Retry
Checking Date & Time
It looks like your message just says "Retry" — could you clarify what you'd like me to retry or help you with?
Drug interactions comprehensive review
Asking for Preferences
Checking Date & Time
Reading File
Finding Sources
Searching PubMed
"drug interactions" AND "clinical significance"
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Loading Image
Finding Sources
Reading File
I now have comprehensive source material. Let me compile the full review.
Drug Interactions: A Comprehensive Clinical Review
Overview
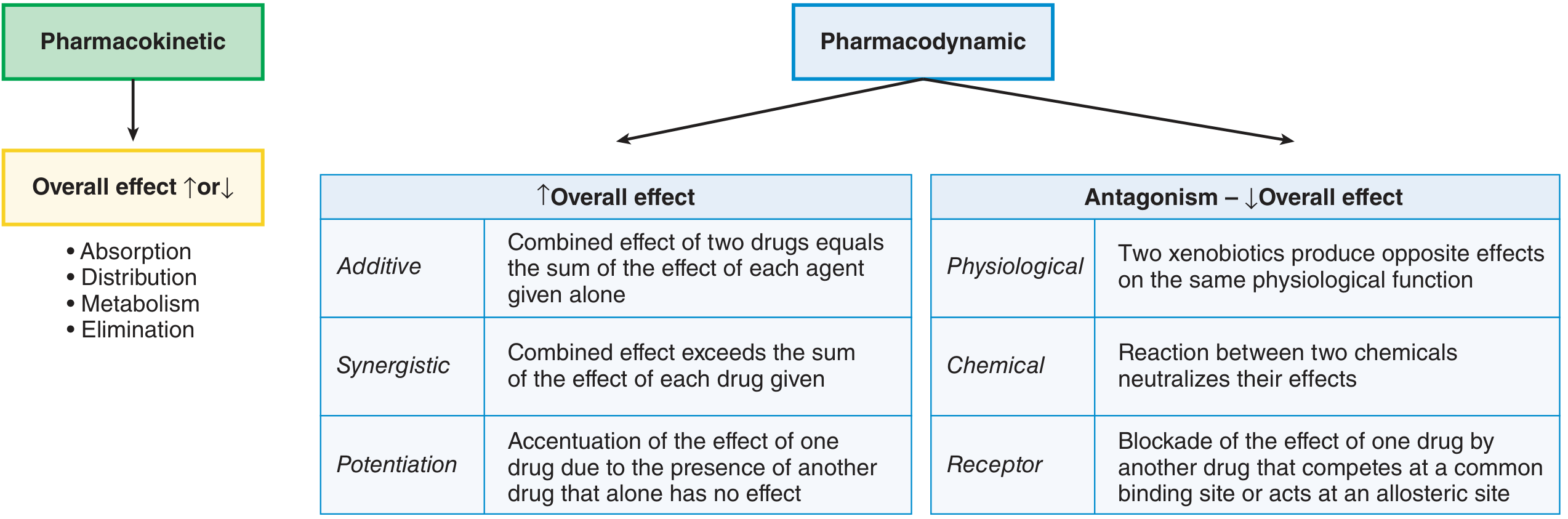
A drug interaction occurs when the activity or toxicity of one drug is altered by the concurrent use of another drug, food, or supplement. Drug interactions are a major source of preventable harm — virtually all hospitalized patients on antimicrobials receive concurrent medications with interaction potential. Two primary mechanisms drive clinically relevant interactions: pharmacokinetic (PK) and pharmacodynamic (PD).
Part 1: Pharmacokinetic Drug Interactions
Pharmacokinetic interactions occur when one drug alters the absorption, distribution, metabolism, or elimination (ADME) of another, resulting in increased or decreased drug effect.
1.1 Absorption Interactions
Interactions at the absorption step alter how much drug enters systemic circulation:
| Mechanism | Example | Clinical Effect |
|---|---|---|
| GI pH alteration | Ranitidine + triazolam | Raised GI pH → ↑ absorption of basic drugs |
| Physical adsorption | Levothyroxine + calcium antacids | Calcium binds levothyroxine → ↓ absorption |
| Chelation | Fluoroquinolones + divalent cations (Fe²⁺, Mg²⁺, Al³⁺) | Chelate formation → ↓ antibiotic absorption |
| GI motility | Opioids slow gastric emptying | Alters absorption rate of co-administered drugs |
1.2 Transporter-Mediated Interactions
Drug transporters are membrane proteins that regulate intracellular drug concentrations at the gut, liver, kidney, and blood-brain barrier. Clinically important transporters include:
- P-glycoprotein (P-gp / MDR1) — efflux transporter; highly expressed in gut epithelium, BBB, liver, and kidney
- Organic anion transporters (OAT1, OAT3) — renal secretion of anionic drugs
- Breast cancer resistance protein (BCRP/ABCG2) — efflux in gut and liver
- Organic cation transporters (OCT1, OCT2) — hepatic and renal uptake
Key clinical examples:
| Perpetrator | Victim | Transporter | Effect |
|---|---|---|---|
| Verapamil (P-gp inhibitor) | Dabigatran (P-gp substrate) | P-gp | ↑ dabigatran AUC by 71%, ↑ Cmax by 91% → bleeding risk |
| P-gp inhibitors (e.g., quinidine) | Loperamide | P-gp | Loperamide crosses BBB → CNS opioid toxicity, cardiac arrhythmia |
| Probenecid | Penicillin, methotrexate | OAT | Blocks renal tubular secretion → ↑ drug levels |
| Cyclosporine | Statins (rosuvastatin) | OATP1B1 | ↑ statin exposure → myopathy risk |
Clinical Pearl: Loperamide is normally a safe antidiarrheal because P-gp efflux prevents CNS penetration. When combined with P-gp inhibitors, it becomes a CNS-active opioid with life-threatening cardiovascular toxicity.
1.3 Protein Binding Interactions
Many drugs are highly plasma protein–bound (albumin, α₁-acid glycoprotein). Displacement interactions can transiently alter free drug concentrations:
- Drugs prone to displacement: warfarin, phenytoin, valproic acid, sulfonamides, barbiturates, aspirin
- Clinical relevance is often overstated — displacement is usually transient as free drug redistributes and elimination increases
- Clinical concern is highest in patients with hypoalbuminemia, overdose (binding site saturation), or with drugs of narrow therapeutic index
1.4 Metabolism Interactions: CYP Enzymes
The cytochrome P450 (CYP) enzyme system in the liver (and gut) is the most common site of clinically significant drug interactions. Key isoforms:
| CYP Isoform | Substrates (examples) | Inhibitors | Inducers |
|---|---|---|---|
| CYP3A4 (most abundant; ~50% of drugs) | Statins, protease inhibitors, CCBs, benzodiazepines, cyclosporine, fentanyl | Ketoconazole, itraconazole, erythromycin, clarithromycin, ritonavir, grapefruit juice | Rifampin, carbamazepine, phenytoin, St. John's wort, dexamethasone |
| CYP2D6 | Codeine, tramadol, TCAs, SSRIs, β-blockers, antipsychotics | Fluoxetine, paroxetine, bupropion, quinidine | None significant |
| CYP2C9 | Warfarin (S-), NSAIDs, phenytoin, glipizide | Fluconazole, amiodarone | Rifampin, carbamazepine |
| CYP2C19 | PPIs, clopidogrel (prodrug), diazepam | Omeprazole, fluoxetine | Rifampin |
| CYP1A2 | Theophylline, clozapine, olanzapine, caffeine | Ciprofloxacin, fluvoxamine | Smoking, omeprazole |
| CYP2E1 | Acetaminophen (toxic pathway), ethanol | Disulfiram | Ethanol (chronic), isoniazid |
Inhibition (rapid onset — occurs as soon as the inhibitor reaches steady state):
- Reduces metabolism of victim drug → elevated plasma levels → toxicity
- Example: Ketoconazole (CYP3A4 inhibitor) + HIV protease inhibitors → ↑ protease inhibitor levels
Induction (delayed onset — requires days to weeks for new enzyme synthesis):
- Increases metabolism of victim drug → reduced efficacy
- Critical examples:
- Rifampin (potent pan-inducer of CYP3A4, CYP2C9, transporters) → reduces efficacy of oral contraceptives, warfarin, HIV antiretrovirals, immunosuppressants
- St. John's wort → induces CYP3A4 → reduces levels of cyclosporine, antiretrovirals, digoxin
Grapefruit juice (furanocoumarins, naringin) irreversibly inhibits intestinal CYP3A4, increasing bioavailability of statins, CCBs, and immunosuppressants. The effect persists for up to 72 hours after a single glass.
The withdrawn drug terfenadine is a landmark case: as a CYP3A4 substrate, its metabolism was inhibited by erythromycin and grapefruit juice → elevated parent compound levels → fatal QT prolongation/torsades de pointes. Its active metabolite (fexofenadine), which is not cardiotoxic, replaced it.
Acetaminophen–Ethanol Interaction (CYP2E1)
- Acute co-ingestion: Ethanol competitively inhibits CYP2E1 → less NAPQI (toxic metabolite) formed → possibly protective
- Chronic alcohol use: Induces CYP2E1 + depletes glutathione → far more NAPQI generated at standard doses → hepatotoxicity
1.5 Elimination Interactions
Renal interactions are critical for drugs with narrow therapeutic indices excreted renally:
-
Lithium is entirely renally eliminated. Clearance depends on sodium balance. Drugs that reduce renal blood flow or tubular secretion increase lithium levels dramatically:
- NSAIDs (inhibit prostaglandins → ↓ renal perfusion) → 12–66% ↓ lithium clearance
- ACE inhibitors and ARBs → ↓ GFR
- Thiazide and loop diuretics → sodium depletion → compensatory lithium reabsorption
-
Methotrexate + NSAIDs — NSAIDs reduce renal OAT-mediated secretion → ↑ methotrexate toxicity (mucositis, bone marrow suppression)
-
Probenecid + penicillin — historically used therapeutically to prolong penicillin levels by blocking tubular secretion
Part 2: Pharmacodynamic Drug Interactions
Pharmacodynamic interactions occur at the level of receptor binding or physiological effect, independent of drug concentration changes. They are classified by direction of effect:
2.1 Agonistic (↑ Overall Effect)
| Type | Definition | Example |
|---|---|---|
| Additive | Combined effect = sum of individual effects | Aspirin + warfarin = additive bleeding risk |
| Synergistic | Combined effect > sum of individual effects | Trimethoprim + sulfamethoxazole (TMP-SMX) block sequential steps in folate synthesis; bactericidal synergy |
| Potentiation | One drug (with no relevant effect alone) amplifies another | Clavulanate (no antibiotic activity alone) + amoxicillin → β-lactamase inhibition greatly potentiates amoxicillin |
2.2 Antagonistic (↓ Overall Effect)
| Type | Definition | Example |
|---|---|---|
| Receptor antagonism | Drug B competes at or allosterically blocks Drug A's receptor | Naloxone reverses opioid toxicity; flumazenil reverses benzodiazepine sedation |
| Physiological antagonism | Two drugs produce opposing physiological effects | Insulin + glucocorticoids (opposing glycemic effects) |
| Chemical antagonism | Direct chemical neutralization | Protamine + heparin (ionic binding inactivates heparin) |
Part 3: Clinically Critical Interaction Categories
3.1 QT Prolongation and Torsades de Pointes
This is one of the most dangerous drug interaction patterns. QT-prolonging drugs have additive/synergistic effects on cardiac repolarization:
- High-risk drug classes: antiarrhythmics (Class IA: quinidine, procainamide; Class III: sotalol, amiodarone), fluoroquinolones, macrolides, antipsychotics (haloperidol, thioridazine), methadone, some antiemetics (ondansetron at high doses), azole antifungals
- Amplifiers: hypokalemia, hypomagnesemia, bradycardia, female sex, congenital long QT syndrome
- Key interaction: Ciprofloxacin or macrolides + antipsychotics → additive QT prolongation
3.2 Serotonin Syndrome
Occurs when serotonergic activity is excessive — typically from combining drugs that increase synaptic serotonin through different mechanisms:
- Mechanisms: ↑ serotonin synthesis (L-tryptophan), ↓ serotonin reuptake (SSRIs, SNRIs, TCAs, tramadol, meperidine), ↓ serotonin breakdown (MAOIs), direct serotonin agonists (triptans, LSD)
- Critical combination: Linezolid (reversible, non-selective MAO inhibitor) + SSRIs, opioid analgesics, or antiepileptics → serotonin syndrome
- Clinical features: agitation, tremor, myoclonus, hyperthermia, diaphoresis, tachycardia → life-threatening
3.3 Bleeding Risk Interactions
- Warfarin is a CYP2C9 substrate and is also affected pharmacodynamically:
- Amiodarone inhibits CYP2C9 → ↑ warfarin levels → ↑ INR
- Rifampin induces CYP2C9 → ↓ warfarin levels → ↓ INR
- Aspirin + warfarin: dual PD effect — antiplatelet effect (pharmacodynamic) + displacement from protein binding
- Direct oral anticoagulants (DOACs) are P-gp and CYP3A4 substrates (apixaban, rivaroxaban, edoxaban)
3.4 CNS Depression
- Opioids + benzodiazepines → synergistic respiratory depression (Black Box Warning)
- CNS depressants + antihistamines, alcohol, muscle relaxants → additive sedation
3.5 Antimicrobial-Specific Interactions
- Triazoles (voriconazole, itraconazole, posaconazole) are potent CYP3A4/2C9/2C19 inhibitors → toxic accumulation of co-administered drugs
- Ciprofloxacin inhibits CYP1A2 → elevates theophylline, clozapine, olanzapine levels
- Rifampin induces CYP3A4/P-gp → reduces oral contraceptive efficacy, warfarin effect, immunosuppressant levels
Part 4: Special Populations and Risk Factors
| Risk Factor | Why It Matters |
|---|---|
| Polypharmacy (≥5 drugs) | Exponentially increases interaction probability |
| Narrow therapeutic index drugs | Small changes in PK → toxicity or failure (warfarin, digoxin, lithium, phenytoin, theophylline, cyclosporine) |
| Elderly patients | ↓ hepatic CYP activity, ↓ renal function, ↓ albumin, ↑ drug sensitivity |
| Hepatic impairment | Reduced phase I and II metabolism; affects metronidazole, voriconazole, caspofungin |
| Renal impairment | Reduced drug/metabolite clearance; lithium, metformin, methotrexate, DOACs |
| Genetic polymorphisms | CYP2D6 ultra-rapid metabolizers: codeine → morphine toxicity; poor metabolizers: codeine → no effect |
| Herbal/OTC products | St. John's wort (CYP3A4 inducer), grapefruit juice (CYP3A4 inhibitor), fish oil (antiplatelet) |
Part 5: Prevention and Clinical Management
- Reconcile the medication list at every transition of care — admission, discharge, clinic visits
- Check interaction databases (Lexicomp, Micromedex, Epocrates) for all new prescriptions
- Know your narrow-TI drugs and treat them with extra scrutiny: warfarin, digoxin, lithium, phenytoin, theophylline, methotrexate, cyclosporine, DOACs
- Time separation can reduce absorption interactions (e.g., take levothyroxine 4 hours apart from calcium)
- Monitor and adjust — when interactions cannot be avoided, increase monitoring frequency (INR, drug levels, renal function)
- Consider CYP genotyping for high-stakes drugs in selected patients (clopidogrel + CYP2C19 loss-of-function alleles → reduced activation → stent thrombosis risk)
- Educate patients about foods and herbal products — grapefruit juice, St. John's wort, alcohol
Quick Reference: Highest-Risk Combinations in Clinical Practice
| Drug A | Drug B | Mechanism | Hazard |
|---|---|---|---|
| Warfarin | Amiodarone | CYP2C9 inhibition (PK) | ↑ bleeding |
| Warfarin | Rifampin | CYP2C9/CYP3A4 induction (PK) | ↓ anticoagulation |
| SSRI/SNRI | Linezolid or MAOI | Serotonin excess (PD) | Serotonin syndrome |
| Dabigatran | Verapamil | P-gp inhibition (PK) | ↑ bleeding |
| Statins | Clarithromycin/azole | CYP3A4 inhibition (PK) | Myopathy/rhabdomyolysis |
| Clopidogrel | PPIs (omeprazole) | CYP2C19 inhibition (PK) | ↓ antiplatelet efficacy |
| Lithium | NSAIDs/ACEi/diuretics | ↓ renal clearance (PK) | Lithium toxicity |
| Opioids | Benzodiazepines | CNS/respiratory depression (PD) | Respiratory arrest |
| QT-prolonging drugs | QT-prolonging drugs | Additive channel blockade (PD) | Torsades de pointes |
| Loperamide | P-gp inhibitors | P-gp inhibition (PK) | CNS opioid toxicity |
| Oral contraceptives | Rifampin/St. John's wort | CYP3A4 induction (PK) | Contraceptive failure |
| Methotrexate | NSAIDs | OAT inhibition (PK) | MTX toxicity |
Sources:
- Goodman & Gilman's The Pharmacological Basis of Therapeutics, Chapters 5 & 9
- Goldman-Cecil Medicine International Edition, Chapter 42 (Antimicrobial Drug-Drug Interactions)
This is a shared conversation. Sign in to Orris to start your own chat.