How to use you properly
Here's how to get the best out of me:
What I Can Do
Research & Knowledge
- Answer questions across all domains — science, history, law, technology, culture, math, and more
- Search the web for current information (news, prices, recent events)
- Search and summarize peer-reviewed medical literature (PubMed)
- Look up content in a medical textbook library for clinical questions
Document & File Work
- Create and edit Word documents (.docx), PowerPoint presentations (.pptx), Excel spreadsheets (.xlsx), and PDFs
- Read and extract content from uploaded files (PDFs, Word docs, spreadsheets, images, etc.)
- Generate images and visual diagrams from descriptions
Code & Data
- Write, debug, and explain code in any language
- Run code in a live sandbox (Python, Node.js, shell scripts, etc.)
- Analyze data, generate charts, and produce reports
How to Get the Best Results
Be specific about your goal. Instead of "tell me about diabetes," try "explain the mechanism of insulin resistance in Type 2 diabetes for a medical student."
Mention the format you want. "Give me a bullet-point summary," "write this as a formal email," or "create a Word document" all produce different outputs.
Attach files when relevant. If you have a PDF, spreadsheet, or image, attach it and I'll work with it directly.
Ask follow-up questions. I iterate — if my first answer misses the mark, tell me what to adjust.
For medical questions, I consult authoritative textbooks and PubMed automatically before answering, so you'll get evidence-based responses.
Tips
- You can ask me to do complex multi-step tasks ("research X, then create a PowerPoint from the findings")
- I'll ask clarifying questions when your request is ambiguous — this saves time
- I can run things in parallel (e.g. research multiple topics at once)
- If I make a mistake, just point it out and I'll correct it
What would you like to work on?
Let’s study copd
Asking for Preferences
Reading File
Finding Sources
Searching PubMed
"chronic obstructive pulmonary disease"[MeSH Terms] AND "management"[MeSH Terms]
Reading File
Reading File
Reading File
Reading File
Finding Sources
Searching Images
COPD emphysema pathology diagram
I have comprehensive content. Let me now build the full study guide.
COPD — Complete Medical Student Study Guide
1. Definition
Chronic Obstructive Pulmonary Disease (COPD) is a progressive, not fully reversible airflow limitation caused by an abnormal inflammatory response of the lung to noxious particles or gases — most commonly cigarette smoke. It most commonly manifests as emphysema and/or chronic bronchitis, which frequently coexist.
Key distinction from asthma: COPD obstruction is not fully reversible with treatment.
2. Epidemiology & Risk Factors
| Factor | Details |
|---|---|
| #1 Risk Factor | Cigarette smoking (15–30% of smokers develop COPD*) |
| Genetic | α₁-antitrypsin (AAT) deficiency → panacinar emphysema |
| Environmental | Occupational dust, air pollution, biomass fuel smoke |
| Age | Typically older patients (>40 years) |
| Burden | 3rd most common cause of death in the US; >$40 billion/year in healthcare costs |
*Note: CT studies now show progressive bronchial wall changes and lung tissue loss even in smokers with normal spirometry, challenging the classic 15–30% figure — Katzung's Pharmacology, 16th Ed.
3. Pathology: Two Major Subtypes
A. Emphysema ("Pink Puffer")
- Definition: Permanent enlargement of airspaces distal to terminal bronchioles with destruction of alveolar walls
- Mechanism: Proteases (especially neutrophil elastase) destroy elastic support → loss of elastic recoil → air trapping
- Protease–antiprotease imbalance is the core concept: smoking activates neutrophils/macrophages → ↑ elastase; simultaneously, smoke inactivates α₁-antitrypsin
| Subtype | Location | Cause |
|---|---|---|
| Centriacinar (centrilobular) | Upper lobes; central part of acinus | Smoking (most common) |
| Panacinar (panlobular) | Lower lobes; entire acinus | AAT deficiency |
| Paraseptal | Subpleural | Associated with spontaneous pneumothorax |
Clinical features:
- Barrel chest (increased AP diameter)
- Pursed-lip breathing, tripod positioning
- Dyspnea >> cough
- Relatively preserved oxygenation at rest ("pink puffer")
- Hyperresonance on percussion
B. Chronic Bronchitis ("Blue Bloater")
- Definition (clinical): Productive cough for ≥3 consecutive months in ≥2 consecutive years
- Mechanism: Mucus overproduction from hyperplasia of tracheal/large airway mucous glands + goblet cell metaplasia; airway obstruction from small airway inflammation (chronic bronchiolitis)
- Reid index (gland thickness / bronchial wall thickness) is increased (>0.4)
- Histology: Enlarged mucus glands, goblet cell metaplasia, inflammation, bronchiolar wall fibrosis
Clinical features:
- Prominent productive cough
- Cyanosis (hypoxemia + hypercapnia → "blue bloater")
- Frequent respiratory infections (impaired mucociliary clearance → persistent Haemophilus influenzae infection)
In practice, most patients have mixed features of both subtypes.
4. Pathophysiology
The core defect is obstructive ventilatory pattern:
| Parameter | COPD | Normal |
|---|---|---|
| FEV₁ | ↓↓ | Normal |
| FVC | Normal or mildly ↓ | Normal |
| FEV₁/FVC ratio | < 0.70 (post-bronchodilator) | ≥ 0.70 |
| TLC | ↑ (air trapping) | Normal |
| RV | ↑↑ | Normal |
| DLCO | ↓ (emphysema destroys alveolar surface) | Normal |
V/Q mismatch is the central gas exchange defect:
- Poorly ventilated alveoli receive blood → shunt-like effect → ↓ PaO₂
- Compensatory hyperventilation (if possible) → ↓ PaCO₂ in early disease
- As disease progresses → hypercapnia (CO₂ retention) + hypoxemia
Clinical physiology case (Costanzo Physiology): A 65-year-old with 40 pack-years had PaO₂ = 60 mmHg vs. calculated PAO₂ = 113 mmHg. The A-a gradient confirms V/Q mismatch. His low PaCO₂ (30 mmHg) reflects compensatory hyperventilation from hypoxic drive. FEV₁/FVC < 0.70 confirms obstructive disease.
5. GOLD Staging (Spirometry-Based)
Based on post-bronchodilator FEV₁ % predicted (all require FEV₁/FVC < 0.70):
| GOLD Grade | Severity | FEV₁ % predicted |
|---|---|---|
| GOLD 1 | Mild | ≥ 80% |
| GOLD 2 | Moderate | 50–79% |
| GOLD 3 | Severe | 30–49% |
| GOLD 4 | Very Severe | < 30% |
Staging also incorporates symptom burden (mMRC dyspnea scale or CAT score) and exacerbation history to guide treatment.
6. Clinical Features & Diagnosis
Symptoms:
- Progressive exertional dyspnea (cardinal symptom)
- Chronic productive cough and sputum
- Wheezing
Signs:
- Barrel chest, prolonged expiratory phase
- Use of accessory muscles
- Decreased breath sounds, wheeze on auscultation
- Cyanosis (in bronchitic type)
- Cor pulmonale (RV failure from pulmonary HTN) in advanced disease → JVD, peripheral edema
Investigations:
| Test | Finding |
|---|---|
| Spirometry | FEV₁/FVC < 0.70 (confirmatory) |
| CXR | Hyperinflation, flattened diaphragms, bullae |
| CT chest | Gold standard for emphysema subtype; detects bullae, air trapping |
| ABG | ↓ PaO₂, ↑ or ↓ PaCO₂ depending on stage |
| α₁-AT level | Screen if young, non-smoker, or lower-lobe predominant emphysema |
| CBC | Secondary polycythemia in chronic hypoxemia |
CT imaging of COPD — centrilobular emphysema:
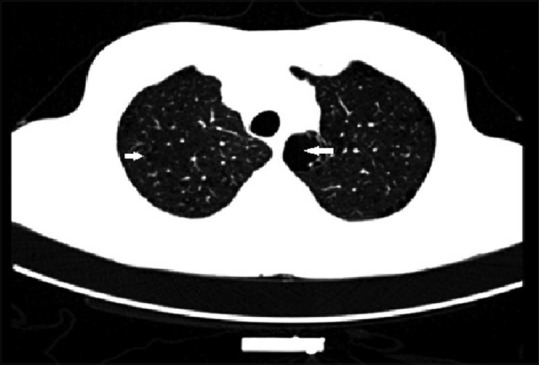
CT chest: Centrilobular emphysema (focal low-attenuation intraparenchymal lucencies) and paraseptal emphysema (subpleural lucencies). Hallmark of smoking-related COPD.
7. Management
Non-Pharmacological (cornerstone)
- Smoking cessation — only intervention proven to slow FEV₁ decline
- Pulmonary rehabilitation
- Supplemental O₂ if PaO₂ ≤55 mmHg (or ≤59 with cor pulmonale/polycythemia) → shown to reduce mortality
- Vaccinations (influenza, pneumococcal, COVID-19)
Pharmacological (stepwise by symptom/severity)
| Severity | Drug class | Examples |
|---|---|---|
| All symptomatic | SABA (rescue) | Albuterol, salbutamol |
| Persistent dyspnea | LAMA | Tiotropium |
| Persistent dyspnea | LABA | Salmeterol, formoterol |
| Severe/frequent exacerbations | LABA + LAMA | Dual bronchodilator |
| High eosinophils + exacerbations | ICS + LABA | Fluticasone/salmeterol |
| Triple therapy | ICS + LABA + LAMA | Most severe patients |
| Chronic bronchitis + exacerbations | Roflumilast (PDE4 inhibitor) | Reduces exacerbation frequency |
Important caveats (Katzung, 16th Ed.):
- ICS are less effective in COPD than asthma; associated with ↑ bacterial pneumonia risk → reserve for high exacerbation risk or high blood eosinophils
- Low-dose theophylline failed to reduce exacerbations in a large RCT — no longer recommended as standard
- Blood eosinophil count guides ICS use (low eosinophils = unlikely benefit; high eosinophils = reasonable benefit)
8. COPD Exacerbations
Definition: Acute worsening of respiratory symptoms beyond normal day-to-day variation, requiring a change in medication.
Triggers: Viral respiratory infections (most common), bacterial infections (H. influenzae, S. pneumoniae, M. catarrhalis), air pollution.
Management of acute exacerbation:
- O₂ — titrate to SpO₂ 88–92% (avoid over-oxygenation → hypercapnic drive suppression)
- SABA ± SAMA (nebulized bronchodilators)
- Systemic corticosteroids (oral prednisolone 40mg × 5 days)
- Antibiotics — β-lactams, doxycycline, azithromycin (cover H. influenzae) for purulent sputum or respiratory failure
- NIV (BiPAP) for hypercapnic respiratory failure (pH < 7.35, PaCO₂ > 45 mmHg)
9. Complications
| Complication | Mechanism |
|---|---|
| Cor pulmonale | Chronic hypoxemia → pulmonary vasoconstriction → right heart failure |
| Secondary polycythemia | Chronic hypoxemia → ↑ EPO → ↑ RBC production |
| Spontaneous pneumothorax | Rupture of subpleural blebs (especially paraseptal emphysema) |
| Respiratory failure | Progressive loss of lung function |
| Malnutrition/cachexia | Systemic inflammation, increased WOB |
10. Key Comparisons to Remember
| Feature | Emphysema | Chronic Bronchitis | Asthma |
|---|---|---|---|
| Inflammation | Neutrophilic | Neutrophilic | Eosinophilic |
| Reversibility | Irreversible | Poorly reversible | Reversible |
| Main complaint | Dyspnea | Cough/sputum | Episodic wheeze |
| PaO₂ | Relatively preserved (early) | Low | Normal (inter-episode) |
| PaCO₂ | Low (early), rises late | High | Low |
| Prognosis | Progressive | Progressive | Generally good |
High-Yield Points for Exams
- FEV₁/FVC < 0.70 post-bronchodilator = diagnostic criterion
- Smoking cessation + long-term O₂ are the only interventions that reduce mortality
- α₁-AT deficiency → panacinar emphysema, lower-lobe predominant
- Roflumilast = PDE4 inhibitor, used when chronic bronchitis + frequent exacerbations
- ICS use guided by eosinophil count (not routine in all COPD)
- COPD exacerbations: treat with SABDs + steroids + antibiotics ± NIV; target SpO₂ 88–92%
- Cor pulmonale = late complication; indicated by peripheral edema, JVD, elevated JVP
Sources: Robbins & Kumar Basic Pathology; Costanzo Physiology 7th Ed.; Katzung's Basic & Clinical Pharmacology 16th Ed.; Fishman's Pulmonary Diseases & Disorders.
Want me to go deeper on any section — pathophysiology, pharmacology, ABG interpretation in COPD, or practice questions?
This is a shared conversation. Sign in to Orris to start your own chat.