Cerebral circulation and factors which govern the blood supply of brain in conscious and anaesthetised patients.
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
Loading Image
I now have comprehensive material from multiple authoritative sources. Let me compose the complete, well-structured answer.
Cerebral Circulation and Factors Governing Brain Blood Supply
1. Anatomy of the Cerebral Circulation
Arterial Supply
The brain receives blood from four arteries: two internal carotid arteries (anterior circulation) and two vertebral arteries (posterior circulation). The vertebral arteries unite to form the basilar artery. The internal carotids and basilar artery connect to form the circle of Willis — a vascular loop below the hypothalamus that permits collateral circulation between right–left and anterior–posterior systems.
From the circle arise three paired arteries perfusing the cortex:
- Anterior cerebral artery (ACA) — medial frontal and parietal lobes
- Middle cerebral artery (MCA) — lateral frontal, parietal, temporal lobes
- Posterior cerebral artery (PCA) — occipital lobe, inferior temporal
The anterior communicating artery connects the two ACAs; posterior communicating arteries link the ICAs to the PCAs, completing the loop. Under normal circumstances, pressures in the two systems are equal and blood from anterior and posterior circulations does not admix. In pathologic arterial occlusion, however, the circle acts as a shunt to increase collateral flow. Note: a complete circle of Willis is present in only ~50% of individuals — significant anatomical variation exists.
Venous Drainage
Three venous systems drain the brain:
- Superficial cortical veins — within the pia mater
- Deep cortical veins — drain deeper structures
- Both empty into dural sinuses (superior/inferior sagittal, straight, transverse, sigmoid) → internal jugular veins
In ~65% of patients, right IJV flow predominates; this has clinical relevance when placing jugular bulb catheters for SjvO₂ monitoring.
2. Normal Values
| Parameter | Value |
|---|---|
| Total CBF (adult) | 750 mL/min (~50 mL/100 g/min) |
| Gray matter CBF | ~80 mL/100 g/min |
| White matter CBF | ~20 mL/100 g/min |
| % of cardiac output | 12–15% |
| CMRO₂ | ~3.5 mL O₂/100 g/min |
| % of total body O₂ consumption | ~20% |
| EEG slowing threshold | <20–25 mL/100 g/min |
| Isoelectric EEG | <20 mL/100 g/min |
| Irreversible ischaemia | <10 mL/100 g/min |
Despite representing only 2% of body weight, the brain receives 12–15% of cardiac output because of its high metabolic rate. It has no energy stores — any interruption in CBF causes rapid functional impairment.
3. Factors Governing CBF
CBF is determined by cerebral perfusion pressure (CPP) and cerebrovascular resistance (CVR):
$$\text{CBF} = \frac{\text{CPP}}{\text{CVR}} = \frac{\text{MAP} - \text{ICP}}{\text{CVR}}$$
The factors regulating CVR — and hence CBF — include:
A. Myogenic Autoregulation (Pressure–Flow Autoregulation)
The cerebral circulation maintains constant CBF over a MAP of ~65–150 mmHg in normal conscious subjects (classical Lassen curve). Outside this range, CBF becomes pressure-passive.
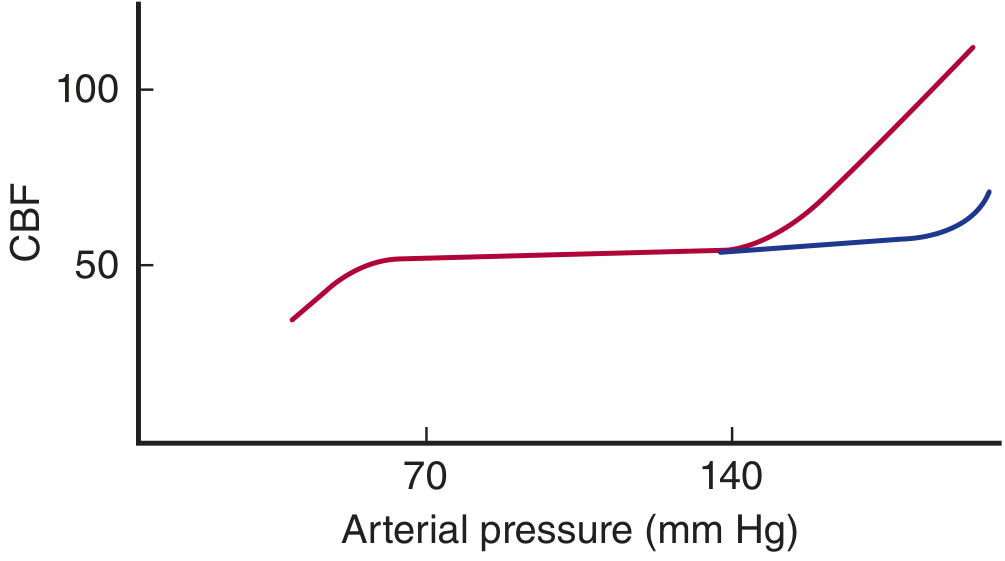
Mechanism: When arterial pressure rises, stretch-activated calcium influx into smooth muscle cells (via voltage-gated Ca²⁺ channels) causes vasoconstriction → increased CVR → maintained CBF. Nitric oxide (NO) from the endothelium, perivascular nerves, and astrocytic paracrine mediators modulate vascular tone.
Contemporary view (Miller's): The autoregulatory plateau is not truly flat but has a gentle positive slope. The plateau range is narrower (±10% MAP) than classically taught. Both the lower limit of autoregulation (LLA) and upper limit of autoregulation (ULA) show considerable inter-individual variability. Autoregulation is best viewed as a dynamic, not static process.
Chronic hypertension shifts the entire curve to the right — higher pressures are tolerated, but the LLA is also higher (these patients tolerate less hypotension).
B. Chemical and Metabolic Regulation
Carbon Dioxide (most potent cerebrovascular regulator)
CO₂ is the dominant vasomotor regulator of the cerebral circulation:
- Hypercapnia (↑PaCO₂): potent vasodilation → ↑CBF. CBF changes ~2–4% per mmHg change in PaCO₂ in the range 20–80 mmHg
- Hypocapnia (↓PaCO₂): vasoconstriction → ↓CBF. This is the basis of controlled hyperventilation to reduce ICP in neurosurgical patients
Mechanism: CO₂ diffuses freely across the blood–brain barrier; extracellular [H⁺] is the actual effector — perivascular acidosis causes vasodilation via smooth muscle relaxation.
Oxygen
- Hypoxia (PaO₂ < 50 mmHg): causes vasodilation → ↑CBF
- Normal PaO₂ has little effect on CBF
- Hyperoxia causes mild vasoconstriction
The diagram below summarises factors affecting overall CBF (Ganong's, Figure 33-8):
Metabolic/Flow–Metabolism Coupling (Neurovascular Coupling)
- Local CBF tightly matches local neuronal metabolic activity — the neurovascular unit (neurons, astrocytes, pericytes, endothelium)
- Increased neuronal firing → glutamate release → astrocyte activation → release of vasoactive mediators (K⁺, adenosine, NO, arachidonic acid metabolites) → local arteriolar dilation → regional CBF increase
- This is the physiologic basis of fMRI (BOLD signal)
- Gray matter CBF is 4× white matter CBF, matching metabolic demand
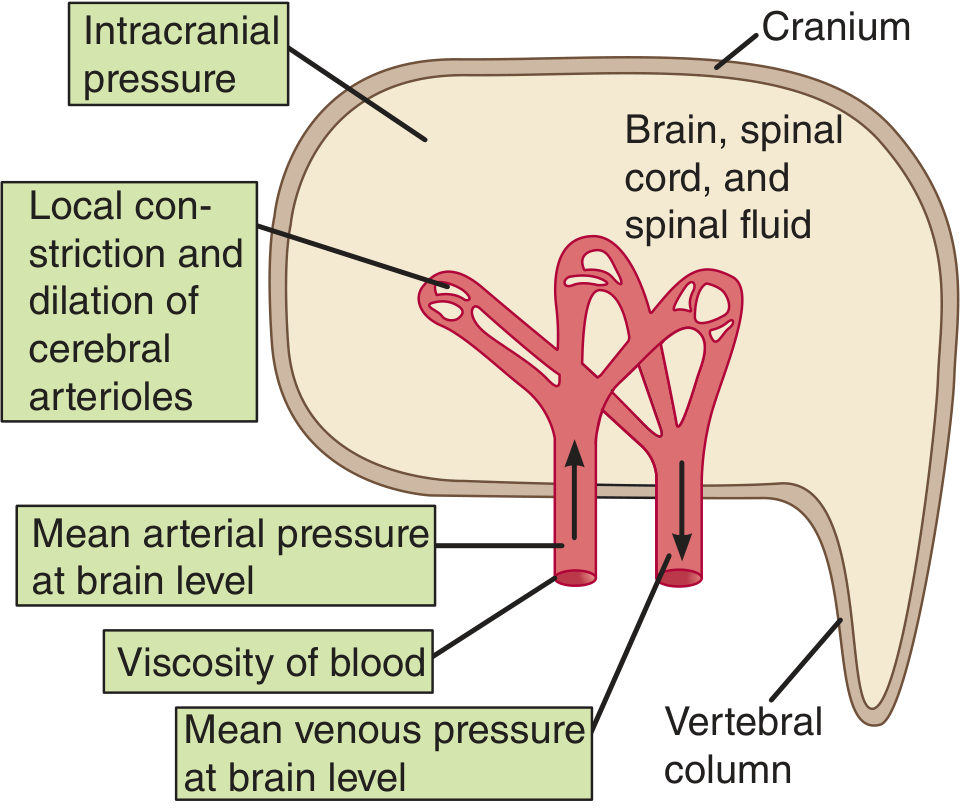
C. Intracranial Pressure (ICP) — Monro-Kellie Doctrine
The skull is a rigid compartment containing brain (~1400 g), blood (~75 mL), and CSF (~75 mL). Volume of any component can only increase at the expense of others:
$$\text{CPP} = \text{MAP} - \text{ICP}$$
A rise in ICP directly reduces CPP and therefore CBF. Rising venous pressure also raises ICP (which is why head-up positioning is used to lower ICP in neurosurgical patients). Normal ICP = 5–15 mmHg.
D. Neurogenic Regulation
- Large cerebral vessels receive extrinsic sympathetic (vasoconstriction via noradrenaline), parasympathetic (vasodilation via acetylcholine, VIP), and sensory (trigeminal — substance P, CGRP) innervation
- Intraparenchymal arterioles are primarily under intrinsic/metabolic control
- Sympathetic activation extends the plateau of autoregulation to the right (Fig. 33-9 blue line) — i.e., provides protection against hypertension-induced breakthrough
- Role of autonomic nerves in routine CBF regulation is modest; they become important during extreme BP changes
E. Blood Viscosity and Rheology
- Haematocrit is the main determinant of viscosity
- Increasing viscosity → ↑CVR → ↓CBF (and vice versa)
- Moderate haemodilution (Hct ~30–35%) may paradoxically improve CBF by reducing viscosity, though this must be balanced against reduced O₂-carrying capacity
F. Cardiac Output
Cardiac output influences CBF both via effects on MAP and via sympathetic nervous activity. Heart failure, reduced preload, or arrhythmias that reduce CO will compromise autoregulatory capacity, particularly at the lower limit.
4. Integrated Regulation Framework
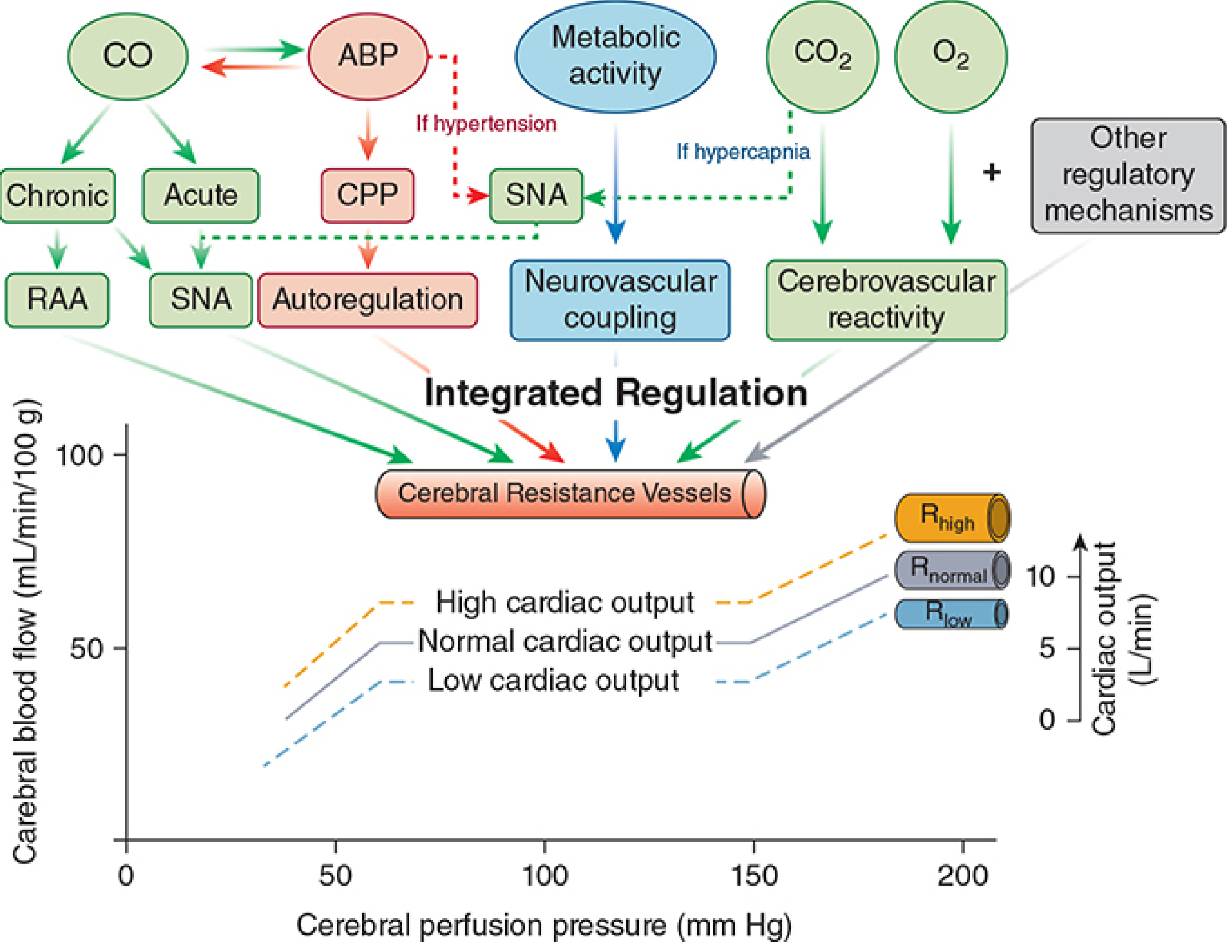
All regulatory mechanisms converge on cerebrovascular resistance vessels (pial arterioles account for ~50% of total CVR). The net CBF at any moment reflects the integrated balance of:
- Myogenic tone (MAP/CPP)
- Metabolic demand (neurovascular coupling)
- Blood gas tensions (PaCO₂ > PaO₂)
- Autonomic tone
- Cardiac output and circulating volume
5. CBF in the Conscious Patient
In the awake, resting individual:
- Total CBF ≈ 750 mL/min (54 mL/100 g/min)
- Flow is highest in the premotor and frontal cortex (cognitive/sensory decoding)
- Voluntary movement → increased regional CBF in corresponding motor/sensory cortex
- Language, visual tasks → increased occipital/temporal CBF
- These dynamic regional changes underlie PET and fMRI neuroimaging
- Autoregulation is intact; cerebrovascular reactivity to CO₂ is robust
- EEG and metabolic coupling are fully preserved
6. CBF Under General Anaesthesia
General anaesthesia produces complex, drug-specific alterations in CBF and CMRO₂. The key principle is:
CBF = f(CMRO₂, direct vascular effects, autoregulatory status)
Cerebral Metabolic Rate (CMRO₂) Under Anaesthesia
- ~60% of brain energy consumption is for electrophysiologic function (ion gradients, neurotransmitter cycling)
- ~40% is for cellular homeostasis ("housekeeping")
- Most anaesthetics (barbiturates, propofol, isoflurane, sevoflurane, desflurane, etomidate) suppress the electrophysiologic component in a dose-dependent manner, tracked by progressive EEG suppression
- Once EEG is isoelectric, no further reduction in CMRO₂ occurs — the housekeeping component is irreducible by anaesthetics
- Exceptions: Ketamine and N₂O increase CMRO₂ and CBF
Effects of Individual Anaesthetic Agents
Volatile Anaesthetics (Halothane, Isoflurane, Sevoflurane, Desflurane)
- All are direct cerebral vasodilators → tend to ↑CBF
- Simultaneously ↓CMRO₂ via EEG suppression → coupled tendency to ↓CBF
- Net effect = balance of direct vasodilation vs. metabolic suppression (uncoupling):
- Halothane: greatest cerebral vasodilation per MAC — significant CBF increase even at low doses
- Isoflurane: CBF minimally changed at <1 MAC (metabolic suppression offsets vasodilation); increases at ≥1.5 MAC
- Sevoflurane and Desflurane: minimal change in CBF up to 1.5 MAC; sevoflurane best preserves autoregulation
- Autoregulation: All volatile agents impair pressure autoregulation in a dose-dependent manner. At high doses, CBF becomes pressure-passive. Sevoflurane best preserves dynamic autoregulation; desflurane and isoflurane impair it more
- CO₂ reactivity is partially preserved under volatile anaesthesia
- ICP: Volatile agents increase CBF (and therefore ICP); this can be attenuated by prior hyperventilation or barbiturate coadministration
Intravenous Agents
| Agent | CBF | CMRO₂ | Autoregulation | ICP |
|---|---|---|---|---|
| Propofol | ↓↓ | ↓↓ | Preserved | ↓ |
| Barbiturates (thiopental) | ↓↓ | ↓↓ | Preserved | ↓↓ |
| Etomidate | ↓ | ↓ | Preserved | ↓ |
| Ketamine | ↑↑ | ↑ | Impaired | ↑↑ |
| Benzodiazepines | ↓ | ↓ | Preserved | ↓ |
| Dexmedetomidine | ↓ modest | ↓ modest | Preserved | ↓ |
| N₂O | ↑ | ↑ | Variable | ↑ |
Propofol and barbiturates are preferred in neurosurgery/raised ICP due to coupled reductions in CBF and CMRO₂ while preserving autoregulation.
Key principle: When complete EEG suppression is achieved, CMRO₂ is similar regardless of the agent used. However, the regional pattern differs — barbiturates produce uniform depression; isoflurane/sevoflurane produce preferential neocortical suppression.
Temperature and Anaesthesia
- CMRO₂ decreases 6–7% per °C of hypothermia
- Unlike anaesthetic drugs, hypothermia reduces both electrophysiologic and housekeeping components
- At 18–20°C, EEG is isoelectric; CMRO₂ at 18°C is <10% of normothermic values — basis of hypothermic circulatory arrest in cardiac surgery
Glymphatic System and Anaesthesia
The brain's glymphatic waste clearance system (perivascular aquaporin-mediated CSF flow) is enhanced during general anaesthesia — the periarterial space expands, increasing waste clearance. This is less active during volatile anaesthesia; dexmedetomidine better preserves glymphatic function.
Summary: Key Differences — Conscious vs. Anaesthetised
| Feature | Conscious | Anaesthetised |
|---|---|---|
| CBF | 50 mL/100g/min | Variable (drug-dependent) |
| CMRO₂ | 3.5 mL O₂/100g/min | Reduced (except ketamine/N₂O) |
| Flow–metabolism coupling | Intact | May be uncoupled by volatile agents |
| Autoregulation | Intact (MAP 65–150) | Impaired by volatiles (dose-dependent); preserved with IVA |
| CO₂ reactivity | Robust | Partially preserved (all agents) |
| Regional flow variation | High (task-dependent) | Blunted |
| ICP | Normal | ↑ with volatiles; ↓ with IVA |
Sources:
- Miller's Anesthesia, 10th Ed., Ch. 10 — Cerebral Physiology and the Effects of Anesthetic Drugs
- Morgan & Mikhail's Clinical Anesthesiology, 7th Ed., Ch. 26
- Barash, Cullen & Stoelting's Clinical Anesthesia, 9th Ed., Ch. 18
- Ganong's Review of Medical Physiology, 26th Ed., Ch. 33 — Cerebral Circulation
- Guyton & Hall Textbook of Medical Physiology
Add just a diagram of the anatomy as well and make a pdf
Reading File
Loading Image
Running Command
Running Command
Writing File
Running Command
Editing File
Running Command
The PDF was built successfully. Here's your download link:
Cerebral Circulation
PDF Document · PDF
The PDF contains:
- Cover page — titled with source credits
- Section 1 — Anatomy — Circle of Willis, arterial branches, venous drainage, normal values table, plus the full Fig. 1 anatomy diagram from Miller's (showing circle of Willis variants + cross-sections of vessel wall from MCA to capillary)
- Section 2 — Factors governing CBF — Ganong's factors diagram, autoregulation curve, integrated regulation diagram, with subsections covering: myogenic autoregulation, CO₂/O₂/metabolic coupling, ICP (Monro-Kellie), neurogenic, viscosity, and cardiac output
- Section 3 — Conscious patient — regional flow, intact coupling, measurement methods
- Section 4 — Anaesthetised patient — CMRO₂ principles, volatile agent table, IV agent table, temperature effects, glymphatic system
- Section 5 — Comparison table — Conscious vs. IVA vs. Volatile anaesthesia across 8 parameters
This is a shared conversation. Sign in to Orris to start your own chat.