75 yr old male with op poisoning
Reading File
Finding Sources
Finding Sources
Searching PubMed
organophosphate poisoning management elderly
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Here is a comprehensive clinical overview of organophosphate (OP) poisoning in a 75-year-old male.
Organophosphate (OP) Poisoning — 75-Year-Old Male
Mechanism
Organophosphates irreversibly inhibit acetylcholinesterase (AChE), causing accumulation of acetylcholine (ACh) at all cholinergic synapses — muscarinic (parasympathetic), nicotinic (NMJ + autonomic ganglia), and CNS. This produces a cholinergic crisis.
"Aging" of the OP–enzyme complex occurs within hours, after which the inhibition becomes permanent and oximes can no longer reactivate AChE.
Clinical Features
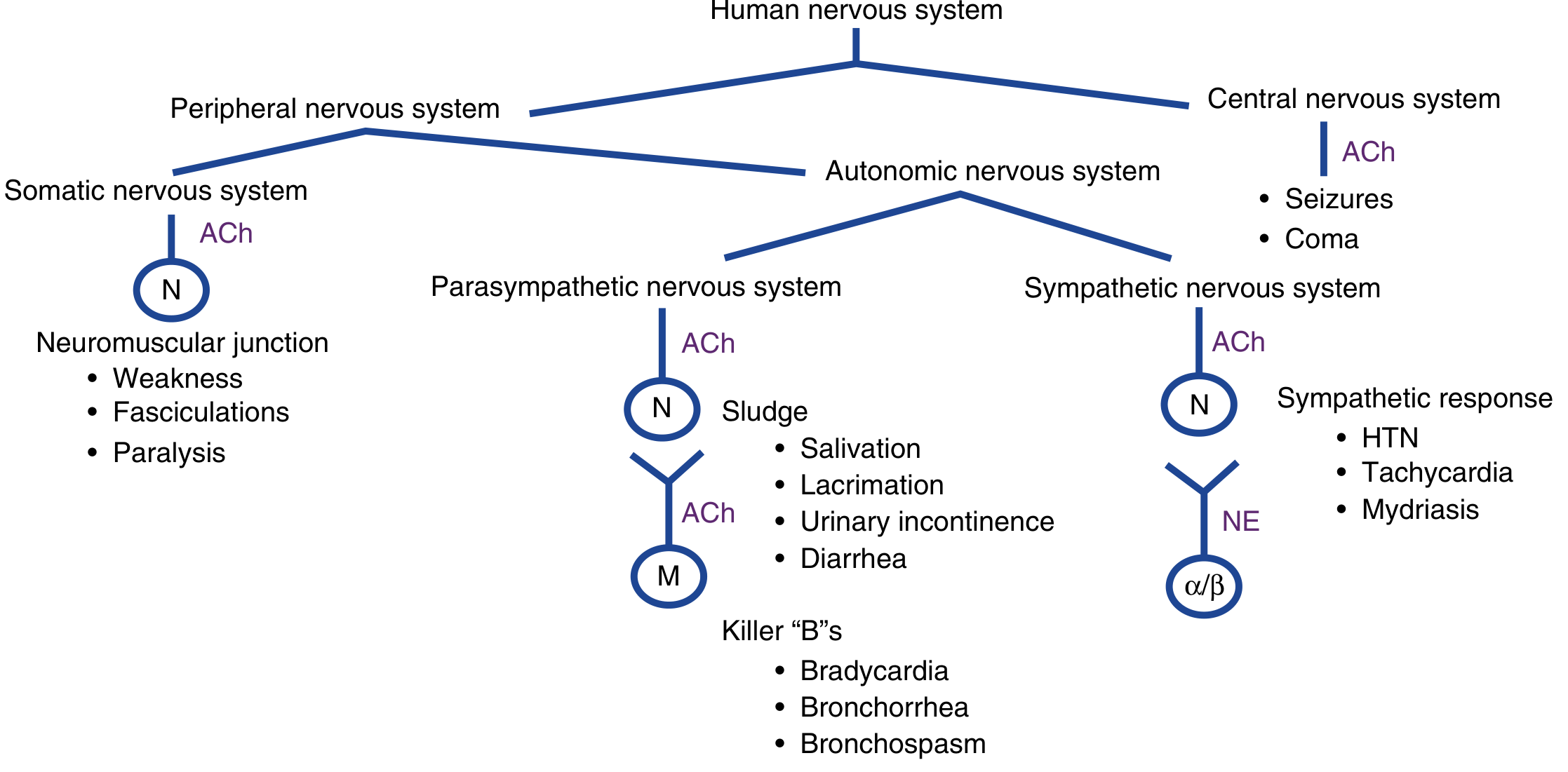
Muscarinic Effects (SLUDGE + Killer B's)
| Mnemonic | Features |
|---|---|
| SLUDGE | Salivation, Lacrimation, Urination, Defecation, GI cramps, Emesis |
| Killer B's | Bradycardia, Bronchorrhea, Bronchospasm |
| Other | Miosis, diaphoresis, urinary incontinence |
Nicotinic Effects (NMJ + Sympathetic ganglia)
- Muscle fasciculations → weakness → paralysis
- HTN, tachycardia (sympathetic ganglia stimulation)
CNS Effects
- Anxiety, confusion, altered mental status, seizures, coma
In a 75-year-old, altered mental status may be the dominant presentation. Respiratory failure from bronchospasm + bronchorrhea + neuromuscular paralysis is the primary cause of death. The elderly are at higher risk for aspiration, respiratory muscle fatigue, and prolonged mechanical ventilation needs.
Three Clinical Phases
| Phase | Timing | Features |
|---|---|---|
| Acute cholinergic crisis | Hours | SLUDGE, Killer B's, seizures, coma |
| Intermediate syndrome | 24–96 h after acute phase | Proximal limb weakness, neck flexor weakness, cranial nerve palsy, respiratory paralysis — does NOT respond to atropine |
| Delayed polyneuropathy | 2–5 weeks | Distal sensorimotor neuropathy, motor > sensory |
Investigations
- Plasma butyrylcholinesterase (easier to assay, decreases first) — ↓ ≥50% even in asymptomatic patients
- Red cell acetylcholinesterase (more specific): ↓ to 10–20% of normal in moderate, <10% in severe poisoning; takes up to 120 days to normalize
- ECG: QTc prolongation, ST changes, peaked T waves, AV block, torsades de pointes, VF
- CXR: pulmonary edema in severe cases
- Routine labs: pancreatitis, hypo/hyperglycemia, elevated LFTs, leukocytosis
Note: Cholinesterase levels have poor standardization across labs; clinical assessment drives management, not the number alone.
Management
1. Decontamination (FIRST priority)
- Remove ALL clothing (double-bag as hazardous waste)
- Copious water/soap skin flush — protects healthcare workers too
- PPE mandatory (level C: full-face air-purifying respirator, chemical-resistant suit, nitrile gloves)
- Gastric lavage and activated charcoal have no proven benefit (rapid absorption + early vomiting/diarrhea)
2. Airway & Supportive Care
- Suction secretions; supplemental O₂ (100%)
- Early intubation — use rocuronium 1 mg/kg (preferred; non-depolarizing, not metabolized by cholinesterases)
- Avoid succinylcholine if possible — prolonged paralysis (4–6 h) due to cholinesterase inhibition; if used, anticipate prolonged ventilation
- Benzodiazepines for seizures/agitation (after airway secured)
- Avoid β-blockers (tachyarrhythmias usually resolve with antidotes)
3. Antidotes
🅐 Atropine — Targets MUSCARINIC effects only
| Parameter | Detail |
|---|---|
| Initial dose | 1.2–3 mg IV bolus (severity-dependent) |
| Titration | Double dose every 5 min until adequate atropinization |
| Endpoint | ✔ Clear chest (dry secretions) ✔ HR >80 bpm ✔ SBP >80 mmHg |
| Maintenance | Continuous infusion at 10–20% of total loading dose per hour |
| Elderly note | High doses are still needed — do not underdose; miosis is NOT an atropinization endpoint |
| Toxicity to avoid | Absent bowel sounds, hyperthermia, delirium |
Do NOT use pupil dilation or heart rate alone as the atropinization endpoint — bronchial secretion drying is the key target.
🅑 Pralidoxime (2-PAM) — Reactivates AChE (before aging)
| Parameter | Detail |
|---|---|
| Dose | 1–2 g IV bolus over 15–30 min; then infusion 200–400 mg/h |
| Timing | Give as early as possible — becomes ineffective after AChE "aging" |
| Effect | Reverses nicotinic (NMJ) effects; complements atropine |
| Caveat | Rosen's recommends use only in severe poisoning or when unable to distinguish from carbamate poisoning |
🅒 Benzodiazepines (Diazepam/Lorazepam)
- For seizures, anxiety, agitation
- First-line for seizure control — atropine does NOT control seizures
Special Considerations in the Elderly (75 yr)
| Concern | Relevance |
|---|---|
| Reduced cholinesterase reserve | More severe cholinergic toxicity at lower doses |
| Polypharmacy | Drugs that lower plasma cholinesterase (codeine, morphine) worsen toxicity |
| Hepatic dysfunction | Reduces pralidoxime metabolism; impairs drug clearance |
| Respiratory reserve | Pre-existing COPD/restrictive disease worsens respiratory failure |
| CNS vulnerability | Delirium/coma may dominate; atropine-induced delirium easily confused with CNS poisoning |
| Cardiovascular comorbidity | QTc prolongation → higher risk of torsades and ventricular arrhythmias |
| Aspiration risk | Excessive secretions + altered consciousness → high aspiration pneumonia risk |
Disposition
- All symptomatic patients require ICU admission
- Monitor for intermediate syndrome (days 1–4) — respiratory paralysis can be fatal even after apparent recovery
- Watch for delayed polyneuropathy at 2–5 weeks follow-up
Sources:
- Rosen's Emergency Medicine, 9e, Ch. 152
- Tintinalli's Emergency Medicine, 9e, Ch. 201
- Adams and Victor's Principles of Neurology, 12e
- Bradley and Daroff's Neurology in Clinical Practice
- Lippincott Illustrated Reviews: Pharmacology
This is a shared conversation. Sign in to Orris to start your own chat.