Central retinal vein occlusion refer ak khurana
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Searching the Web
AK Khurana ophthalmology central retinal vein occlusion CRVO
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Loading Image
Loading Image
I now have all the material needed. Let me compile a comprehensive AK Khurana-style answer on Central Retinal Vein Occlusion.
Central Retinal Vein Occlusion (CRVO)
Note: AK Khurana's Comprehensive Ophthalmology is not in the library catalogue. The following is structured according to Khurana's well-known syllabus format, drawing from Kanski's Clinical Ophthalmology (10th ed.) — the authoritative ophthalmology reference available — supplemented with standard teaching points from Khurana's curriculum.
Anatomy Recap
The central retinal vein (CRV) and central retinal artery share a common adventitial sheath as they pass through the optic nerve, posterior to the lamina cribrosa. Atherosclerotic changes in the artery at this confined crossing can compress and occlude the CRV — this is the anatomical basis of CRVO.
Pathogenesis
Thrombosis occurs at or posterior to the lamina cribrosa, where the CRV is most vulnerable. Three mechanisms contribute (Virchow's triad):
- Endothelial damage — from adjacent atherosclerotic artery or inflammation
- Turbulent flow — at the narrow shared sheath crossing
- Hypercoagulability — thrombophilic states amplify risk
Once occlusion occurs → ↑ venous and capillary pressure → stagnation → retinal hypoxia → capillary endothelial damage → extravasation of blood + fluid → liberation of VEGF → macular oedema and neovascularization.
Risk Factors
| Category | Factors |
|---|---|
| Most important | Age >65 years (>50% of cases) |
| Systemic | Hypertension (2/3 of patients >50 yrs), hyperlipidaemia (1/3), diabetes mellitus (~15%) |
| Ocular | Open-angle glaucoma/ocular hypertension (major ocular risk factor), hypermetropia, shorter axial length |
| Young patients | Oral contraceptive pill (most common in young females), thrombophilia (hyperhomocysteinaemia, antiphospholipid Ab, Factor V Leiden), vasculitis (Behçet, sarcoidosis, Wegener's), myeloproliferative disorders |
| Others | Smoking, dehydration, chronic renal failure |
Classification
CRVO is classified into three types:
1. Impending (Partial) CRVO
- Occurs in younger patients; may be artificial distinction from mild non-ischaemic CRVO
- Symptoms: absent or minor transient blurring, worse on waking
- Fundus: mild venous dilatation and tortuosity, few scattered dot-blot haemorrhages, mild macular oedema
- Prognosis: usually good; some progress to ischaemic CRVO
2. Non-ischaemic CRVO (Venous Stasis Retinopathy)
- More common than ischaemic; ~1/3 progress to ischaemic CRVO within months
Clinical features:
- Symptoms: Sudden painless monocular fall in VA (variable degree)
- RAPD: Absent or mild
- Fundus:
- Tortuosity and dilatation of all 4 quadrant retinal veins
- Dot, blot, and flame haemorrhages — mild to moderate extent
- Cotton-wool spots, disc oedema, macular oedema — common but mild
- Patchy (perivenular) areas of capillary non-perfusion on FA
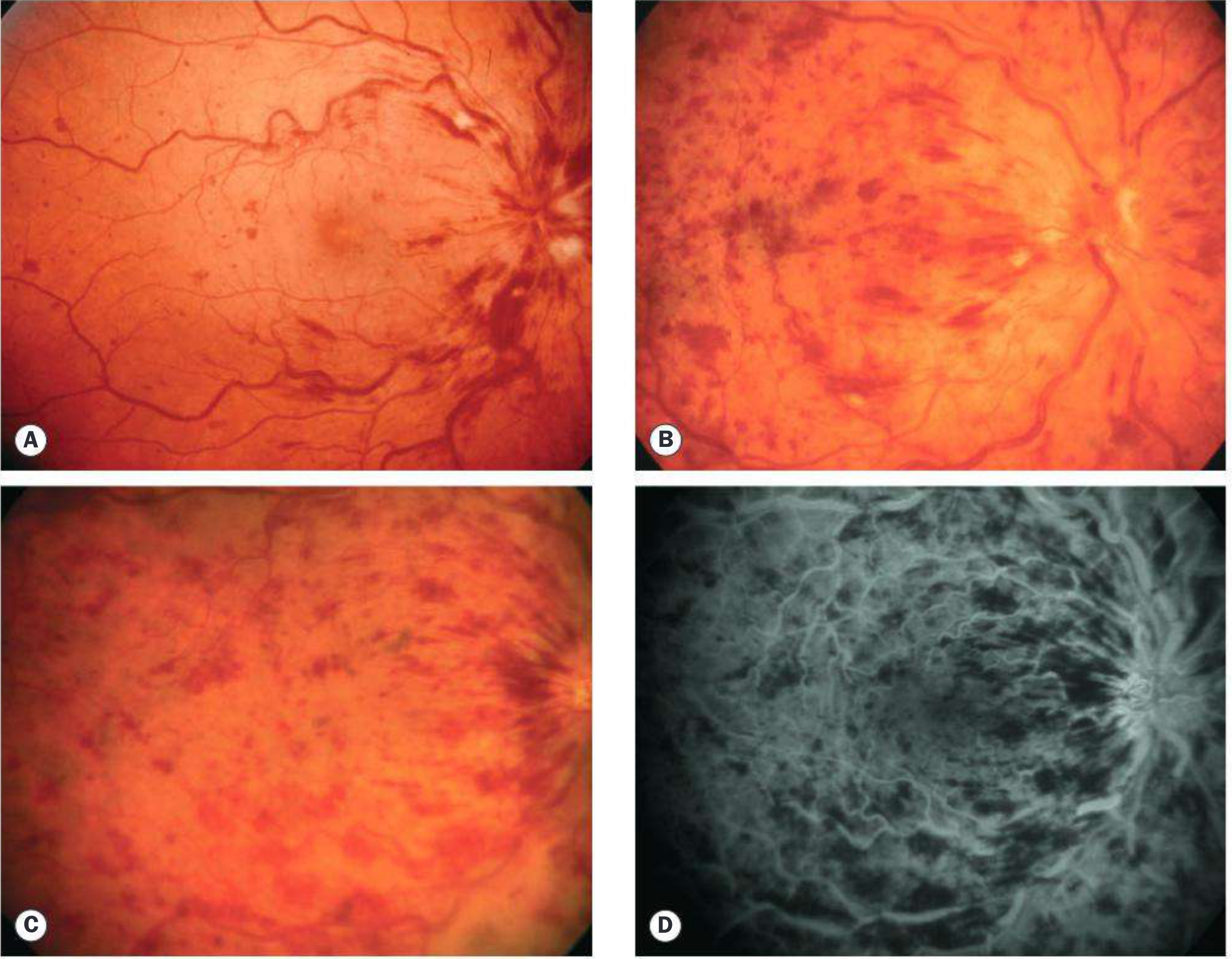
- Prognosis: Vision returns to normal/near-normal in ~50% if non-ischaemic; VA >6/60 usually portends better outcome
3. Ischaemic CRVO (Haemorrhagic Retinopathy)
- Substantially decreased retinal perfusion with extensive capillary closure and retinal hypoxia
Clinical features:
- Symptoms: Sudden, severe monocular painless visual loss; occasionally painful (NVG)
- VA: Usually counting fingers or worse
- RAPD: Present (important differentiating sign from non-ischaemic)
- Fundus:
- Severe tortuosity and engorgement of all CRV branches
- Extensive deep blot + flame haemorrhages in all 4 quadrants (periphery + posterior pole) — classic "Blood and Thunder" fundus
- Prominent cotton-wool spots (infarcts)
- Optic disc swelling and hyperaemia
- NVI (Rubeosis iridis): Develops in ~50%, usually 2–4 months post-occlusion → "100-day glaucoma" / "90-day glaucoma"
- FA: Markedly delayed arteriovenous transit, extensive capillary non-perfusion (>10 disc areas), vessel wall staining and leakage
- ERG: Depressed (used to assess neovascular risk)
Differentiating Ischaemic vs Non-ischaemic CRVO
| Feature | Non-ischaemic | Ischaemic |
|---|---|---|
| VA | Variable, often better than 6/60 | Usually CF or worse |
| RAPD | Absent/mild | Present |
| Cotton-wool spots | Few | Numerous/prominent |
| Haemorrhages | Moderate | Extensive ("blood and thunder") |
| Capillary non-perfusion on FA | <10 disc areas | >10 disc areas |
| NVI/NVG | Rare | ~50% |
| ERG | Near normal | Depressed |
| Prognosis | Better | Very poor |
Systemic Assessment
All patients:
- Blood pressure, fasting glucose, lipids
- ESR/plasma viscosity, FBC, urea/electrolytes/creatinine
- ECG
Additional in patients <50 years, bilateral CRVO, or recurrent CRVO:
- Thrombophilia screen: antiphospholipid Ab, protein C/S, factor V Leiden, homocysteine
- Autoimmune screen: ANA, ANCA
Complications
- Macular oedema — commonest cause of chronic poor vision
- Rubeosis iridis (NVI) → Neovascular glaucoma — "100-day glaucoma"
- Vitreous haemorrhage (from retinal NV)
- Macular ischaemia → permanent VA loss
- Epiretinal membrane, subretinal fibrosis (rare)
Management
Systemic
Control hypertension, diabetes, hyperlipidaemia. Investigate for underlying cause.
Treatment of Macular Oedema
Indicated when VA <6/9 and/or central macular thickness >250 μm on OCT:
| Treatment | Key Trial | Outcome |
|---|---|---|
| Intravitreal anti-VEGF (ranibizumab, aflibercept, bevacizumab) | CRUISE (ranibizumab), COPERNICUS (aflibercept) | First-line; monthly injections × 6 months improve VA vs placebo |
| Intravitreal dexamethasone implant (Ozurdex) | GENEVA study | Effective; risk of IOP rise and cataract |
| Intravitreal triamcinolone | SCORE study | >3 lines improvement in 25% vs 7% controls; 1 mg dose preferred |
| Laser photocoagulation | — | Not beneficial for visual outcome in CRVO |
Treatment of Neovascularization
- PRP (panretinal photocoagulation): 1500–2000 burns when NVI or angle NV develops; do NOT perform before NVI
- Adjunctive intravitreal anti-VEGF: every 6 weeks until stable; reduces macular oedema concurrently
- Neovascular glaucoma: managed as per NVG protocol
Key principle: No proven treatment can "unblock" the occluded vein. Treatment aims to control complications.
Prognosis
Non-ischaemic CRVO:
- VA 6/18 or better → likely to remain so
- VA 6/24–6/60 → variable, difficult to predict
- VA worse than 6/60 → usually poor
Ischaemic CRVO: Extremely poor — due to macular ischaemia and neovascular glaucoma.
Review schedule:
- Ischaemic: monthly for 6 months, then 3-monthly for 2–3 years
- Non-ischaemic: 3 months initially, discharge at 18–24 months if stable
Key Points for Exam (Khurana-style)
- Site of occlusion: at or posterior to lamina cribrosa
- Most common age: >65 years
- Most important systemic risk factor: hypertension
- Most important ocular risk factor: open-angle glaucoma
- Classic fundus: "blood and thunder" appearance (all 4 quadrants)
- NVI develops at 2–4 months → "100-day glaucoma"
- FA criterion for ischaemia: >10 disc areas of capillary non-perfusion
- RAPD present in: ischaemic CRVO
- First-line treatment of macular oedema: intravitreal anti-VEGF
- Key trials: CRUISE (ranibizumab), COPERNICUS (aflibercept), SCORE (triamcinolone), GENEVA (dexamethasone implant)
- Young females: think OCP, thrombophilia
— Kanski's Clinical Ophthalmology, 10th Ed., Chapter 13 (Retinal Vascular Disease)
This is a shared conversation. Sign in to Orris to start your own chat.