pathophysiology behind all the sign and symptoms in iron deficiency anemia
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive data from multiple authoritative textbooks. Let me compile the complete answer.
Pathophysiology Behind Every Sign and Symptom in Iron Deficiency Anemia
Iron deficiency anemia (IDA) produces manifestations in two broad categories: (1) non-hematologic effects from tissue iron depletion (appear early, before anemia), and (2) hematologic effects from impaired erythropoiesis (appear late). Understanding each requires knowing what iron does beyond carrying oxygen.
Pathophysiologic Foundation
Iron is essential not only for hemoglobin, but for:
- Myoglobin (oxygen storage in muscle)
- Cytochromes and oxidative phosphorylation (mitochondrial ATP production)
- Iron-sulfur cluster enzymes (succinate dehydrogenase, aconitase)
- DNA synthesis (ribonucleotide reductase is iron-dependent)
- Neurotransmitter synthesis (tyrosine hydroxylase for dopamine/norepinephrine)
- Thyroid hormone metabolism
- Epithelial cell proliferation and integrity
Depletion follows a predictable sequence:
- Storage iron (ferritin/hemosiderin) depleted → ↓ serum ferritin
- Transport iron depleted → ↑ TIBC, ↓ transferrin saturation
- Tissue/enzyme iron depleted → non-hematologic symptoms appear
- Erythroid iron depleted → anemia, hypochromia, microcytosis appear
1. Fatigue and Reduced Exercise Tolerance
Mechanism (multi-factorial):
- Reduced O₂ delivery: ↓ hemoglobin → ↓ oxygen-carrying capacity → tissues shift to anaerobic metabolism → lactic acid accumulation → early exhaustion.
- Impaired mitochondrial function: Iron is a cofactor in the electron transport chain (Complexes I, II, III) and in cytochromes. Even before hemoglobin falls, skeletal muscle mitochondrial oxidative phosphorylation is impaired → reduced ATP yield per glucose molecule → muscle weakness and fatigue.
- Reduced myoglobin: Myoglobin facilitates intracellular O₂ diffusion and storage within myocytes. Iron deficiency depletes myoglobin, reducing the muscle's oxygen reserve during high demand.
- Heart and skeletal muscles become iron-depleted earlier than the erythron, so fatigue and reduced exercise tolerance are often the first symptoms — before any anemia is detectable. — Harrison's Principles of Internal Medicine 22E
"Isolated iron deficiency should not be underestimated since the resulting suboptimal metabolic function of myocytes, cardiomyocytes, and neurons may lead to fatigue, irritability, poor concentration, restless legs syndrome, and reduced exercise capability." — Harrison's 22E
2. Pallor
Mechanism:
- Reduced hemoglobin in RBCs → less oxyhemoglobin (which imparts the red-pink color to skin, mucous membranes, conjunctiva, and nail beds) → pallor.
- The hypochromic RBCs also carry less pigment per cell, contributing to the pale appearance of mucous membranes even at modest degrees of anemia.
Peripheral smear (from Rosen's Emergency Medicine):
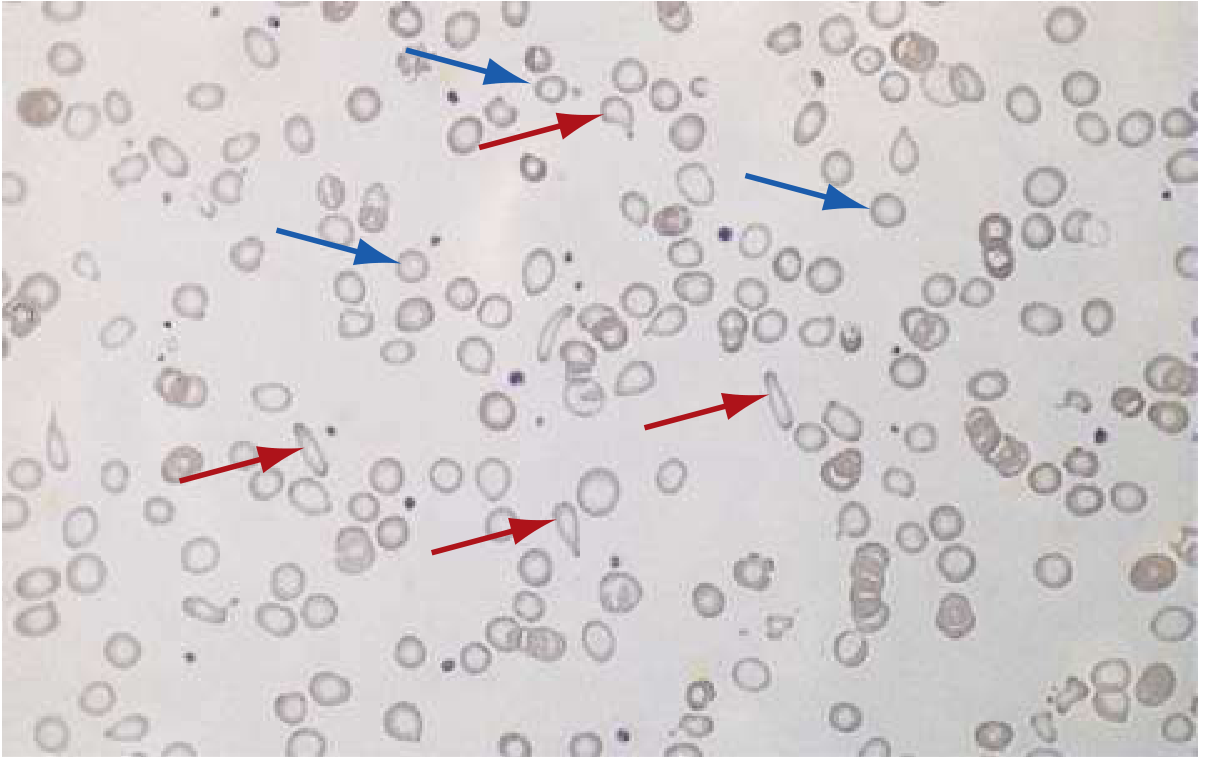
Red arrows = poikilocytes (pencil/cigar cells); Blue arrows = microcytic hypochromic RBCs. From Rosen's Emergency Medicine.
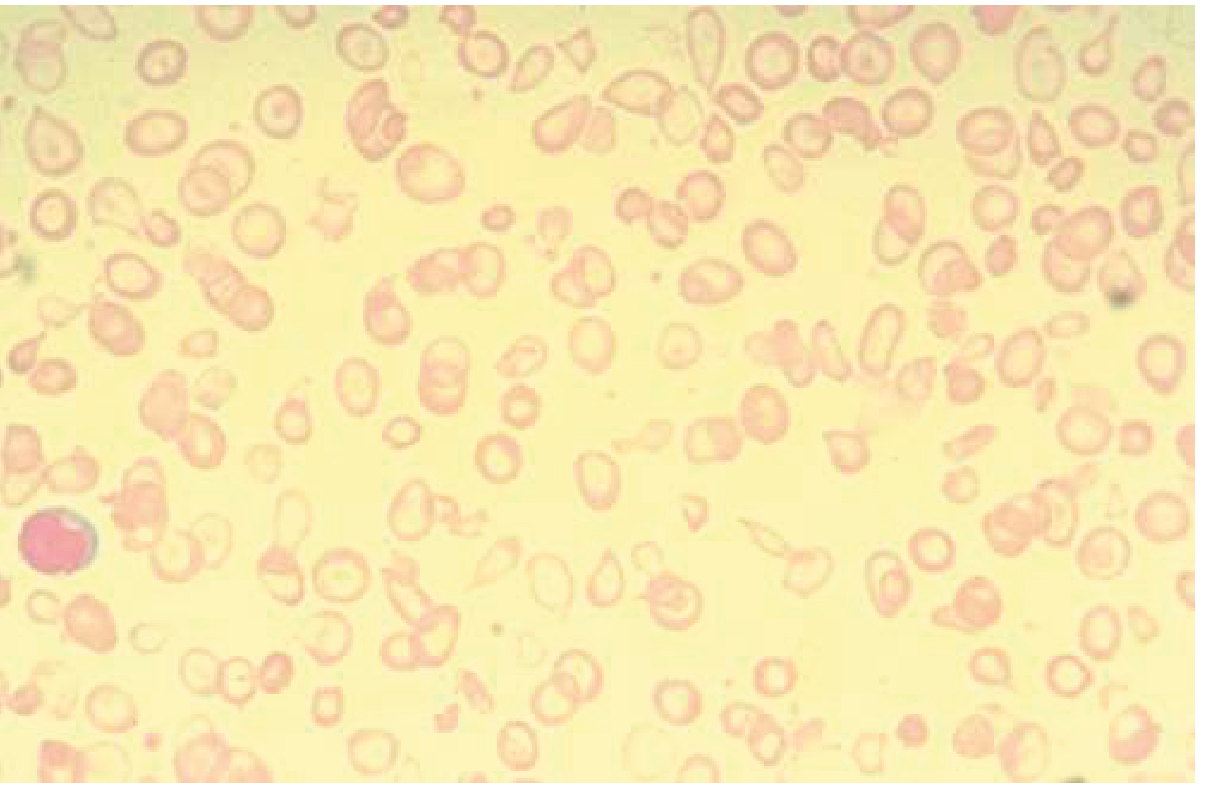
Classic IDA smear: cells have a narrow rim of hemoglobin at the periphery with a large area of central pallor. — Goldman-Cecil Medicine
3. Dyspnea and Palpitations
Mechanism:
- Reduced O₂ delivery triggers compensatory responses: increased heart rate (tachycardia), increased stroke volume, and vasodilation → palpitations.
- Chemoreceptors sense tissue hypoxia → increased respiratory drive → dyspnea, especially on exertion.
- As anemia worsens, high-output cardiac state → flow murmur, and in patients with coronary artery disease, new-onset or worsening angina may occur due to increased myocardial oxygen demand combined with decreased O₂ supply. — Goldman-Cecil Medicine
4. Headache, Irritability, Poor Concentration, Cognitive Impairment
Mechanism:
- Cerebral iron depletion: Iron is required for the synthesis of dopamine (via tyrosine hydroxylase) and other monoamine neurotransmitters. Iron-containing enzymes in the brain (e.g., monoamine oxidase) are impaired.
- Reduced cerebral O₂ delivery adds to neuronal dysfunction.
- In children, iron deficiency during critical periods of myelination causes lasting developmental impairment (iron is needed for myelin synthesis via fatty acid desaturases).
- Reduced ATP production in neurons → impaired axonal transport and synaptic function → headache, poor attention span, irritability.
5. Koilonychia (Spoon Nails)
Mechanism:
- Iron-containing enzymes are critical for keratinocyte proliferation and differentiation in the nail matrix.
- Iron deficiency → reduced keratinocyte iron → impaired cytoskeletal protein cross-linking → softening of the nail plate.
- Softened nails are molded by mechanical and capillary forces into a concave, spoon shape (koilonychia).
- Nails also become brittle, thin, and may develop longitudinal ridging.
"In severe long-standing iron deficiency, depletion of iron-containing enzymes in cells throughout the body causes koilonychia, alopecia, atrophic changes in the tongue and gastric mucosa." — Robbins Pathologic Basis of Disease
6. Alopecia (Hair Loss)
Mechanism:
- Hair follicle matrix cells are among the most rapidly proliferating cells in the body and have high iron requirements for DNA synthesis (ribonucleotide reductase is iron-dependent).
- Iron deficiency impairs DNA synthesis → shortened anagen (growth) phase → diffuse non-scarring scalp alopecia.
- Pattern: diffuse loss rather than focal, mimicking telogen effluvium.
- Goldman-Cecil notes hair loss is common in women with ferritin < 100 ng/dL — well above the threshold for anemia, confirming this is a tissue-iron effect.
7. Atrophic Glossitis (Smooth Painful Tongue)
Mechanism:
- The tongue's epithelium is rapidly renewing and highly iron-dependent.
- Iron deficiency → impaired proliferation of lingual papillary epithelium → atrophy of filiform and fungiform papillae → smooth, shiny, beefy-red tongue.
- Associated with burning, soreness, and sensitivity to hot/spicy foods.
- Iron-containing enzymes (succinate dehydrogenase, cytochrome c oxidase) in mucosal cells fail → mucosal atrophy and inflammation.
8. Angular Stomatitis (Cheilosis)
Mechanism:
- Same epithelial iron-depletion mechanism as glossitis, affecting the commissures of the mouth.
- Rapidly renewing mucosal epithelium at the mouth corners becomes thin and friable → fissuring, cracking, and secondary infection.
- Results in painful cracks and erythema at the corners of the lips.
9. Dysphagia and Esophageal Webs (Plummer-Vinson / Paterson-Brown-Kelly Syndrome)
Mechanism:
- The postcricoid esophageal mucosa, like other rapidly-dividing epithelia, is iron-dependent for normal renewal.
- Iron deficiency → mucosal atrophy → submucosal fibrosis → formation of thin, membranous postcricoid esophageal webs → narrowing of the lumen (symptomatic when < 12 mm).
- Results in dysphagia predominantly for solids.
- The full triad = microcytic hypochromic anemia + atrophic glossitis + esophageal webs.
- Of clinical importance: Plummer-Vinson syndrome is a premalignant condition — it raises the risk of postcricoid esophageal squamous cell carcinoma. — Harrison's 22E, Yamada's Gastroenterology
10. Pica (Pagophagia / Geophagia)
Mechanism:
- Pagophagia (craving for ice) is the most specific form; geophagia (eating clay/soil) is also classic.
- Mechanism not fully understood, but iron depletion from the CNS appears central:
- Depleted iron in the basal ganglia and hypothalamus → altered dopaminergic signaling → abnormal appetitive behavior.
- Iron-containing enzymes in the CNS regulate taste perception and satiety; their deficiency may drive craving for non-nutritive substances.
- Interestingly, chewing ice may temporarily stimulate cerebral blood flow, providing brief cognitive relief, which may reinforce pagophagia.
- Pica resolves with iron supplementation, confirming the causal relationship. — Robbins Pathologic Basis of Disease
11. Restless Legs Syndrome (RLS)
Mechanism:
- Iron is required as a cofactor for tyrosine hydroxylase, the rate-limiting enzyme in dopamine synthesis.
- Iron deficiency in the substantia nigra and basal ganglia → reduced dopamine synthesis/activity → impaired inhibitory control of spinal sensorimotor pathways → abnormal leg sensations and urge to move at night.
- RLS also occurs in renal failure, pregnancy, and Parkinson's disease — all states where iron bioavailability to the CNS is compromised.
- Symptoms (uncomfortable crawling/burning leg sensations, worse at rest, worse at night) improve with iron supplementation. — Goldman-Cecil Medicine
12. Cold Intolerance
Mechanism:
- Iron is necessary for the activity of thyroid peroxidase (an iron hemoprotein) which synthesizes thyroid hormones.
- Iron deficiency reduces thyroid hormone biosynthesis and impairs peripheral deiodination of T4 → reduced basal metabolic rate → impaired thermogenesis → cold intolerance.
- Also, reduced mitochondrial ATP production limits heat generation in peripheral tissues.
- Note: cold intolerance can persist even after hemoglobin normalizes if iron stores remain depleted. — Goldman-Cecil Medicine
13. Pruritus and Easy Bruising
Mechanism:
- Pruritus: iron-dependent enzymes maintain skin barrier integrity; their depletion → dry, atrophic skin → generalized itch.
- Easy bruising: in severe IDA, platelet function may be mildly impaired (iron depletion affects platelet mitochondria and membrane lipid metabolism), though thrombocytosis (reactive, from EPO stimulation and marrow stress) is more common than thrombocytopenia.
14. Splenomegaly (Uncommon)
Mechanism:
- Extramedullary hematopoiesis: in severe, prolonged IDA (especially in children), the spleen may attempt to compensate for inadequate bone marrow red cell production.
- Also, increased RBC destruction of abnormally shaped microcytic cells occurs in the splenic sinusoids. — Henry's Clinical Diagnosis and Management
15. Microcytic Hypochromic Anemia on Blood Smear
Mechanism:
- Microcytosis (MCV < 80 fL): Iron deficiency → insufficient hemoglobin synthesis → developing erythroblasts must undergo extra divisions to achieve critical hemoglobin threshold → smaller cells with fewer divisions needed.
- Hypochromia: Reduced hemoglobin content per cell → enlarged zone of central pallor (> 1/3 diameter of the cell).
- Anisocytosis (elevated RDW): Mixed population of normal residual cells and newly produced microcytic cells → variable cell sizes.
- Poikilocytosis: Misshapen cells including pencil cells (elliptocytes), target cells, and occasionally ring forms.
- These morphological changes are late findings — they appear only after bone marrow and cytochrome iron stores are both depleted. — Rosen's Emergency Medicine
Summary Table
| Sign / Symptom | Primary Mechanism | Pathophysiologic Basis |
|---|---|---|
| Fatigue | ↓ ATP, ↓ O₂ delivery | Impaired mitochondrial oxidative phosphorylation; ↓ myoglobin |
| Pallor | ↓ Hb per RBC | Hypochromia; ↓ oxyhemoglobin in tissues |
| Dyspnea / Palpitations | ↓ O₂ delivery | Compensatory ↑ HR, ↑ RR; high-output state |
| Angina | ↑ cardiac demand + ↓ O₂ supply | Anemia-induced high-output state |
| Headache / poor concentration | ↓ CNS iron | ↓ Dopamine synthesis; ↓ cerebral O₂ |
| Koilonychia | ↓ Nail keratinocyte iron | Impaired keratin cross-linking → nail softening → spoon deformity |
| Alopecia | ↓ Hair matrix cell iron | ↓ DNA synthesis → shortened anagen phase |
| Glossitis | ↓ Tongue epithelial iron | Mucosal atrophy; ↓ papillary proliferation |
| Angular stomatitis | ↓ Oral mucosal iron | Epithelial thinning, fissuring at commissures |
| Dysphagia / Esophageal webs | ↓ Esophageal mucosal iron | Mucosal atrophy → submucosal fibrosis → postcricoid webs (Plummer-Vinson) |
| Pica / Pagophagia | ↓ CNS iron | Altered dopaminergic signaling; hypothalamic iron depletion |
| Restless Legs Syndrome | ↓ CNS dopamine | ↓ Tyrosine hydroxylase activity (iron-dependent) |
| Cold intolerance | ↓ Thyroid hormone | ↓ Thyroid peroxidase (iron hemoprotein) activity |
| Microcytosis / Hypochromia | ↓ Hb synthesis in marrow | Extra cell divisions; insufficient heme production per cell |
Key clinical principle: Non-hematologic symptoms (fatigue, cognitive impairment, koilonychia, pica, RLS, cold intolerance) reflect tissue iron depletion and often precede overt anemia by weeks to months. Treating to a normal hemoglobin alone is insufficient — iron stores (ferritin) must be replenished to resolve the full symptom burden.
Sources: Robbins, Cotran & Kumar Pathologic Basis of Disease; Goldman-Cecil Medicine; Harrison's Principles of Internal Medicine 22E; Rosen's Emergency Medicine; Henry's Clinical Diagnosis and Management by Laboratory Methods
why target cells in iron deficiency anemia
Finding Sources
Finding Sources
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.