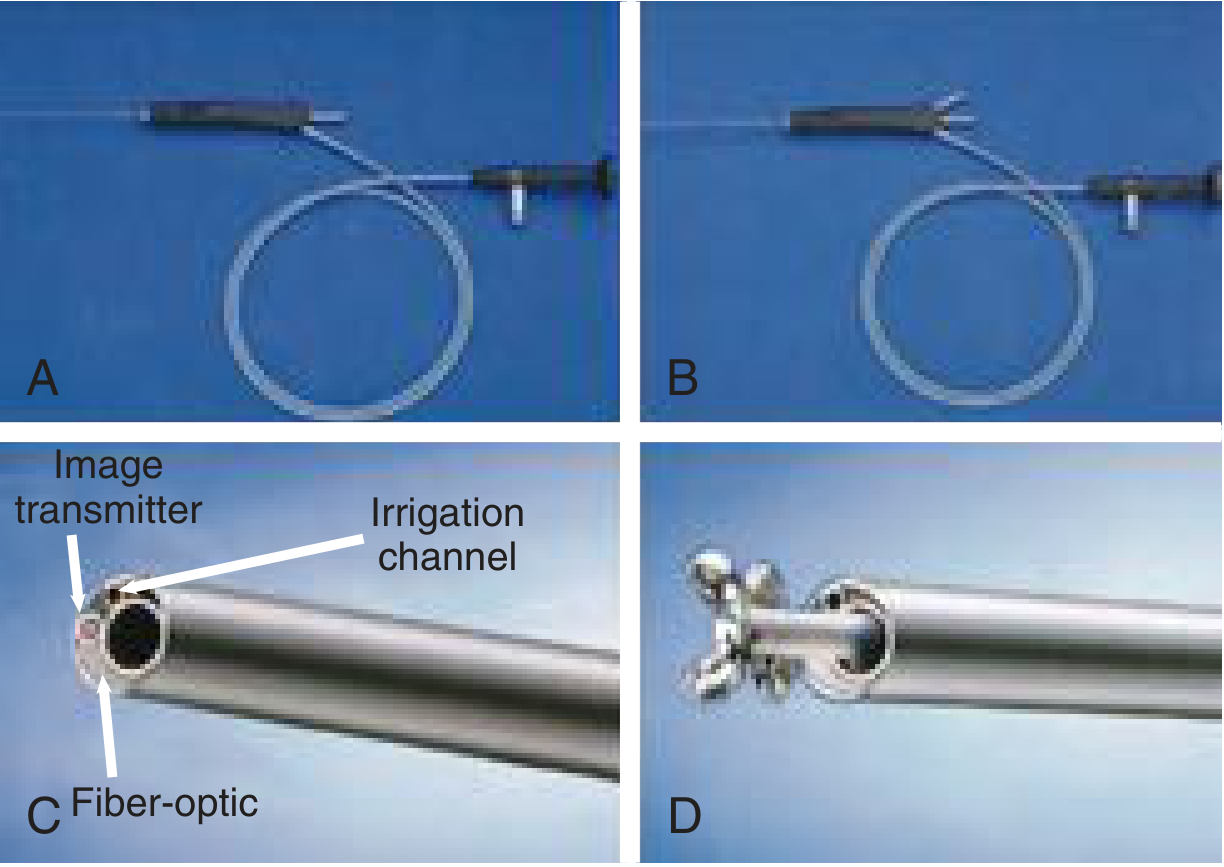
Types of Sialendoscopes
| Type | Features |
|---|---|
| Diagnostic | Fiberoptic light, image transmission, irrigation channel; no working channel |
| Therapeutic — compact | Single-unit; combines fiber light, image transmission, working channel, irrigation channel in one instrument |
| Therapeutic — modular | Optical probe inserted into sheaths of variable size; gap used as irrigation; more versatile |
The Erlangen (Karl Storz) Sialendoscope
| Size | Features |
|---|---|
| 0.8 mm | Integrated lens + irrigation channel only; no working channel |
| 1.1 mm | Lens + irrigation + working channel (instruments passable) |
| 1.6 mm | Largest; all features; may be too large for some pediatric patients |
Instruments Passed Through the Working Channel
- Microdrill — fragments larger stones mechanically
- Holmium laser fiber — endoscopic lithotripsy for large/hard stones
- Pneumatic (intraductal) lithotripsy probe — newer modality for stone fragmentation
- Wire basket (Dormia-type) — for stone fragment extraction
- Balloon dilator — for ductal stenosis dilation
- Stent — to maintain ductal patency after dilation (kept for up to 4 weeks)
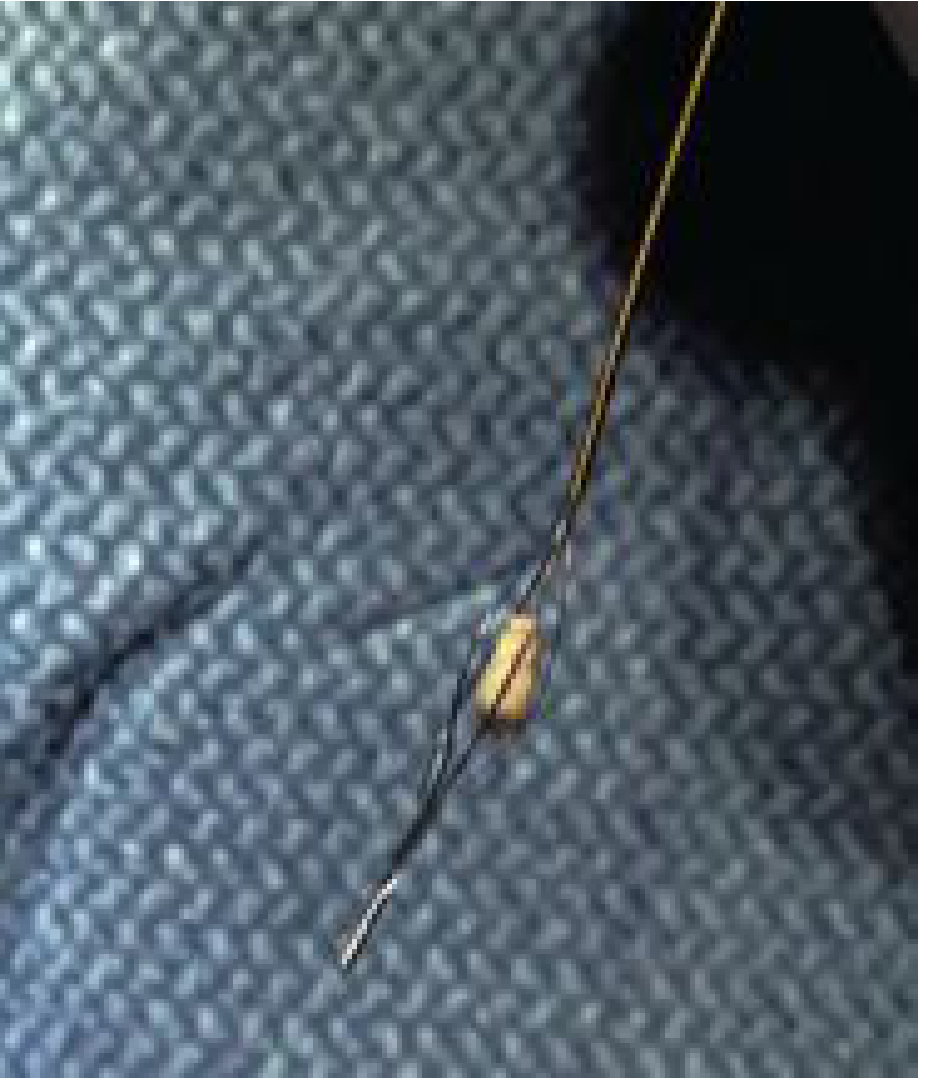
Anesthesia
- General anesthesia — standard; required for younger children and uncooperative patients
- Local anesthesia — possible in children aged >8 years who can cooperate, and in adults
Technique (Step-by-Step)
1. Patient Positioning and Setup
- Appropriate bite block placed on the contralateral side to access the papilla
2. Papilla Identification
- The ductal papilla of Stensen (parotid) or Wharton (submandibular) duct is identified
- Secretagogue (lemon juice) or gentle massage helps identify the papilla by inducing salivary flow
3. Serial Dilation
- Salivary duct probes and dilators are used sequentially to progressively widen the papilla
- Lacrimal dilators may be used as an adjunct
4. Scope Introduction
- The sialendoscope (attached to a camera and monitor) is introduced into the duct via the dilated orifice
- Continuous saline irrigation is maintained throughout to:
- Maintain visualization
- Provide hydraulic dilation
- Flush out mucus, debris, and small stone fragments
5. Ductal Exploration
- The scope is advanced to the first branching point of the main duct
- Each branch is examined as far as the scope can comfortably pass
- The ductal mucosa, caliber, and any filling defects are systematically assessed
6. Therapeutic Interventions (as needed)
| Pathology | Intervention |
|---|---|
| Small stones (<4 mm) | Wire basket extraction directly |
| Large/impacted stones | Holmium laser lithotripsy or pneumatic intraductal lithotripsy → basket extraction of fragments |
| Stones too large/distal | Combined approach: endoscope localizes, then small external/intraoral incision extracts |
| Ductal stenosis | Balloon dilation ± stent placement |
| Chronic sialadenitis / JRP | Saline washout + intraglandular corticosteroid instillation (triamcinolone 40 units in 3–5 mL saline) |
| Radioiodine sialadenitis | Washout, mucus plug/debris removal, dilation, stent placement |
| Mucus plugs / inflammatory debris | Lavage and mechanical removal |
Endoscopic Views
Endoscopic Stone Identification and Basket Retrieval
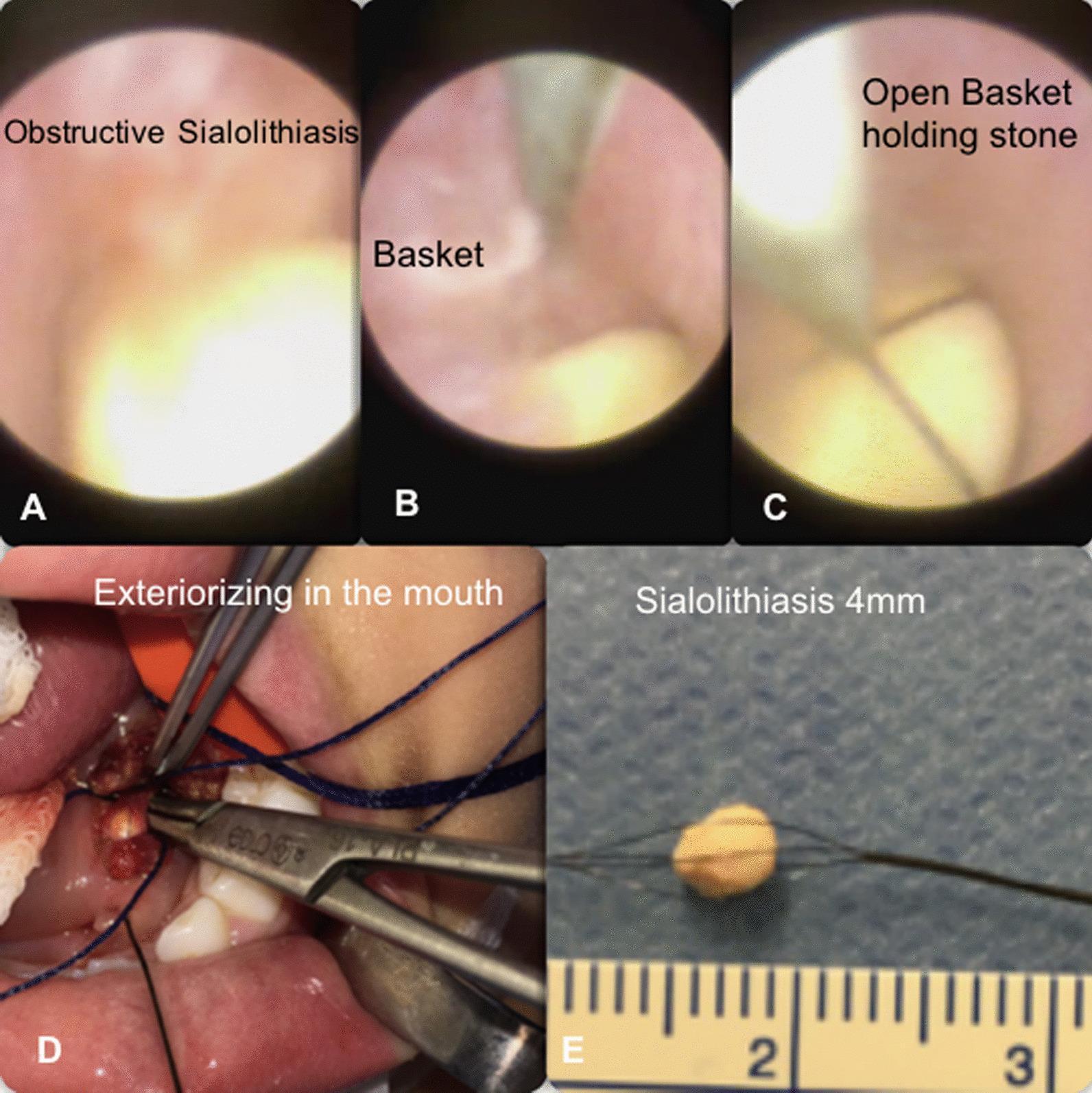
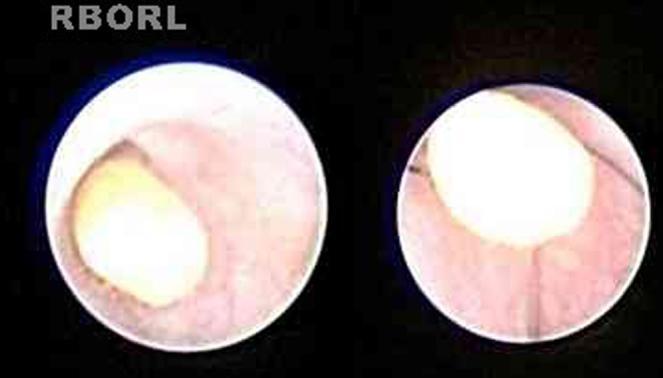
Endoscopic Findings
| Finding | Appearance |
|---|---|
| Normal duct | Pale pink, smooth, glistening mucosa; uniform caliber; regular branching |
| Sialolith | Yellowish-white, hard, irregular mass obstructing ductal lumen |
| Stenosis / stricture | Narrowing of ductal lumen; pale, fibrotic mucosa |
| Mucus plug | Semi-transparent, whitish, gelatinous material |
| Sialodochitis (ductal inflammation) | Erythema, friable mucosa, irregular walls |
| Sialectasis | Focal or diffuse ductal dilation |
| Polyp | Pedunculated mucosal projection |
Role in Specific Conditions
Juvenile Recurrent Parotitis (JRP)
- Most common pediatric indication
- Sialendoscopy provides:
- Ductal washout with saline
- Hydrostatic dilation of peripheral ducts
- Intraglandular corticosteroid instillation (triamcinolone)
- Dilation of strictures
- Sialography (with iodinated oil) may also serve both diagnostic and therapeutic roles in JRP — the antiseptic solution lingers in the duct for days/weeks
- Virtually all cases of JRP resolve spontaneously at puberty
Sjögren Syndrome
- Sialendoscopy dilates and washes out affected ducts
- MR sialography or sialography can help stage disease
- A multidisciplinary approach (salivary endoscopist + rheumatologist) is recommended
Radioiodine-Induced Sialadenitis
- Gland washout, removal of debris and mucus plugs, dilation ± stenting
- Reported symptomatic success rates: 50–100%
Sialolithiasis
- Sialendoscopy is now first-line for gland-preserving stone removal
- Digital subtraction sialography: 95–100% sensitivity for radiolucent stones
- Ultrasound: first-line, detects >90% of stones >2 mm
- Combined endoscope + external approach for large/distal stones
Combined Approach (Endoscope-Assisted External Surgery)
- Sialendoscope localizes the stone and transilluminates the overlying mucosa/skin
- A small intraoral or external incision is made directly over the stone
- Stone is extracted via sialoductotomy
- Gland is preserved; the duct is repaired
Advantages Over Conventional Surgery
| Feature | Sialendoscopy | Open Surgery / Gland Excision |
|---|---|---|
| Gland preservation | Yes | No |
| Minimally invasive | Yes | No |
| Facial/lingual nerve risk | Minimal | Significant |
| Applicability in acute inflammation | No (contraindicated) | Possible |
| Day procedure | Usually | Sometimes inpatient |
| Recurrence management | Repeatable | Limited |
Complications
| Complication | Notes |
|---|---|
| Ductal perforation | Most common; usually minor |
| Ductal avulsion | Rare; excessive force during dilation |
| Post-procedure sialadenitis | Ascending infection |
| Ranula / sialocele | Rare |
| Basket entrapment | If stone too large for basket extraction |
| Incomplete stone removal | Residual fragments — may require repeat procedure or combined approach |
| Ductal stenosis (post-procedure) | Managed with stenting |
| Failure to cannulate | Small/fibrotic papilla |
Summary: Diagnostic vs. Therapeutic Sialendoscopy
| Mode | Purpose | Features |
|---|---|---|
| Diagnostic | Visualization; inspection | Fiberoptic + irrigation; no working channel |
| Therapeutic | Treatment | Working channel for basket, laser, balloon, stent |
"Sialendoscopic techniques include endoscopic visualization of stenosis or calculi, balloon dilatation of stenoses, fragmentation of large stones with Holmium laser, and ensnarement of stones with a wire basket." — Cummings Otolaryngology
Describe the methods of identification of facial nerve during parotid surgery and various methods to preserve the nerve during parotid surgeries
Facial Nerve Identification and Preservation in Parotid Surgery
Surgical Anatomy — Brief Review
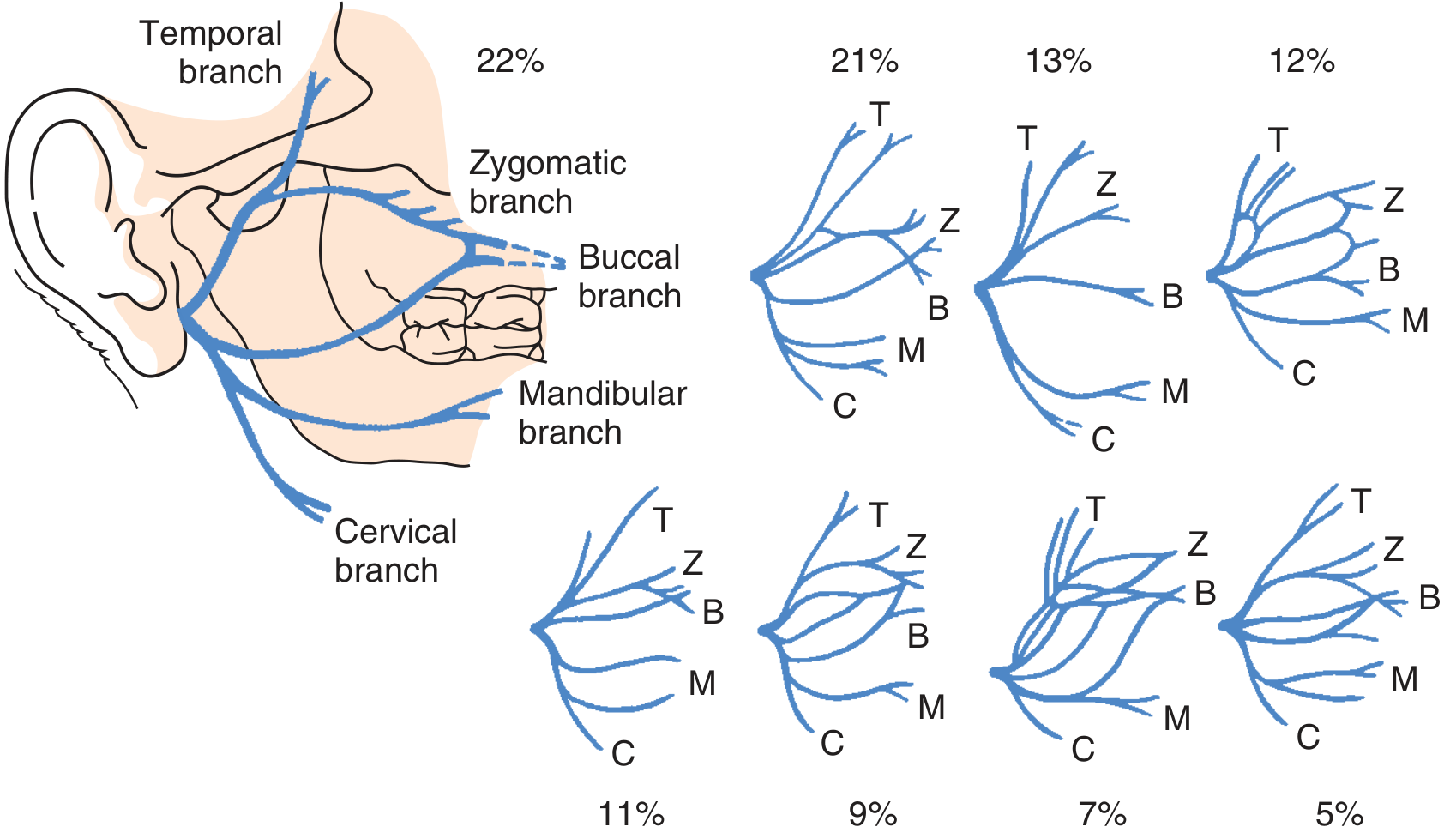
A. Methods of Identification of the Facial Nerve
1. Antegrade (Anterograde) Approach — Identification at the Main Trunk
Landmark 1: Tragal Pointer (Most Common Method)
- The medial tip of the tragal cartilage — the "pointer" — points directly toward the facial nerve
- The main trunk is located ~1 to 1.5 cm deep and inferior to the tragal pointer
- The parotid tissue is elevated off the anterior surface of the tragal cartilage; retraction anteriorly exposes the pointer
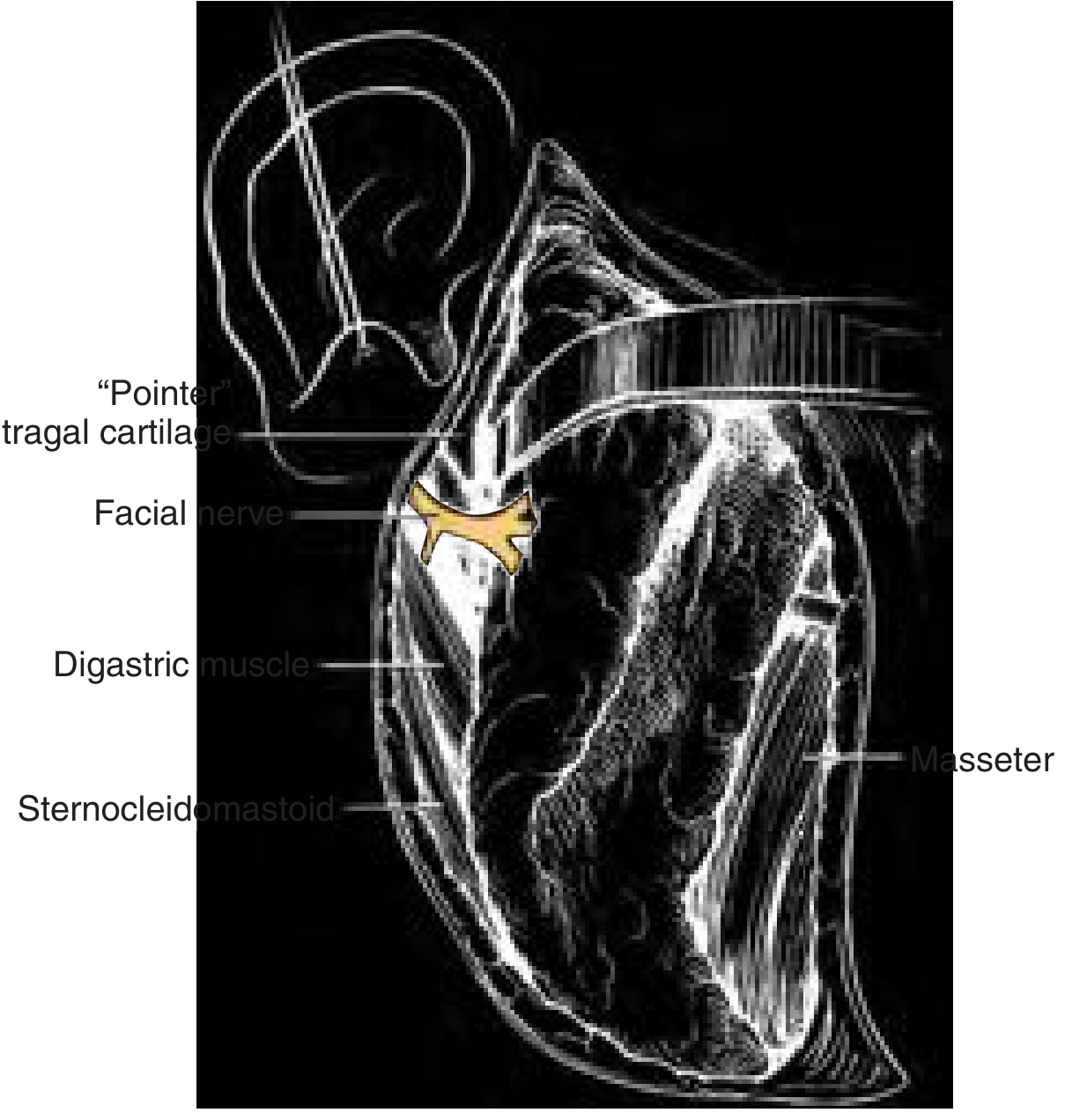
Landmark 2: Tympanomastoid Suture Line
- The tympanomastoid suture (junction of the tympanic bone and mastoid) is followed medially
- The facial nerve lies 6–8 mm deep to the tympanomastoid suture
- Described as a more constant landmark than the tragal pointer
- The parotid gland is dissected proceeding deep to the tympanic and mastoid bones along this suture line
Landmark 3: Posterior Belly of the Digastric Muscle
- The posterior belly of the digastric muscle is exposed at its attachment to the mastoid bone
- The facial nerve lies in the tissue plane between the tragal pointer and the attachment of the digastric to the mastoid
- The digastric serves as the inferior boundary of the nerve's expected location
- The tissue between these two landmarks is carefully divided to expose the nerve
Stylomastoid Artery
- The stylomastoid artery (a branch of the posterior auricular artery) runs in close association with the facial nerve at the stylomastoid foramen
- Its identification can guide the surgeon to the nerve — listed as a named landmark in K.J. Lee's
2. Retrograde (Peripheral → Central) Approach
- Tumor overlies the region of the main trunk
- Recurrent tumors with significant scarring at the main trunk
- Previous surgery has distorted normal anatomy
Via the Marginal Mandibular Branch
- Most commonly used peripheral branch for retrograde identification
- Located below the lower border of the mandible, crossing the superficial branch of the facial vessels in the plane immediately beneath the deep cervical fascia
- Identified by following the posterior facial vein superiorly — the marginal mandibular branch crosses this vein
- Once identified, it is traced proximally and retrograde back to the main trunk
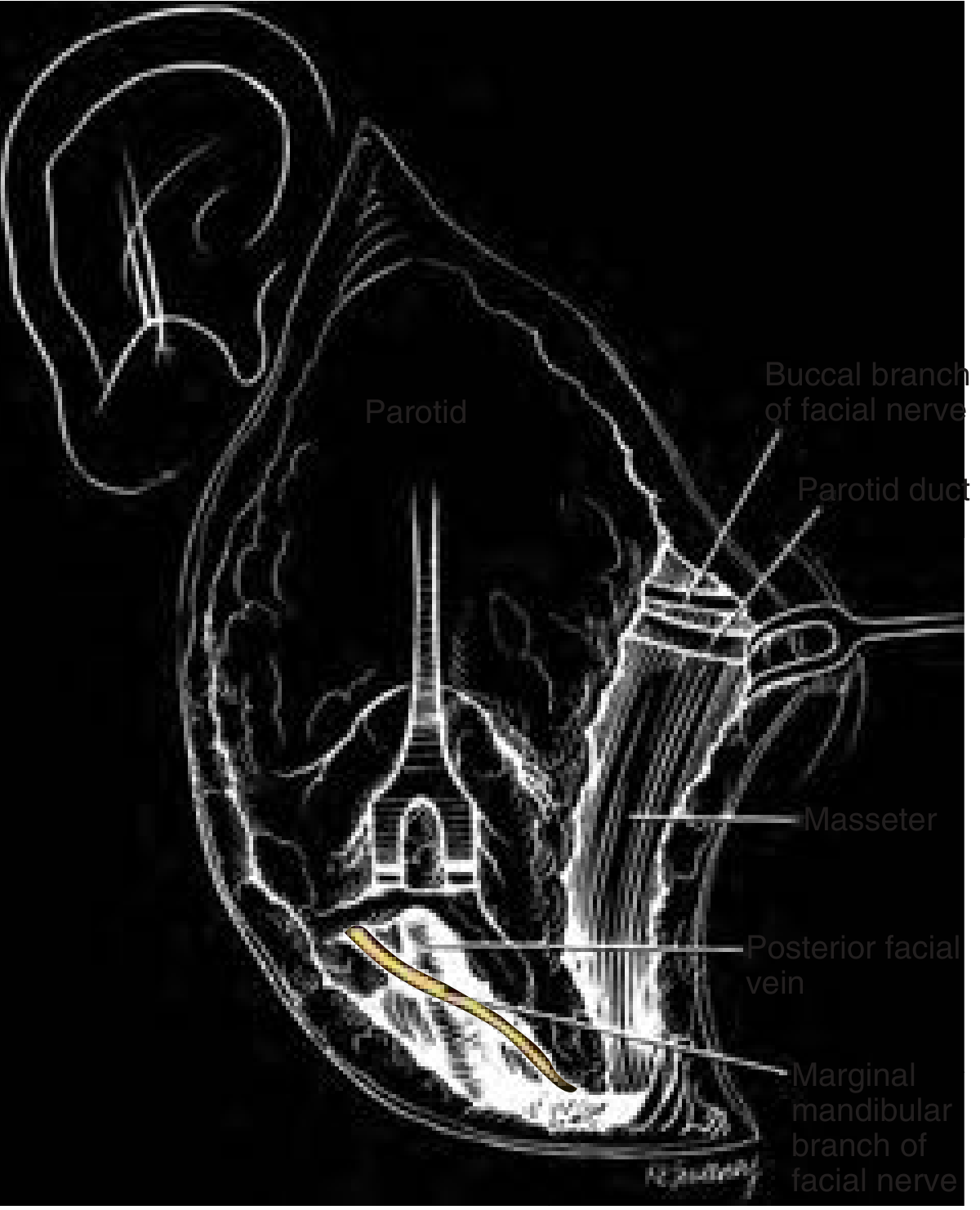
Via the Buccal Branch
- Found beneath the parotidomasseteric fascia, coursing parallel to the parotid duct
- Can be identified and traced back to the pes anserinus and main trunk
Via the Temporal Branch
- Can be identified in the preauricular region and traced posteriorly
3. Mastoid (Proximal/Intratemporal) Approach
- Extensively scarred fields (e.g., prior surgery, malignancy with perineural invasion requiring proximal margin)
- When tumor extends to or invades the facial nerve requiring resection to negative margins at the stylomastoid foramen or within the mastoid
- Requires mastoidectomy to expose the vertical segment of the facial nerve within the mastoid
- Nerve is identified within the mastoid and followed distally into the parotid
- Nerve mobilization after mastoidectomy is required to minimize tension on any reconstruction anastomosis
4. Electrical Nerve Stimulation / Intraoperative Facial Nerve Monitoring
- A nerve stimulator probe is used to identify suspected neural tissue by observing twitching of the ipsilateral facial musculature
- Intraoperative electromyographic (EMG) monitoring with needle electrodes in the facial muscles provides real-time feedback
- Particularly important when: normal anatomy is distorted, patient has had prior surgery, tumor is large, or revision cases
- Critically: Neuromuscular blocking agents (NMBs) must be avoided or reversed before and during dissection to allow nerve stimulation to function — the anesthesiologist must be informed
- Spontaneous EMG activity alerts the surgeon to proximity of the nerve even without direct stimulation
B. Methods of Preserving the Facial Nerve During Parotidectomy
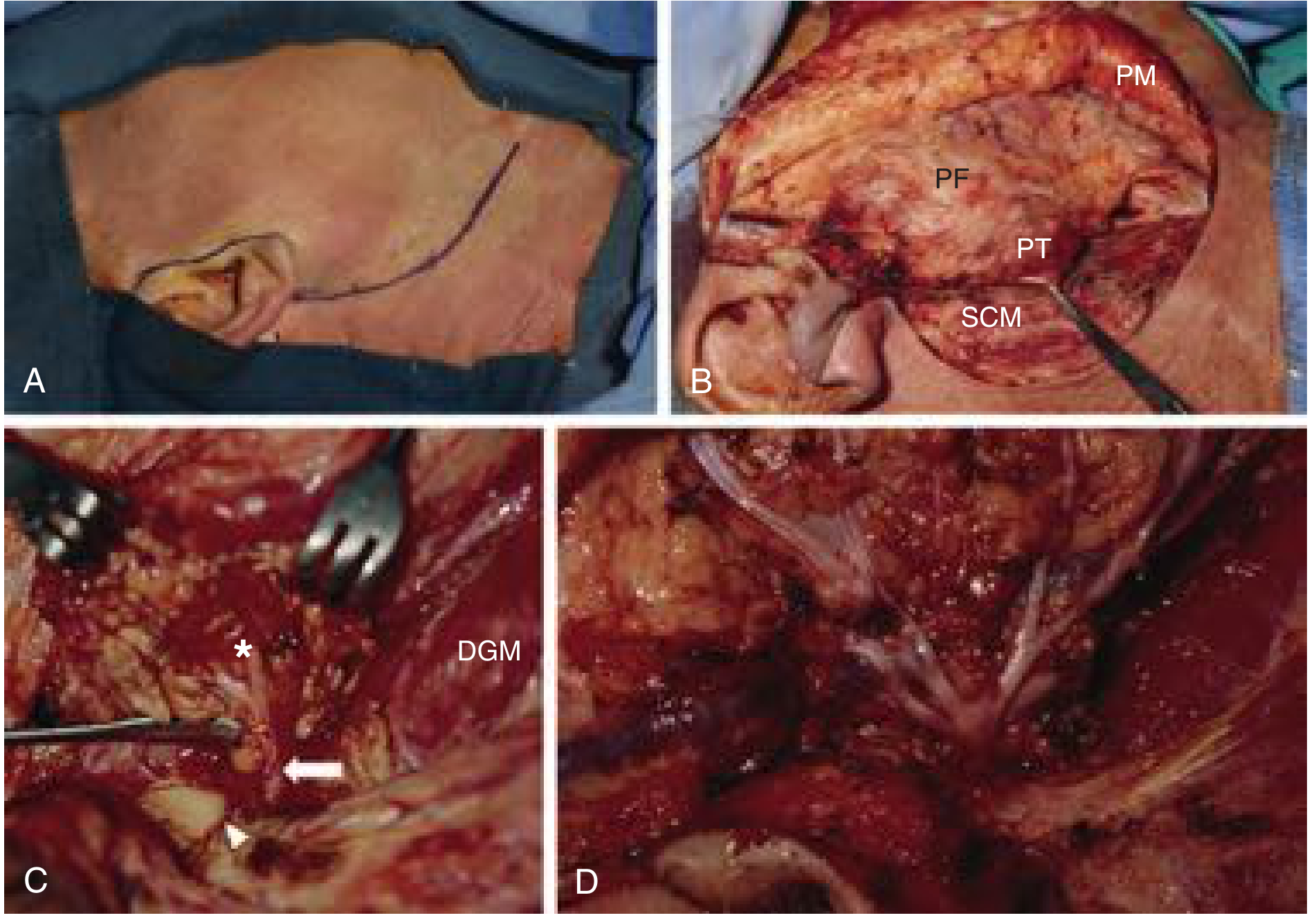
1. Systematic Anterograde Dissection
- The overlying parotid tissue is elevated off the nerve with a fine clamp — blades of the clamp straddle the nerve, and the intervening bridge of tissue is carefully divided
- Dissection proceeds distally from the main trunk to the first division (pes anserinus), then to each branch sequentially
- Each division and branch is individually and meticulously dissected in the same anterograde fashion
- The parotid tissue lateral to the nerve plane is progressively removed until the entire superficial lobe is delivered
2. Atraumatic Technique Principles
| Principle | Detail |
|---|---|
| Sharp, meticulous dissection | Avoid blunt tearing; use fine scissors or clamp to tent tissue off the nerve |
| Avoid thermal injury | Bipolar cautery preferred; monopolar kept well away from the nerve |
| Avoid traction | Excessive retraction on parotid tissue can stretch and injure the nerve |
| Continuous saline irrigation | Keeps the field clear, reduces thermal risk |
| Fine instruments | Mosquito clamps, tenotomy scissors — not large instruments |
| Bloodless field | Careful hemostasis prevents obscuring the nerve; uncontrolled bleeding leads to blind maneuvers |
3. Avoidance of Neuromuscular Blockade
- As above, NMBs are omitted or avoided so the nerve can be identified by electrical stimulation
- If NMB was used for induction, adequate reversal is confirmed before dissection begins
4. Preservation of Greater Auricular Nerve
- The greater auricular nerve is identified and traced inferiorly during flap elevation
- It is divided as distally as possible — preserving maximum length
- The posterior branch of the greater auricular nerve can often be preserved, reducing post-operative auricular numbness
- The proximal portion is preserved as a potential cable graft donor if the facial nerve requires reconstruction
5. Management When Tumor Abuts/Displaces the Nerve
- If the tumor abuts but does not invade the nerve, careful sharp dissection in the peritumoral plane is used
- The facial nerve can be gently mobilized off of deep lobe tumors (see deep lobe parotidectomy)
- If preoperative facial nerve function is fully intact → nerve most likely not invaded → all attempts made to preserve
- If preoperative evaluation reveals paresis or paralysis → nerve likely invaded → proceed with resection to negative margins (frozen sections guide extent)
6. Deep Lobe / Total Parotidectomy — Nerve Elevation Technique
- After complete superficial lobe removal with nerve dissection, the main trunk and all branches are carefully elevated off the underlying deep lobe tissue
- Multiple intraglandular and periglandular vessels must be controlled (branches of external carotid, retromandibular vein)
- The facial nerve is mobilized over the tumor surface using a plane of dissection immediately on the nerve surface
- The gland/tumor deep to the nerve is then dissected off deep musculature, mandible, and temporal bone
7. When Facial Nerve Must Be Sacrificed — Reconstruction
- Resection proceeds until proximal and distal margins are histologically negative (frozen section)
- May require mastoidectomy to achieve negative proximal margin
- Reconstruction options:
| Method | Notes |
|---|---|
| Primary neurorrhaphy | If tension-free end-to-end repair is possible |
| Greater auricular nerve graft | First choice for short gaps; same operative field |
| Sural nerve graft | Used for longer gaps; requires harvesting from leg |
| Nerve mobilization post-mastoidectomy | Reduces tension on anastomosis |
8. Defect Reconstruction to Prevent Complications
- Sialocele
- Frey syndrome (gustatory sweating)
- Cosmetic depression
- Abdominal fat graft
- AlloDerm (acellular dermal matrix)
- SMAS (superficial musculoaponeurotic system) flap
- Sternocleidomastoid (SCM) transposition flap
- Free tissue transfer (for large defects)
Summary Table: Methods of Facial Nerve Identification
| Method | Landmark / Technique | Key Measurement | When Preferred |
|---|---|---|---|
| Tragal pointer | Tip of tragal cartilage | ~1–1.5 cm deep and inferior | Standard, first choice |
| Tympanomastoid suture | Tympanic–mastoid bone junction | 6–8 mm deep | Most constant; routine |
| Posterior belly digastric | Digastric–mastoid attachment | Between digastric and tragal pointer | Routine; confirms zone |
| Stylomastoid artery | Accompanies nerve at foramen | — | Adjunct landmark |
| Retrograde — marginal mandibular | Posterior facial vein → nerve | Below mandible | Tumor overlying trunk; revision |
| Retrograde — buccal branch | Parallel to parotid duct | — | Alternative peripheral ID |
| Mastoid approach | Facial nerve in mastoid | — | Malignancy, perineural invasion |
| Nerve stimulator / EMG | Electrical stimulation → twitch | — | Distorted anatomy; all difficult cases |
Recent advances in investigation and management of salivary gland disease
"salivary gland" AND ("advances" OR "novel" OR "new")
"salivary gland" AND "sialendoscopy"
"salivary gland carcinoma" AND ("targeted therapy" OR "immunotherapy" OR "molecular")
"salivary gland" AND ("robotic surgery" OR "robot-assisted" OR "transoral")
"salivary gland" AND ("ultrasound elastography" OR "sonoelastography" OR "cone beam CT" OR "diffusion weighted")
"Sjogren syndrome" AND ("salivary gland" OR "saliva") AND ("biomarker" OR "diagnosis" OR "ultrasound")
"salivary gland" AND "core needle biopsy"
Recent Advances in Investigation and Management of Salivary Gland Disease
I. ADVANCES IN INVESTIGATION
1. Imaging
A. Ultrasound — Salivary Gland Elastography (Novel)
- Measures tissue stiffness by assessing shear wave velocity or strain — fibrotic/inflamed salivary glands (as in Sjögren syndrome) are stiffer than normal
- Two main techniques: strain elastography and shear wave elastography (SWE)
- A 2024 systematic review and meta-analysis (Dai et al., Eur Radiol, PMID 37658892) across 15 studies (816 pSS patients) showed:
- Pooled sensitivity: 80% | Pooled specificity: 87% for primary Sjögren syndrome (pSS)
- Better performance in patients aged ≤51 years
- SWE technique and measurement location are key determinants of performance
- A non-invasive, cost-effective technique that can differentiate pSS from healthy/disease controls without minor salivary gland biopsy
- A separate 2024 systematic review (Kise et al., Oral Radiol, PMID 38308723) also confirmed a role for USE in pSS diagnosis
- Point-of-care ultrasound (POCUS) is increasingly being integrated into rheumatology and ENT clinics for real-time salivary gland assessment
B. MRI with Advanced Sequences
- MRI is the primary modality for characterising salivary gland tumours
- Required sequences include:
- Diffusion-weighted imaging (DWI) — apparent diffusion coefficient (ADC) maps help differentiate benign from malignant lesions; malignant lesions have restricted diffusion (low ADC)
- Dynamic contrast-enhanced (DCE) MRI — perfusion curves distinguish Warthin tumour (type III washout) from pleomorphic adenoma and malignancies
- MR Sialography — heavily T2-weighted; non-invasive ductal mapping; validated for Sjögren syndrome, ductal stenosis, and post-radiation xerostomia
- For malignancy: CT neck + chest for lymph node and metastasis assessment
- FDG-PET — not currently recommended in routine salivary gland tumour management (high false-positive rate from parotitis)
C. Cone Beam CT (CBCT) Sialography
- CBCT sialography provides 3D reconstruction of the ductal system with lower radiation than conventional CT
- Comparable to MR sialography for detecting sialoceles and sialoliths
- Allows virtual salivary endoscopy from 3D reconstructions in complex cases
D. Endosonography
- Emerging application of intraductal ultrasound during sialendoscopy — the endoscopic probe provides high-resolution intraluminal sonographic assessment of duct walls and periluminal structures (Brandt et al., Ultraschall Med, 2025, PMID 39706218)
2. Tissue Sampling — Core Needle Biopsy vs. FNA
| Parameter | FNA | Core Needle Biopsy |
|---|---|---|
| Sensitivity | ~68% | ~99% |
| Specificity | High (both similar) | High |
| Non-diagnostic rate | Higher | Lower |
| Repeat procedures needed | More | Fewer |
| Complications | Rare | Slightly higher hematoma risk |
| Operator dependency | Significant | Less |
- CNB shortens time-to-diagnosis, reduces repeat visits, and improves surgical planning
- Performed under ultrasound guidance with 18–22G needles
- Early meta-analyses had suggested increased risk of facial nerve damage and tumour seeding — but no major complications (nerve injury, tumour seeding) have been confirmed in current meta-analyses
3. Molecular Profiling and Biomarkers
-
International guidelines (ESMO, NCCN) now urge routine testing of:
- Androgen receptor (AR) — in salivary duct carcinoma
- HER2 (ERBB2) — by IHC/FISH, particularly in salivary duct carcinoma
- NTRK gene fusions — in all non-adenoid cystic SGC eligible for systemic therapy
- MYB/MYBL1 rearrangements — pathognomonic for adenoid cystic carcinoma (AdCC)
- CRTC1/3::MAML2 fusion — diagnostic for mucoepidermoid carcinoma (MEC)
-
Advanced panels (NGS) also identify actionable mutations: NOTCH1/2, PI3KCA, BRAF, EGFR, FGFR
- Saliva is rich in proteins, metabolites, RNA, and cell-free DNA
- Salivary metabolomics may detect oral and systemic disease biomarkers (Garcia et al., Clin Oral Investig, 2024, PMID 39377832)
- Circulating tumour DNA (ctDNA) in saliva holds promise for early malignancy detection
- Novel IHC markers now used diagnostically: NR4A3 (nuclear receptor — acinic cell carcinoma), p63/p40, SOX10, GATA3, INSM1, PLAG1 (pleomorphic adenoma)
- These improve classification in diagnostically challenging cases
II. ADVANCES IN MANAGEMENT
1. Sialendoscopy — Expanded Role and Evidence Base
| Indication | Weighted Pooled Success Rate |
|---|---|
| Sialolithiasis (all) | 89.6% |
| Submandibular gland | 88.3% |
| Parotid gland | 81.2% |
| Ductal stenosis | 56.3% |
| Juvenile recurrent parotitis (JRP) | 67.0% |
| Radioiodine-induced sialadenitis (RAIS) | 45.8% |
| Combined endoscope + transoral | 86.3% |
- Intraductal pneumatic lithotripsy — newer addition alongside Holmium laser for fragmentation of hard/large stones
- Holmium laser lithotripsy — now used safely for most stones regardless of size
- Balloon dilation + stenting — for ductal stenosis; stents kept 4 weeks to prevent re-stenosis
- Steroid irrigation — A 2026 systematic review (Teng et al., Clin Otolaryngol, PMID 41501981) found symptomatic relief irrespective of steroid type/dose; however, evidence is insufficient to determine whether benefit is from steroid or mechanical flushing alone
2. Transoral Robotic Surgery (TORS) for Salivary Gland Disease
- TORS has been applied to submandibular sialolithiasis and parapharyngeal space tumours
- A 2025 systematic review and meta-analysis (Lazzeroni et al., Am J Otolaryngol, PMID 40311492) — 23 studies, 2,520 patients:
- TORS success rate: 95.7% vs conventional transoral: 92.6%
- Transient lingual nerve neuropraxia: more common with TORS (15.8% vs 8.1%)
- No permanent lingual nerve injuries in either group
- Conclusion: TORS is a valid option; high cost mandates strict patient selection
3. Targeted Molecular Therapy — Salivary Gland Malignancies
| Target | Drug | Histotype | Evidence |
|---|---|---|---|
| NTRK fusion | Larotrectinib (TRK inhibitor) | Any SGC with NTRK fusion | ~75% response rate; FDA-approved tissue-agnostic |
| NTRK fusion | Entrectinib | Any SGC with NTRK fusion | FDA-approved |
| HER2 | Trastuzumab ± pertuzumab | Salivary duct carcinoma | Phase II trials; responses seen |
| HER2 | Trastuzumab deruxtecan (T-DXd) | HER2-positive SGC | Promising phase II data |
| Androgen receptor | Androgen deprivation therapy (ADT) | Salivary duct carcinoma (AR+) | Used in clinical practice |
| NOTCH1/2 | NOTCH inhibitors (investigational) | Adenoid cystic carcinoma | Clinical trials |
| PI3K/AKT/mTOR | Investigational | Multiple histotypes | Trials ongoing |
| BRAF V600E | Vemurafenib/dabrafenib | BRAF-mutated SGC | Case reports/small series |
| MYB/MYBL1 | No approved target yet | AdCC | Active research |
4. Adenoid Cystic Carcinoma (AdCC) — New Molecular Insights
- MYB::NFIB and MYBL1::NFIB fusions — present in ~60–80% of AdCC; ongoing therapeutic targeting
- NOTCH1 mutations — present in high-grade transformation cases; NOTCH inhibitors under trial
- Epigenetic modifications (DNMT3A, KDM6A) — potential therapeutic targets
- AdCC is characterised by indolent but relentless progression; targeted therapy offers hope for unresectable/metastatic disease (Zupancic et al., Anticancer Res, 2024, PMID 38537991)
5. Management of Cancer Therapy-Related Salivary Dysfunction
| Approach | Details |
|---|---|
| IMRT / proton therapy | Parotid-sparing techniques; reduces xerostomia incidence |
| Amifostine | Radioprotector; reduces radiation-induced dry mouth (modest effect) |
| Pilocarpine / cevimeline | Muscarinic agonists; current standard sialogogues |
| Botulinum toxin (novel use) | Intra-glandular injection to manage sialocele and drooling; reduces Frey syndrome |
| Acupuncture | Growing evidence for XRT-induced xerostomia |
| Stem cell therapy (emerging) | Salivary gland stem/progenitor cell transplantation post-radiation; early-phase trials |
| Gene therapy (emerging) | AQP1 (aquaporin-1) gene transfer to restore secretory function — Phase I trial (NIH) showed promising results |
| Cellular organoids (emerging) | Salivary gland organoids under development for gland regeneration after radiation damage (Wu et al., Int J Oral Sci, 2024, PMID 39482304) |
6. Radioiodine-Induced Sialadenitis (RAIS)
- Sialendoscopy (washout, mucus plug removal, dilation, stenting) has success rates of 50–100%
- Amifostine and vitamin E have been explored as protective agents with limited evidence
- Botulinum toxin injection into the parotid pre-radioiodine therapy has been studied as a protective measure (reduces salivary gland uptake)
- Intensity-modulated proton therapy — avoidance of salivary glands in thyroid cancer treatment planning
7. Sjögren Syndrome — Advances in Management
- Rituximab (anti-CD20, B-cell depletion) — used for systemic manifestations; TRACTISS and TEARS trials showed limited benefit for glandular symptoms; role still debated
- Belimumab and abatacept — under active clinical investigation
- Sialendoscopy with steroid irrigation for recurrent sialadenitis episodes (multidisciplinary salivary endoscopist + rheumatologist model)
- Salivary gland ultrasonography (B-mode + elastography) is now being integrated into 2022 ACR/EULAR classification criteria revisions and monitoring protocols
8. Surgical Advances
- Partial (extracapsular) parotidectomy — growing evidence that partial superficial parotidectomy for benign tumours has lower rates of facial nerve dysfunction and Frey syndrome compared to complete superficial parotidectomy, with no higher recurrence rates (K.J. Lee)
- Facelift (retrotragal) incision — adopted for improved cosmesis in benign disease
- Intraoperative nerve monitoring (EMG) — now standard in many centres, especially revision surgery and malignancy
- 3D virtual planning and surgical navigation for complex deep-lobe and parapharyngeal tumours
Summary of Key Recent Evidence
| Area | Advance | Level | PMID |
|---|---|---|---|
| Investigation | Ultrasound elastography for pSS (Se 80%, Sp 87%) | Meta-analysis | 37658892 |
| Investigation | Core needle biopsy superior to FNA (Se ~99% vs ~68%) | Meta-analysis | 41264980 |
| Investigation | MRI (DWI + DCE) as primary imaging for tumours (REFCOR guidelines) | Consensus | 38036312 |
| Management | Sialendoscopy overall success rate 80.9% (stones 89.6%) | Meta-analysis | 37486613 |
| Management | Intraductal steroids in sialendoscopy — symptomatic benefit but insufficient quantitative data | Systematic Review | 41501981 |
| Management | TORS for submandibular stones: 95.7% success | Meta-analysis | 40311492 |
| Management | Personalised molecular therapy (NTRK/HER2/AR) in SGC | Review | 38401478 |
| Management | Cancer therapy-related xerostomia: stem cell/gene therapy emerging | Review | 39225092 |
| Emerging | Salivary gland organoids for regenerative medicine | Review | 39482304 |
| Evolving landscape | AdCC: NOTCH/MYB targets; salivary duct carcinoma: HER2/AR | Review | 37490348 |