Explain the congenital heart diseases in pediatrics mbbs
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Searching Images
tetralogy of Fallot congenital heart disease diagram
Loading Image
I now have comprehensive content from multiple authoritative textbooks and excellent diagrams. Let me compile the complete MBBS-level answer.
Congenital Heart Diseases (CHD) in Pediatrics
Definition & Epidemiology
Congenital heart diseases are abnormalities of the heart or great vessels present at birth. They account for 20–30% of all birth defects and affect nearly 1% of all live births (~40,000 infants/year in the USA). Incidence is higher in premature infants and stillborns (about one-fourth of stillborns have significant cardiac malformations). Twelve entities account for 85% of all CHD.
Frequency of Congenital Cardiac Malformations
| Malformation | Incidence per 1M Live Births | % |
|---|---|---|
| Ventricular septal defect (VSD) | 4,482 | 42 |
| Atrial septal defect (ASD) | 1,043 | 10 |
| Pulmonary stenosis | 836 | 8 |
| Patent ductus arteriosus (PDA) | 781 | 7 |
| Tetralogy of Fallot | 577 | 5 |
| Coarctation of aorta | 492 | 5 |
| Atrioventricular septal defect | 396 | 4 |
| Aortic stenosis | 388 | 4 |
| Transposition of great arteries | 388 | 4 |
| Truncus arteriosus | 136 | 1 |
| TAPVC | 120 | 1 |
| Tricuspid atresia | 118 | 1 |
(Source: Robbins & Kumar Basic Pathology)
Pathogenesis / Risk Factors
Faulty embryogenesis during gestational weeks 3–8 (when major cardiac structures form). Cause is unknown in ~90% of cases. Identified risk factors include:
- Prematurity, family history
- Maternal conditions: diabetes, hypertension, obesity, PKU, thyroid disorders, connective tissue disorders
- Teratogenic drugs: phenytoin, retinoic acid; smoking; alcohol
- Genetic disorders: Trisomy 21 (Down's), Trisomy 18, Trisomy 13, Turner syndrome, Noonan syndrome
- In utero infections: rubella (most classic), CMV, coxsackievirus, parvovirus B19, toxoplasmosis
- ~10% of defects are associated with genetic syndromes (VACTERL association, DiGeorge syndrome)
Classification
CHD is classified by:
- Haemodynamic/clinical: Cyanotic vs. Acyanotic
- Anatomic defect: Shunt, obstruction, transposition, or complex
Classification at a Glance
| Acyanotic (L→R Shunt initially) | Acyanotic (Obstruction) | Cyanotic (R→L Shunt) |
|---|---|---|
| VSD | Pulmonary stenosis | Tetralogy of Fallot |
| ASD | Aortic stenosis | Transposition of great arteries (TGA) |
| PDA | Coarctation of aorta | Tricuspid atresia |
| AVSD | Truncus arteriosus | |
| Total anomalous pulmonary venous connection (TAPVC) | ||
| Ebstein's anomaly |
Cyanotic lesions = the "Five Ts": Tetralogy of Fallot, Tricuspid anomalies (atresia, Ebstein's), Truncus arteriosus, Total anomalous pulmonary venous return, Transposition of great arteries.
Physiology of Shunts
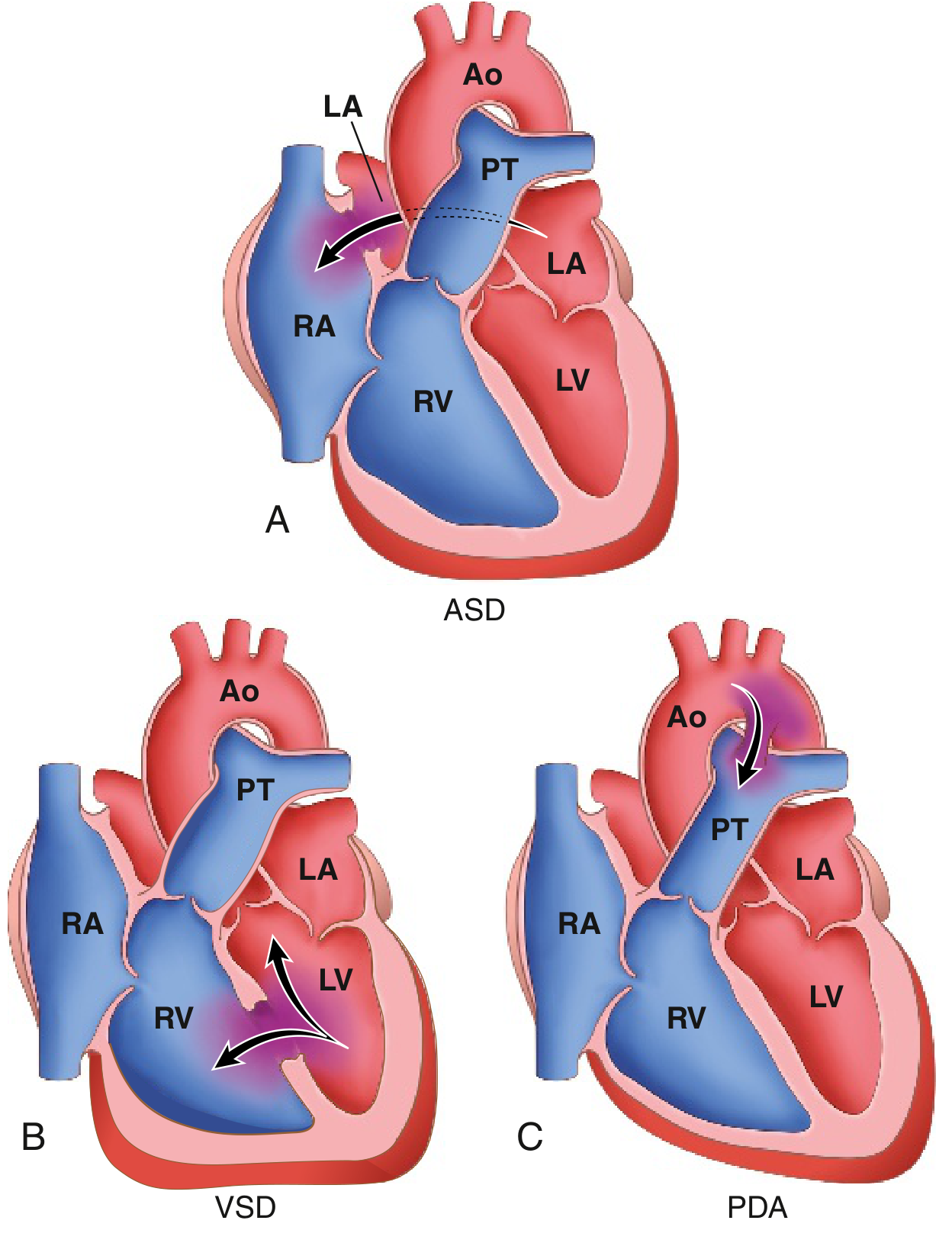
- Left → Right shunt: No early cyanosis. Increases pulmonary blood flow → RV hypertrophy → if untreated, leads to pulmonary hypertension → shunt reversal (R→L) = Eisenmenger syndrome with late cyanosis.
- Right → Left shunt: Immediate cyanosis (poorly oxygenated blood enters systemic circulation). Associated with polycythemia, clubbing, hypoxic spells.
ACYANOTIC HEART DISEASES
1. Ventricular Septal Defect (VSD)
Most common CHD at birth (42%).
- Defect: Opening in the interventricular septum; 90% are in the membranous (perimembranous/subarterial) region
- Physiology: L→R shunt → pulmonary overcirculation → if large, LV failure
- Clinical features:
- Small VSD: Loud, harsh pansystolic murmur at left lower sternal border (LLSB); often asymptomatic; "Maladie de Roger"
- Large VSD: Poor feeding, failure to thrive, recurrent respiratory infections, tachycardia, hepatomegaly, CHF
- Investigations: CXR — cardiomegaly, increased pulmonary vascular markings; Echo is diagnostic
- Management: Most small VSDs close spontaneously (by 2 years). Large VSDs require surgical patch closure or catheter-based device closure. Diuretics (furosemide) + digoxin for CHF
2. Atrial Septal Defect (ASD)
Most common CHD diagnosed in adulthood (spontaneous closure less likely than VSD).
Types:
-
Ostium secundum (90%): Near foramen ovale; isolated defect
-
Ostium primum (5%): Lower atrial septum; associated with mitral/tricuspid valve anomalies; part of AVSD spectrum
-
Sinus venosus (5%): High in atrial septum; associated with anomalous pulmonary venous drainage
-
Clinical features: Usually asymptomatic in childhood. Fixed splitting of S2 (hallmark), systolic ejection murmur at left upper sternal border (pulmonary flow murmur), mid-diastolic murmur at tricuspid area (flow)
-
Complications: Atrial arrhythmias, right heart failure, pulmonary hypertension, paradoxical embolism
-
Management: Device closure (catheter-based, for secundum) or surgical closure. Patent foramen ovale (PFO) ≠ true ASD — usually closes spontaneously; can cause paradoxical embolism
3. Patent Ductus Arteriosus (PDA)
- Defect: Persistence of the ductus arteriosus (fetal communication between aorta and pulmonary artery) after birth; normally closes within 15 hours–3 days of life
- At risk: Premature infants, maternal rubella, high altitude births
- Physiology: L→R shunt (Ao → PA); wide pulse pressure; bounding pulses
- Clinical features:
- Continuous "machinery" murmur at left infraclavicular area (Gibson murmur), best heard at left 2nd intercostal space
- Widened pulse pressure, collapsing/bounding pulses (Corrigan's pulse)
- Signs of CHF in large PDA
- Investigations: CXR — cardiomegaly, increased pulmonary markings; Echo is definitive
- Management:
- Premature infants: Indomethacin or Ibuprofen (COX inhibitors to close) IV
- Term infants/children: Surgical ligation or catheter-based device (coil/Amplatzer)
- Prostaglandin E1 (PGE1) is used to keep PDA open in duct-dependent lesions (e.g., pulmonary atresia, critical coarctation)
4. Coarctation of the Aorta
- Defect: Discrete narrowing of the aorta, usually near the ductus arteriosus insertion (juxtaductal, most common); can be preductal (infantile) or postductal (adult type)
- Associated: Bicuspid aortic valve (50–85%), Turner syndrome (classic association)
- Clinical features:
- Upper limb hypertension, weak/absent femoral pulses
- BP differential: Upper extremities > lower extremities (>20 mmHg)
- Systolic murmur (left infraclavicular/interscapular)
- In neonates: Can present with cardiovascular collapse when PDA closes
- Rib notching on CXR (in older children >6 yrs, from intercostal collaterals) — "3-sign" on CXR
- Management: Balloon angioplasty + stenting (catheter-based) or surgical resection. Recurrence in 10% of cases.
5. Pulmonary Stenosis (PS)
- Defect: Narrowing at pulmonary valve (most common), subvalvular (infundibular), or supravalvular level
- Associated: Noonan syndrome (classic), Williams syndrome (supravalvular), congenital rubella
- Clinical features:
- Systolic ejection murmur at left upper sternal border with ejection click
- Widely split S2 (delayed P2)
- RV heave; cyanosis only if severe (critical PS with R→L shunt through PFO/ASD)
- Management: Balloon pulmonary valvuloplasty (catheter-based); surgery for subvalvular/supravalvular
6. Aortic Stenosis (AS)
- Defect: Obstruction at valvular (most common — bicuspid aortic valve), subvalvular, or supravalvular level
- Clinical: Systolic ejection murmur radiating to neck; exertional syncope, angina, dyspnea (classical triad in older children)
- Critical AS in neonates: Duct-dependent systemic circulation; presents with severe CHF/shock
- Management: Balloon valvuloplasty or surgical valve repair/replacement
CYANOTIC HEART DISEASES
1. Tetralogy of Fallot (TOF) — Most common cyanotic CHD
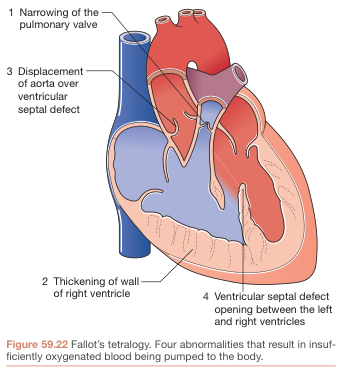
Four components ("PROVE"):
- Pulmonary stenosis (infundibular/valvular)
- Right ventricular hypertrophy (RVH)
- Overriding aorta (dextroposition — sits over VSD)
- Ventricular septal defect (large, perimembranous)
The primary defect is anterior malalignment of the infundibular septum → causes all four components.
- Clinical features:
- Cyanosis (worsening with age and after feeding/exertion)
- Hypercyanotic ("Tet") spells: Paroxysmal hyperpnea, increasing cyanosis, squatting behaviour (squatting increases SVR → decreases R→L shunt); peak 2–4 months–2 years
- Clubbing of fingers and toes (chronic hypoxemia)
- Single S2 (pulmonary component absent), harsh systolic ejection murmur at LUSB (due to RVOTO, not VSD)
- Investigations:
- CXR: "Boot-shaped heart" (coeur en sabot) — upturned apex from RVH, concave pulmonary bay, right-sided aortic arch (25%)
- ECG: Right axis deviation, RVH
- Echo: Diagnostic
- Management:
- Acute Tet spell: Knee-chest position, 100% O2, IV morphine, propranolol, phenylephrine (↑ SVR), IV fluid bolus
- Definitive: Total surgical correction (patch VSD, relieve RVOTO); optimal at 3–6 months
- Palliative (before correction): Blalock-Taussig shunt (subclavian → pulmonary artery)
2. Transposition of the Great Arteries (TGA) — Most common cyanotic CHD presenting in neonates
- Defect: Aorta arises from RV; Pulmonary artery arises from LV → two parallel circulations (incompatible with life unless mixing exists via PFO/ASD/VSD/PDA)
- Clinical: Severe cyanosis from birth, no murmur (unless associated VSD/PS), tachypnea
- CXR: "Egg-on-side" heart (narrow mediastinum), increased pulmonary vascularity
- Management:
- Immediate: PGE1 (keep PDA open), balloon atrial septostomy (Rashkind procedure) to increase mixing
- Definitive: Arterial switch operation (Jatene) — reconnects aorta to LV and PA to RV; done in first 2 weeks of life
3. Tricuspid Atresia
- Defect: Complete absence of tricuspid valve → no direct communication between RA and RV; requires ASD (mandatory) + VSD or PDA for survival
- Clinical: Cyanosis from birth, single S2, hepatomegaly; LV hypertrophy on ECG (unusual for cyanotic CHD)
- Management: Palliative Blalock-Taussig shunt → Fontan procedure (connecting IVC/SVC directly to pulmonary artery)
4. Truncus Arteriosus
- Defect: Single arterial trunk arising from both ventricles (via large VSD), giving rise to systemic, pulmonary, and coronary circulations
- Clinical: Mild cyanosis, loud S2, harsh systolic murmur; early CHF from pulmonary overcirculation
- Association: DiGeorge syndrome (22q11 deletion)
- Management: Surgical repair in early infancy
5. Total Anomalous Pulmonary Venous Connection (TAPVC)
- Defect: All four pulmonary veins drain into right atrium (or systemic veins) instead of left atrium; ASD (or PFO) is mandatory for survival
- Types: Supracardiac (most common), cardiac, infracardiac, mixed
- Clinical: Depends on obstruction — obstructed type presents with severe cyanosis and pulmonary edema at birth; unobstructed type shows mild cyanosis
- CXR: "Snowman" (figure-of-8) sign in supracardiac type
- Management: Surgical re-routing of pulmonary veins to left atrium (emergency if obstructed)
6. Ebstein's Anomaly
- Defect: Downward displacement of abnormal tricuspid valve into RV → "atrialized" RV; associated with maternal lithium use
- Clinical: Variable; cyanosis (via ASD R→L), arrhythmias (WPW syndrome common — accessory pathway), S3/S4 gallop, systolic murmur
- ECG: Tall P waves, RBBB, delta waves (WPW); "Himalayan P waves"
- Management: Medical (arrhythmia control) or surgical valve repair/replacement
Eisenmenger Syndrome
When any large L→R shunt (VSD, ASD, PDA) is left uncorrected:
- Chronic high-pressure/volume pulmonary flow → irreversible pulmonary vascular disease → pulmonary hypertension → shunt reversal to R→L → late-onset cyanosis, clubbing, erythrocytosis
- Surgical correction is contraindicated once established (fixed pulmonary hypertension)
- Management: Pulmonary vasodilators (sildenafil, bosentan), heart-lung transplant
Clinical Approach to CHD in MBBS
Hyperoxia Test (Nitrogen Washout Test)
Administer 100% O2 for 10–20 minutes:
- PaO2 rises >150 mmHg → Pulmonary cause (normal response)
- PaO2 does NOT rise (remains <100 mmHg) → Cyanotic CHD (R→L shunt)
- Blood turns pink in all causes except methemoglobinemia (remains chocolate-colored)
Key Murmur Summary
| Lesion | Murmur |
|---|---|
| VSD | Pansystolic (holosystolic), LLSB |
| ASD | Ejection systolic, LUSB; fixed split S2 |
| PDA | Continuous "machinery" murmur, left infraclavicular |
| PS | Ejection systolic + click, LUSB; wide split S2 |
| TOF | Ejection systolic, LUSB (RVOTO); single S2 |
| AS | Ejection systolic, aortic area, radiates to neck |
| Coarctation | Systolic, interscapular/left infraclavicular |
Key CXR Signs
| Sign | Lesion |
|---|---|
| Boot-shaped heart (coeur en sabot) | TOF |
| Egg-on-side / narrow mediastinum | TGA |
| Snowman / figure-of-8 | TAPVC (supracardiac) |
| Rib notching, 3-sign | Coarctation of aorta |
| Cardiomegaly + increased PVM | VSD, ASD, PDA, TAPVC (unobstructed) |
Surgical Procedures Summary
| Procedure | Indication |
|---|---|
| Blalock-Taussig (BT) shunt | Palliation: TOF, tricuspid atresia, pulmonary atresia |
| Arterial switch (Jatene) | TGA (within 2 weeks of birth) |
| Rashkind balloon septostomy | TGA (emergency mixing) |
| Fontan procedure | Single-ventricle palliation (tricuspid atresia) |
| Mustard/Senning (atrial baffle) | Older TGA repair (historical) |
| VSD/ASD patch closure | VSD, ASD |
| PDA ligation/coiling | PDA |
| Balloon pulmonary valvuloplasty | PS |
| Norwood procedure | Hypoplastic left heart syndrome (HLHS) |
Sources:
- Robbins & Kumar Basic Pathology, p. 347–358
- Tintinalli's Emergency Medicine: A Comprehensive Study Guide, p. 861–870
- Bailey & Love's Short Practice of Surgery, p. 986
This is a shared conversation. Sign in to Orris to start your own chat.