cell structure and differentiation
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Here is a comprehensive overview of Cell Structure and Differentiation drawn from authoritative medical textbooks.
Cell Structure and Differentiation
1. Overview of Cell Compartments
Every human cell is divided into two major compartments:
- Cytoplasm — everything outside the nucleus; contains organelles, cytoskeleton, and the aqueous cytoplasmic matrix (ions, metabolites, proteins, RNA)
- Nucleus — the largest organelle; houses the genome and the machinery for DNA replication and RNA transcription
"The cytoplasm and nucleus not only play distinct functional roles but also work in concert to maintain the cell's viability." — Histology: A Text and Atlas with Correlated Cell and Molecular Biology
2. Cellular Diversity Through Specialization
Although all cells share the same fundamental organelles, individual cells develop certain structures to exceptional degrees, giving rise to distinct morphologies. Examples:
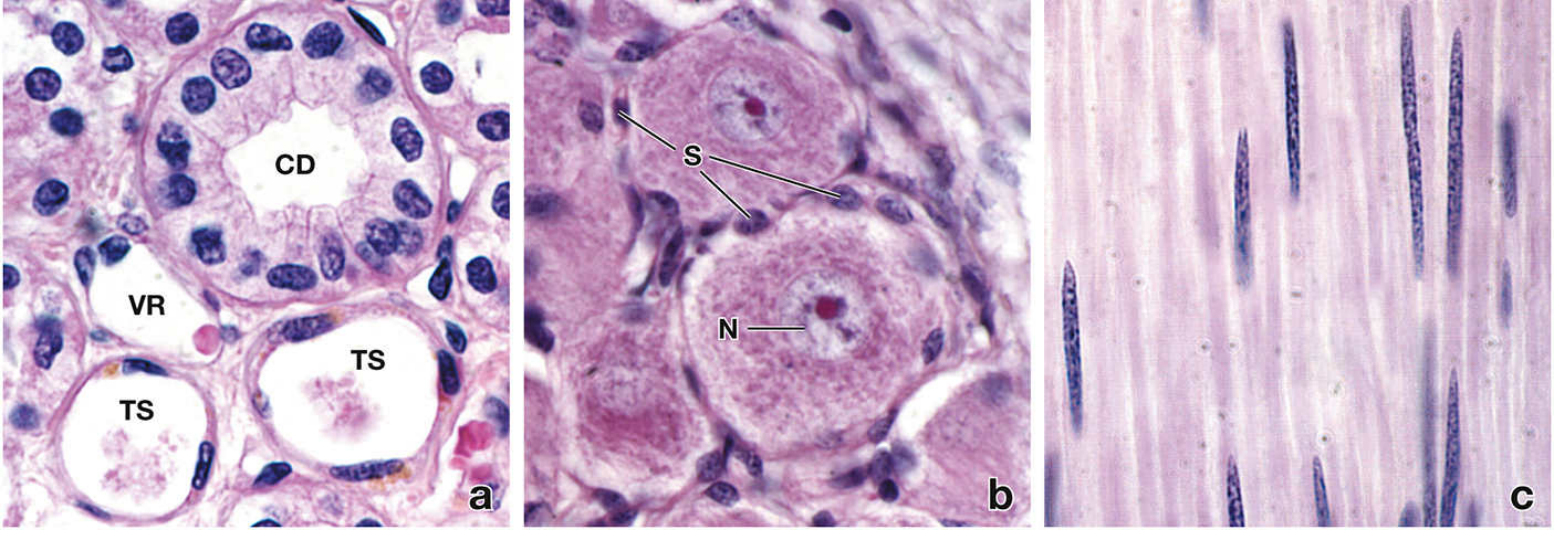
- Muscle cells — huge quantities of contractile filaments (actin/myosin)
- Neurons — large euchromatic nuclei and extensive rough ER (Nissl bodies) for axonal protein synthesis
- Secretory cells — prominent Golgi apparatus and vesicles
- Shape, nuclear morphology, and cytoplasmic content all reflect functional specialization.
3. Key Organelles and Their Functions
| Organelle / Structure | Function |
|---|---|
| Plasma membrane | Lipid bilayer; selectively permeable barrier; houses receptors, transport proteins, channels |
| Nucleus | DNA replication, transcription; genetic control |
| Endoplasmic reticulum (RER/SER) | RER: synthesis of secretory/membrane proteins; SER: lipid synthesis, Ca²⁺ storage |
| Golgi apparatus | Protein sorting and packaging; vesicle formation for secretion and membrane delivery |
| Mitochondria | ATP synthesis via oxidative phosphorylation |
| Lysosomes | Hydrolytic degradation of proteins and macromolecules |
| Peroxisomes | Reactions using/producing H₂O₂; fatty acid oxidation |
| Cytoskeleton | Actin microfilaments, intermediate filaments, tubulin microtubules — maintains shape, anchors organelles, enables motility |
The Plasma Membrane — Fluid Mosaic Model
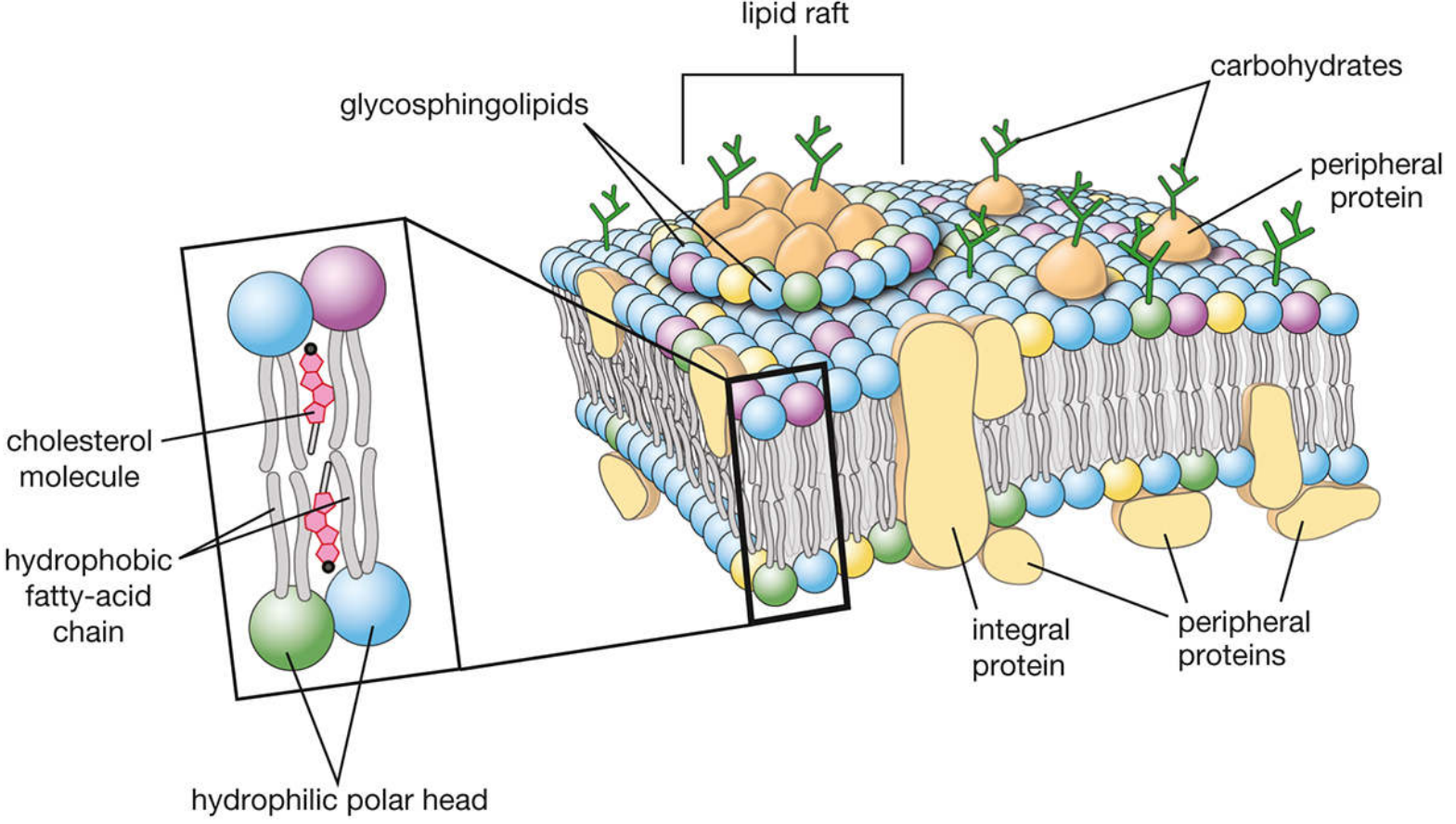
The membrane is a phospholipid bilayer (~8–10 nm thick) with:
- Hydrophobic fatty acid chains facing inward
- Hydrophilic polar heads forming inner/outer surfaces
- Integral proteins spanning the bilayer (transport, receptors)
- Peripheral proteins attached to surfaces (structural, signaling)
- Cholesterol modulating fluidity
- Lipid rafts — microdomains enriched in glycosphingolipids and cholesterol, concentrating signaling proteins
- Glycocalyx — carbohydrate chains on the extracellular surface for cell recognition and adhesion
Transport across the membrane occurs via active transport, facilitated diffusion, or pores/gated channels.
4. Cell Signaling and Chemical Messengers
Differentiated cells integrate signals from their environment. Chemical messengers include:
| Type | Example | Range |
|---|---|---|
| Endocrine | Hormones | Bloodstream (long-range) |
| Paracrine | Growth factors, cytokines | Neighboring cells |
| Juxtacrine | Notch ligands | Direct cell-cell contact |
| Autocrine | Some cytokines | Same cell |
Signals ultimately alter metabolic enzymes, gene regulatory proteins, ion channels, or cytoskeletal proteins in the target cell.
5. Cell Differentiation
General Principle
In most tissues, only primitive undifferentiated (stem) cells are capable of proliferating. As cells differentiate and acquire specialized functions, they progressively lose the ability to divide. This trade-off between proliferative capacity and functional specialization is fundamental to tissue organization.
"Normal cells have a large number of safeguards against uncontrolled proliferation, and cells lose the capacity to proliferate as they differentiate and acquire functional capabilities." — Harrison's Principles of Internal Medicine, 22nd Ed.
Mechanisms Driving Differentiation
- Intrinsic gene expression changes — transcription factors activate lineage-specific programs
- Niche-directed signals — local microenvironment provides adhesion molecules, cytokines, and matrix cues
- Epigenetic regulation — chromatin remodeling silences pluripotency genes and activates lineage genes
- Notch, STING, and other pathways — maintain quiescence or promote transition through the cell cycle
- Cyclin-dependent kinase inhibitors (e.g., p57/CDKN1c) — block G1→S transition, enforcing stem cell quiescence
Progressive Loss of Plasticity
As cells mature: Stem cell → Progenitor → Precursor → Mature effector cell
- Multipotent stem cells give rise to all lineages
- Progenitors are restricted to a narrower set of fates
- Precursors are committed to a single lineage but still proliferate
- Mature cells are terminally differentiated — they perform specialized functions but typically cannot divide
6. Hematopoietic Differentiation — A Model System
The blood system provides the best-characterized example of differentiation in the human body:
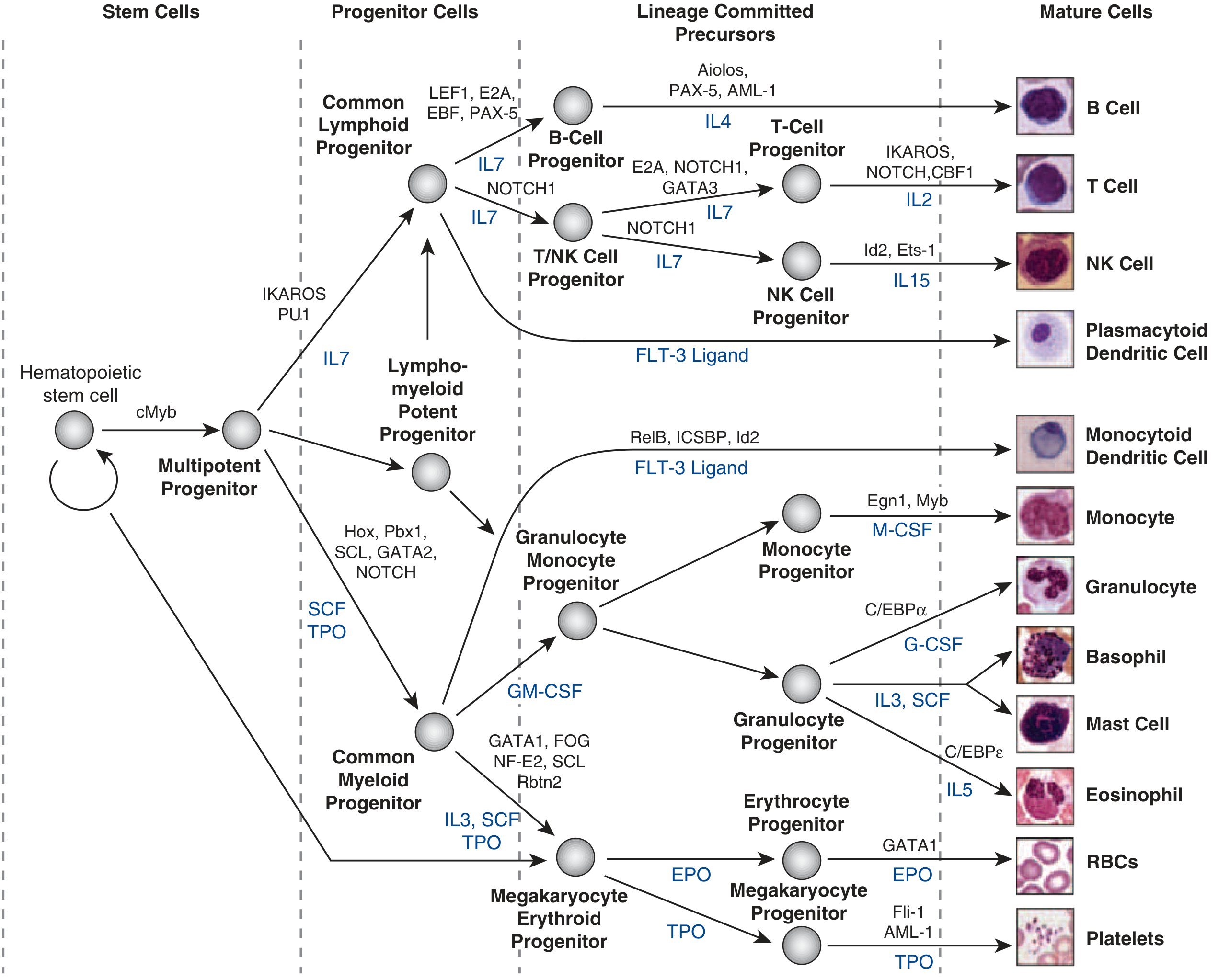
A hematopoietic stem cell (HSC) gives rise to all blood and immune cells through a branching hierarchy:
- HSC → Multipotent Progenitor (driven by cMyb)
- → Common Lymphoid Progenitor (IKAROS, PU.1) → B cells (PAX-5), T cells (NOTCH1, GATA3), NK cells
- → Common Myeloid Progenitor (GATA1, SCL) → Granulocytes (C/EBPα, G-CSF), Monocytes (M-CSF), RBCs (EPO, GATA1), Platelets (TPO)
Key cytokines driving lineage commitment include:
- IL-7 → lymphoid lineages
- EPO (erythropoietin) → red blood cells
- G-CSF → granulocytes
- M-CSF → monocytes
- TPO (thrombopoietin) → megakaryocytes/platelets
- SCF (stem cell factor) → broad HSC maintenance
Mature granulocytes cannot proliferate — numbers increase only by enhanced progenitor production. Lymphocytes retain proliferative capacity, but it is tightly linked to antigen recognition.
7. Differentiation and Cancer
Loss of normal differentiation control is central to cancer. Genetic and epigenetic alterations disrupt the balance between proliferation and differentiation, causing:
- Failure to exit the cell cycle upon differentiation cues
- Reactivation of stem cell programs in mature cells
- Clonal expansion of cells with growth advantage
The multi-step progression — hyperplasia → dysplasia → carcinoma in situ → invasive cancer — reflects accumulating defects in both cell cycle control and differentiation programs.
Sources:
- Histology: A Text and Atlas with Correlated Cell and Molecular Biology, Wolters Kluwer
- Basic Medical Biochemistry: A Clinical Approach, 6th Ed., Wolters Kluwer
- Harrison's Principles of Internal Medicine, 22nd Ed. (2025), McGraw Hill Medical
create its powerpoint pdf start with the meaning of cell
Reading File
Reading File
Reading File
Running Command
Running Command
Writing File
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.