Bronchopleural fistula full details for md exams 15marks answer
Reading File
Finding Sources
Finding Sources
Searching PubMed
"bronchopleural fistula" management
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Searching Images
bronchopleural fistula chest X-ray post pneumonectomy
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive textbook material and excellent images. Let me compose the complete MD exam answer.
Bronchopleural Fistula (BPF) — Complete MD Examination Answer
Definition
A bronchopleural fistula (BPF) is an abnormal communication between the bronchial tree (main, lobar, or segmental bronchus) and the pleural cavity. Occasionally, when there is an additional communication to the surface of the chest wall, it is termed a cutaneous BPF.
- Murray & Nadel's Textbook of Respiratory Medicine: "A bronchopleural fistula is a direct connection between the bronchus and the pleural space."
- Barash's Clinical Anesthesia: "A BPF is an abnormal communication between the bronchial tree and the pleural cavity."
Classification
| Type | Timing | Mechanism |
|---|---|---|
| Post-surgical (early) | Within 7 days of resection | Technical failure of bronchial closure; ischaemia |
| Post-surgical (late) | After 7 days | Stump infection/necrosis; tumour recurrence; empyema erosion |
| Non-surgical | Spontaneous | Necrotising infection, barotrauma, spontaneous rupture |
| Cutaneous BPF | Any | Additional communication to chest wall surface |
Aetiology & Risk Factors
1. Post-Resection (Most Common)
- Pneumonectomy — incidence 2–11%, mortality 5–70% (Miller's Anesthesia)
- Lobectomy — less common; manifests as persistent air leak with purulent drainage
- The key technical point is ensuring the bronchial stump is as flush as possible to the carina to prevent secretion pooling → infection → stump breakdown (Murray & Nadel)
Risk factors for post-resection BPF:
- Right-sided pneumonectomy (longer stump, less vascular supply)
- Pre-operative radiotherapy or chemotherapy
- Malnutrition and poor wound healing
- Empyema or residual tumour at stump
- Prolonged mechanical ventilation post-operatively
- Diabetes mellitus
- Steroid use
2. Necrotising Infections
- Lung abscess rupturing into pleural space
- Necrotising pneumonia (Staphylococcus, Klebsiella, Aspergillus)
- Empyema eroding into bronchus (empyema necessitans)
- Tuberculous cavitation
3. Traumatic / Barotraumatic
- Rupture of bulla, cyst, or parenchymal tissue
- Penetrating chest wounds
- Iatrogenic barotrauma (PEEP-induced rupture)
4. Neoplastic
- Erosion of a bronchus by carcinoma or chronic inflammatory disease (Miller's Anesthesia)
Pathophysiology
- Persistent air communication: Air flows preferentially through the fistula rather than alveoli → loss of tidal volume → impaired ventilation
- Impediment to healing: Continuous airflow through the fistula inhibits granulation tissue formation and wound healing
- Tension pneumothorax: Positive-pressure ventilation forces air through fistula into pleural space → progressive pressure build-up → mediastinal shift → compromised venous return
- Contamination of healthy lung: If empyema is present, purulent material can flood the contralateral lung → aspiration pneumonia/ARDS
- CO₂ retention: Decreased effective alveolar ventilation leads to hypercapnia
- Persistent pleural space: If the lung cannot expand to fill the cavity, the fistula cannot close
The cycle of infection → inflammation → stump necrosis → fistula enlargement → worsening empyema perpetuates the problem.
Clinical Features
Symptoms
- Sudden onset of dyspnoea (most common presenting feature)
- Productive cough — expectoration of sero-sanguineous or purulent sputum (pleural fluid coughed up)
- Subcutaneous emphysema
- Fever, malaise
- Haemoptysis
- Characteristic sign: Patient coughs up the colour of the methylene blue or pleural dye injected into the chest
Signs
- Contralateral tracheal deviation (mediastinal shift)
- Ipsilateral hyperresonance / absent breath sounds
- Subcutaneous crepitus
- Fever, tachycardia, signs of sepsis
- Signs of tension pneumothorax (if large BPF with PPV): hypotension, tracheal deviation, absent breath sounds
Post-Pneumonectomy BPF — Classic Triad:
- Sudden dyspnoea
- Subcutaneous emphysema
- Contralateral deviation of trachea
- Decrease in fluid level on serial chest radiographs (Miller's Anesthesia)
Investigations
1. Chest Radiograph (CXR)
- Persistent pneumothorax / hydropneumothorax
- New air-fluid level in the pleural cavity
- Decrease in the fluid level on serial post-pneumonectomy radiographs — classic sign of BPF (fluid escaping via fistula into airway, coughed up)
- Contralateral mediastinal shift
- Widening of intercostal spaces
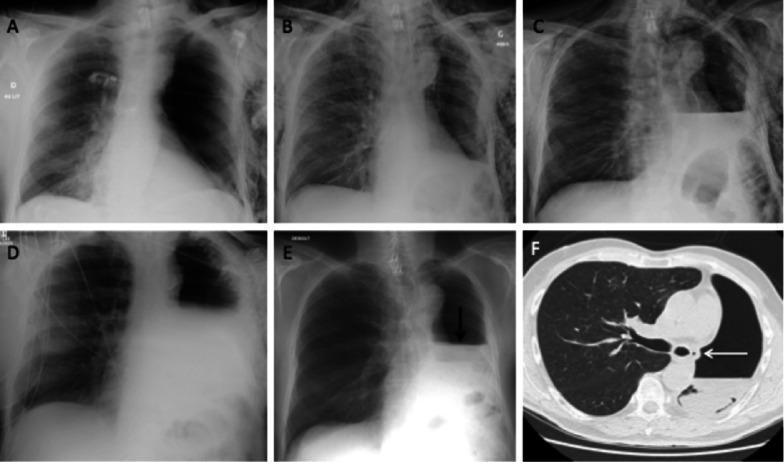
Serial chest X-rays (A–E) and CT (F) demonstrating post-pneumonectomy BPF. The sudden DROP in the fluid level on Day 28 (Panel E, arrow) is pathognomonic. CT shows air bubble adjacent to the bronchial stump (Panel F, arrow).
2. CT Chest
- Most sensitive — shows air bubbles adjacent to bronchial stump
- Localises the fistula
- Detects underlying lung abscess, empyema, malignancy
3. Bronchoscopy (Gold Standard for Confirmation)
- Direct visualisation of fistula (if proximal)
- Balloon sequential occlusion to localise the affected segment (distal fistulae)
- Guides therapeutic intervention

4. Methylene Blue Test
- Inject methylene blue into the pleural space → recovery of dye in sputum confirms BPF
5. Radionuclide Studies
- Inhalation of Xenon-133 or O₂/N₂O mixture → accumulation of radionuclide in pleural space confirms the fistula (Miller's Anesthesia)
6. Sinogram / Bronchography
- Injection of contrast into pleural drain or bronchus with fluoroscopic imaging
7. Quantification of Air Leak
- Chest tube observation: Intermittent bubbling = small fistula; Continuous bubbling = large BPF
- Spirometry: Difference between inhaled and exhaled tidal volumes (via tight-fitting mask or intubated patient) quantifies air leak size (Miller's Anesthesia)
Management
Pre-treatment Principles
- Isolate and drain any empyema before anaesthesia (patient sitting up, leaning towards affected side)
- Chest drain to underwater seal — must remain unclamped at all times
- Position patient with affected side dependent (down) to prevent spillage into healthy lung
A. Conservative / Medical Management
-
Ventilatory strategies to reduce airflow through fistula:
- Low tidal volumes (permissive hypercapnia)
- Minimise PEEP
- Shorten inspiratory time
- High-frequency oscillatory ventilation (HFOV / HFJV) — small tidal volumes → minimal gas loss through fistula → promotes healing
- Isolated contralateral lung ventilation
- Dependent positioning (BPF side down)
-
Chest tube drainage — for pneumothorax and empyema
-
Antibiotics — for associated infection/empyema
B. Bronchoscopic (Interventional) Management
Used when conservative measures fail. Balloon sequential segmental occlusion first localises the fistula, then therapeutic options include:
| Agent/Device | Mechanism |
|---|---|
| Silver nitrate | Chemical cauterisation |
| Cyanoacrylate glue (fibrin glue, BioGlue) | Mechanical sealing |
| Gelatin sponge (Gelfoam) | Plug formation |
| Autologous blood patch | Fibrin clot sealing |
| Thrombin injection | Fibrin clot |
| Endobronchial valves (one-way valves) | Allow outflow, prevent inflow → atelectasis → healing |
| Bronchial blockers | Temporary occlusion |
| Fishing weights / coils | Mechanical occlusion |
The endobronchial valve is the most recent and effective approach — complete/partial air leak resolution in the majority of patients (Fishman's Pulmonary Diseases and Disorders)
Duration: BPF refractory to medical therapy for >7 days requires pleurodesis, endobronchial valve, or surgery (Murray & Nadel)
C. Surgical Management
1. Early Post-Pneumonectomy BPF
- Re-suture of the bronchial stump (if technically feasible)
- Reinforcement with muscle flap (intercostal, pectoralis, latissimus dorsi, omentum)
2. Late/Chronic BPF — Clagett Procedure
- Open pleural drainage (creating a thoracic window — Eloesser flap)
- Irrigation of the cavity with antiseptic solutions (povidone-iodine, antibiotics)
- Once sterile: cavity obliteration with antibiotic solution filling (Barash's Clinical Anesthesia, Harrison's)
- Muscle flap reinforcement of bronchial stump
3. Non-Pneumonectomy BPF
- If lung expands to fill thoracic cavity → chest tube drainage alone usually suffices
- Large fistula + large persistent pleural space → unlikely to close conservatively → surgical resection required (Miller's Anesthesia)
4. VATS (Video-Assisted Thoracoscopic Surgery)
- Endoscopic stapling and removal of bulla or fistula
- Indicated for recurrent pneumothorax with BPF (Washington Manual)
D. Anaesthetic Management (Surgically Important for MD Exams)
The three major intraoperative challenges are (Miller's Anesthesia):
- Need for lung isolation to protect healthy lung from contamination
- Risk of tension pneumothorax with positive-pressure ventilation
- Inadequate ventilation due to air leak from fistula
Preferred technique:
- Awake fibreoptic intubation with a Double-Lumen Tube (DLT) — patient breathing spontaneously (avoids tension pneumothorax)
- Bronchial lumen placed on the side OPPOSITE the BPF
- Immediate suctioning on tracheal lumen if empyema present (pus outpouring)
- Chest drain must remain unclamped throughout
- Extubate as soon as possible after repair (Morgan & Mikhail)
Alternative ventilatory strategies with DLT:
- Healthy lung: normal tidal volume
- Affected lung: smaller tidal volume OR CPAP with oxygen at pressures just below the critical opening pressure of the fistula
- HFJV for large BPF (Barash's Clinical Anesthesia)
Complications of BPF
| Complication | Mechanism |
|---|---|
| Empyema | Infection of persistent pleural space |
| Tension pneumothorax | Air trapping during PPV |
| Aspiration pneumonia / ARDS | Contamination of healthy lung |
| CO₂ retention / respiratory failure | Loss of effective tidal volume |
| Septicaemia | Systemic spread from pleural infection |
| Death | Mortality 5–70% post-pneumonectomy |
Prognosis
- Small fistulae (esp. alveolo-pleural) — heal spontaneously with conservative management
- Persistent BPF after lung resection — mortality 5–70% depending on timing and cause (Miller's Anesthesia)
- Outcomes improved with:
- Early recognition and drainage
- Lung isolation before anaesthesia
- Muscle flap reinforcement
- Endobronchial valve technology
Summary Flowchart
SUSPECT BPF
│
├─► CXR: Dropped fluid level / new air-fluid level
├─► CT Chest: Air near bronchial stump
└─► Bronchoscopy: Confirm + localise
│
▼
DRAIN EMPYEMA FIRST (if present)
│
▼
CONSERVATIVE: Low TV, HFJV, positioning, chest drain
│
Fails >7 days?
│
▼
BRONCHOSCOPIC: Endobronchial valve / glue / silver nitrate
│
Fails?
│
▼
SURGICAL: Clagett procedure / muscle flap / re-suture stump
Key High-Yield Points for MD Exam
| Point | Detail |
|---|---|
| Most common cause | Post-pneumonectomy stump dehiscence |
| Classic radiological sign | Sudden drop in post-pneumonectomy fluid level on serial CXR |
| Best confirmatory test | Bronchoscopy |
| Gold standard localisation | Sequential balloon occlusion bronchoscopy |
| Preferred anaesthetic technique | Awake fibreoptic DLT, spontaneous breathing |
| Medical duration before surgery | >7 days of failed conservative therapy |
| Late chronic BPF surgery | Clagett procedure |
| Newer minimally invasive Rx | Endobronchial one-way valves |
| Key pathophysiology with PPV | Risk of tension pneumothorax |
| Mortality (post-pneumonectomy) | 5–70% |
Sources: Miller's Anesthesia 10e · Murray & Nadel's Textbook of Respiratory Medicine · Barash's Clinical Anesthesia 9e · Morgan & Mikhail's Clinical Anesthesiology 7e · Fishman's Pulmonary Diseases and Disorders · Grainger & Allison's Diagnostic Radiology · Harrison's Principles of Internal Medicine 22e · Washington Manual of Medical Therapeutics
This is a shared conversation. Sign in to Orris to start your own chat.