Toxocariasis. Fascioliasis.
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
Toxocara larvae visceral larva migrans histology eosinophilic granuloma
Searching Images
Fasciola hepatica liver fluke life cycle
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have all the information needed to compile a thorough response on both conditions.
Toxocariasis
Etiology & Epidemiology
Toxocariasis results from accidental ingestion of embryonated eggs of Toxocara canis (dog roundworm) or, less often, Toxocara cati (cat ascarid). It is a globally distributed zoonosis; a nationally representative US survey found 5% of the population ≥6 years old to be seropositive. Infection is most common among children who ingest soil contaminated with dog or cat feces in playgrounds and sandboxes. Eggs become infective 2–4 weeks after shedding and can persist long-term in soil. Direct contact with animals is not required.
- Visceral larva migrans (VLM): predominantly children aged 2–7 years
- Ocular larva migrans (OLM): older children/adolescents (5–10 years); more prevalent in southern US states
Pathobiology
In the natural animal host, Toxocara larvae follow the usual ascarid life cycle (intestine → portal blood → lungs → trachea → swallowed → adult worm). In humans — a dead-end host — larvae released from ingested eggs penetrate the intestinal wall, enter the vasculature, and migrate throughout the body (liver, lungs, CNS, eyes) but cannot mature into adult worms. When larvae eventually die, they trigger intense immediate-type and delayed-type hypersensitivity, producing eosinophilic granulomas at each site of migration.
VLM and OLM appear to be mutually exclusive in most patients.
Clinical Manifestations
| Syndrome | Features |
|---|---|
| Covert toxocariasis | Persistent eosinophilia only; most common presentation; larval migration continues for years |
| Visceral larva migrans | Low-grade fever, cough, wheeze, hepatosplenomegaly, RUQ pain; resolves over 4–8 weeks; less commonly myocarditis, nephritis |
| Neurotoxocariasis | Eosinophilic meningoencephalitis, space-occupying lesions, myelitis, cerebral vasculitis, seizures |
| Ocular larva migrans | Unilateral visual impairment ± strabismus; uveitis, endophthalmitis, retinal granulomas; may mimic retinoblastoma or TB |
Laboratory findings: leukocytosis with marked eosinophilia, hypergammaglobulinemia, elevated isohemagglutinin titers to A/B blood group antigens (in VLM).
Imaging
Liver ultrasound or CT in VLM may show multiple hypodense nodules <2 cm, representing inflammatory granulomas. These resolve to calcific scars after treatment.
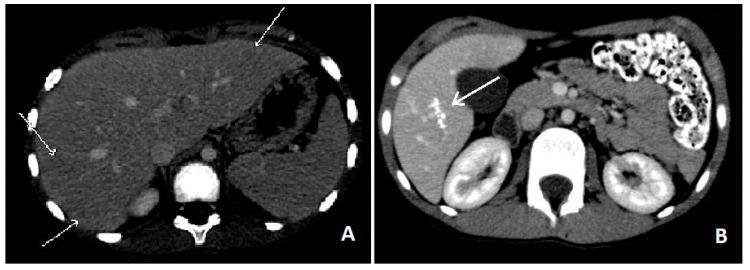
CT of hepatic toxocariasis: (A) acute phase — multiple small hypodense nodules (<10 mm) scattered in the liver parenchyma; (B) follow-up one year later — active nodules have resolved, leaving residual calcific granulomas (arrow). — Goldman-Cecil Medicine
For OLM, CT and fluorescein angiography help differentiate from retinoblastoma. Elevated anti-Toxocara antibodies in aqueous/vitreous humor relative to serum support the diagnosis.
Diagnosis
- ELISA for anti-Toxocara antibodies (available via CDC) + confirmatory immunoblot — does not distinguish active from past infection
- Recombinant larval antigen ELISAs have improved sensitivity/specificity but are not yet widely available
- PCR not currently available
- Liver biopsy (demonstrates eosinophilic granulomas, larvae rarely seen) — low sensitivity, not routinely recommended
- Stool examination is not useful (humans are dead-end hosts; no adult worms form)
Treatment
| Setting | Regimen |
|---|---|
| VLM (acute) | Albendazole 400 mg PO twice daily × 5 days |
| Severe disease (cardiac, CNS, pulmonary) | Albendazole + prednisone 60 mg/day × 5 days |
| OLM | Corticosteroids (prednisone 30–60 mg/day × 2 weeks) ± albendazole ± vitrectomy (with specialist supervision) |
| Alternative | Mebendazole |
— Goldman-Cecil Medicine; Red Book 2021
Fascioliasis
Etiology & Epidemiology
Fascioliasis is a zoonosis caused by the liver fluke Fasciola hepatica (adult: 30 × 13 mm, leaf-shaped) or the larger F. gigantica (adult: up to 75 × 20 mm). Natural definitive hosts include cattle, sheep, and goats. An estimated 2.6–17 million humans are infected globally, mainly in South America (Peru, Bolivia >60% prevalence), Africa, and Asia. Women are more frequently and more severely affected than men.

Gross specimen of Fasciola hepatica extracted via ERCP — brownish, dorsoventrally flattened, leaf-shaped, ~2 cm in length.
Life Cycle
- Eggs excreted in feces → deposited in water
- Miracidia hatch (9–14 days) → infect freshwater snails (Lymnaea spp.) → sporocysts → rediae → cercariae (4–7 weeks)
- Free-swimming cercariae encyst as metacercariae on aquatic vegetation (watercress, water lettuce, alfalfa, mint, parsley, khat)
- Humans infected by ingestion of raw contaminated vegetation or water
- Larvae excyst in duodenum → penetrate bowel wall → migrate through peritoneal cavity → reach liver within 4 weeks → penetrate Glisson's capsule
- Migrate through liver parenchyma for 3–5 months → enter bile ducts → mature adults lay eggs (→ passed via sphincter of Oddi into stool)
- Adults may survive in bile ducts for up to 13 years
Histopathology & Imaging
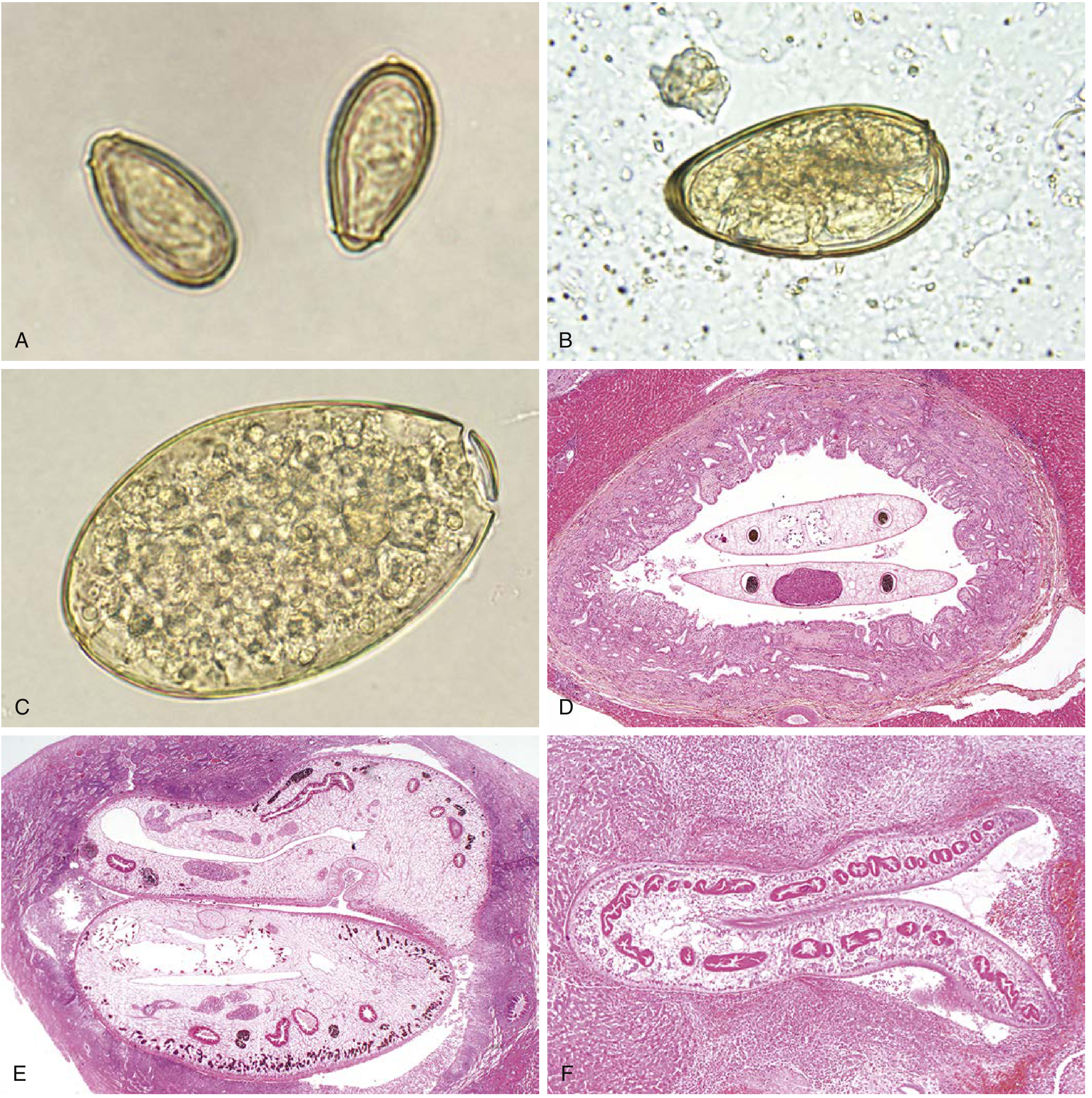
Panel C: Egg of F. hepatica / F. buski in stool (operculate, yellow-brown, 130–150 µm, indistinguishable from Fasciolopsis). Panel F: Adult F. hepatica in a liver bile duct (H&E, 40×). — Henry's Clinical Diagnosis & Management
Clinical Phases
Acute (Invasive/Migratory) Phase
Corresponds to larval migration through liver parenchyma:
- Prolonged fever, hepatomegaly, right upper quadrant / epigastric pain, marked eosinophilia
- Anorexia, weight loss, nausea, vomiting, cough, diarrhea, urticaria, lymphadenopathy, arthralgias
- Acute diarrhea (2–5 days) may precede hepatic invasion
- Hyperbilirubinemia is notably absent in this phase
- CT shows multiple hypodense lesions that change in position, attenuation, and shape over time (migrating parasites) — can mimic metastases; also subcapsular hematoma, tracklike serpiginous lesions (2–10 mm)
- Ectopic/extrahepatic migration (rare): subcutaneous tissue, pancreas, eye, brain, stomach wall
Chronic (Biliary) Phase
Begins when flukes reach the bile ducts:
- Colicky RUQ pain, biliary obstruction symptoms
- Eosinophilia may be absent in up to half of cases
- Bile ducts become dilated, thick-walled, fibrotic, and calcareous
- Complications: cholangitis, choledocholithiasis, hemobilia, liver abscess, liver fibrosis
- Elevated serum alkaline phosphatase (biliary obstruction)
- Adult flukes may be visible on ERCP
- Adults can survive in bile ducts up to 13 years
Diagnosis
| Method | Notes |
|---|---|
| Stool examination | Unembryo-nated, yellow-brown, operculate eggs, 130–150 × 63–90 µm; indistinguishable from Fasciolopsis buski; stool negative in acute phase (adults not yet in biliary tree) |
| Serology (Fas2 ELISA) | Method of choice in acute phase; also used chronically; available at CDC |
| CT/MRI/US | Tracklike hypodense lesions, subcapsular hematomas, nodular lesions, biliary thickening |
| ERCP | Can visualize and extract adult flukes from bile ducts |
| Spurious infection | Positive stool eggs after eating infected raw liver — exclude by repeat stool after dietary exclusion |
Treatment
Triclabendazole (TCZ) is the drug of choice — unique among liver flukes, as Fasciola is resistant to praziquantel:
- Standard adult dose: 10 mg/kg once (or 10 mg/kg × 2 doses 12–24 hours apart for severe infection)
- Praziquantel is ineffective against Fasciola spp.
- Surgical/endoscopic intervention (ERCP) may be required for biliary obstruction
Key Comparison
| Feature | Toxocariasis | Fascioliasis |
|---|---|---|
| Causative agent | T. canis / T. cati (nematode) | F. hepatica / F. gigantica (trematode) |
| Source | Dog/cat feces (soil) | Aquatic vegetation / contaminated water |
| Host status | Humans = dead-end host | Humans = definitive host (adult worms present) |
| Larval vs. adult | Larvae only; no adult worms | Adult flukes in bile ducts |
| Eosinophilia | Prominent | Prominent in acute; may be absent in chronic |
| Stool exam | Not useful | Diagnostic in chronic phase |
| Key diagnosis tool | Serology (ELISA + immunoblot) | Serology (Fas2 ELISA); stool eggs |
| Treatment | Albendazole | Triclabendazole |
| Praziquantel | Not applicable | Ineffective |
— Goldman-Cecil Medicine, 26th ed.; Red Book 2021; Henry's Clinical Diagnosis; Tietz Textbook of Laboratory Medicine
Toxocariasis. Fascioliasis. Etiology and life cycles Epidemiological characteristics Pathogenesis of visceral and hepatic involvement Clinical manifestations Laboratory and serological diagnostics Treatment principles Preventive and hygienic measures
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
Toxocara canis egg embryonated soil life cycle
Searching Images
Fasciola hepatica egg operculate stool microscopy
Loading Image
Loading Image
Loading Image
Loading Image
TOXOCARIASIS
1. Etiology and Life Cycle
Causative agents: Toxocara canis (dog roundworm, most common) and Toxocara cati (cat ascarid). Both are intestinal nematodes (Phylum Nematoda). Rare cases with eosinophilic meningoencephalitis are caused by the raccoon ascarid Baylisascaris procyonis.
Life cycle in the natural (animal) host:
- Infective eggs ingested by dog → larvae hatch, penetrate intestinal wall → migrate via portal blood to liver, then lungs → ascend bronchial tree → swallowed → develop to adult worms in small intestine → lay eggs excreted in feces.
- In pregnant bitches, dormant somatic larvae reactivate → infect puppies transplacentally and via suckling (transmammary transmission) — making puppies the primary egg-shedding reservoir.
- Eggs require 2–4 weeks of embryonation in the environment before becoming infectious. They do NOT hatch immediately upon shedding.
Life cycle in humans (dead-end / accidental host):
Humans ingest embryonated eggs → larvae hatch in the small intestine → penetrate the intestinal mucosa → enter the portal circulation → disseminate to liver, lungs, CNS, eyes, and other organs → cannot develop into adult worms → larvae remain in tissues, continue migrating, and eventually die, triggering eosinophilic granuloma formation.
Because no adult worms ever form in humans, eggs are never found in human stool — stool examination is useless for diagnosis.
2. Epidemiological Characteristics
| Feature | Detail |
|---|---|
| Global distribution | Ubiquitous worldwide; highest seroprevalence in tropical/subtropical zones |
| Seroprevalence (USA) | 5% of the population ≥6 years old (nationally representative survey) |
| Age predilection — VLM | Children 2–7 years (pica, soil contact in playgrounds/sandboxes) |
| Age predilection — OLM | Older children and adolescents 5–10+ years |
| Risk factors | Dog/cat ownership, poverty, pica, living in hot and humid regions (eggs remain viable long-term), frequenting sandboxes or parks |
| Source of infection | Ingestion of soil/sand contaminated with infective eggs; occasionally ingestion of raw or undercooked paratenic hosts (e.g., rodents, chicken livers) |
| Person-to-person spread | None — no direct transmission |
| Key reservoir | Puppies and kittens (highest egg output) |
Eggs can survive in soil for months to years under favorable conditions (warm, moist environment). Direct animal contact is not necessary, as freshly shed feces are not immediately infective (embryonation required).
3. Pathogenesis of Visceral and Ocular Involvement
After penetrating the intestinal wall, larvae enter the portal circulation and are carried to:
- Liver — most common initial target: larvae lodge in sinusoids, triggering focal eosinophilic infiltration and granuloma formation. Granulomas consist of eosinophils, macrophages, epithelioid cells, and giant cells surrounding a central larva or necrotic track. Hepatomegaly results from cumulative granulomatous inflammation.
- Lungs — larvae traverse pulmonary capillaries, eliciting eosinophilic pneumonitis with wheezing and transient infiltrates.
- CNS — larvae cross the blood–brain barrier → eosinophilic meningoencephalitis, cerebral vasculitis, space-occupying granulomas, myelitis.
- Eyes — larvae that reach the retinal vasculature form a single eosinophilic granulomatous mass (usually in the posterior pole) around the entrapped larva, causing fibrous traction, retinal detachment, and loss of vision.
- Myocardium, kidney — less common sites.
Immunological mechanism: Dying larvae release excretory-secretory (ES) antigens that trigger:
- Immediate-type (Type I) hypersensitivity → IgE production, mast cell degranulation, eosinophil recruitment, urticaria, wheezing.
- Delayed-type (Type IV) hypersensitivity → macrophage/T-cell-mediated granuloma formation.
The degree of clinical disease depends on larval burden, tissue distribution, reinfection frequency, and host immune response. VLM and OLM are mutually exclusive in most patients — this likely reflects larval tropism under differing host immune conditions.
4. Clinical Manifestations
Covert Toxocariasis
- Most common form.
- Persistent peripheral eosinophilia, often without systemic symptoms.
- Represents the continuation of larval migration without heavy burden.
Visceral Larva Migrans (VLM)
- Fever (low-grade, prolonged), malaise, anorexia, weight loss.
- Pulmonary: cough, wheezing, dyspnea; transient pulmonary infiltrates on chest X-ray (~50% of symptomatic patients).
- Hepatosplenomegaly → right upper quadrant pain.
- Eosinophilia: can be extreme (up to 90% of leukocytes); leukocytosis, hypergammaglobulinemia.
- Urticaria or rash (IgE-mediated).
- Symptoms develop gradually and resolve over 4–8 weeks with or without treatment.
- Uncommon but serious: myocarditis, nephritis, eosinophilic meningoencephalitis.
- Rare deaths from severe neurologic, pneumonic, or myocardial involvement.
Neurotoxocariasis
- Eosinophilic meningoencephalitis, space-occupying brain lesions, myelitis, cerebral vasculitis.
- Seizures, encephalopathy, neuropsychiatric symptoms.
Ocular Larva Migrans (OLM)
- Affects older children and adolescents; usually no history of pica, no peripheral eosinophilia, no systemic features.
- Unilateral visual impairment, strabismus, eye pain.
- Fundoscopy: whitish eosinophilic granulomatous mass in the posterior pole of the retina, or endophthalmitis, uveitis, chorioretinitis.
- Can mimic retinoblastoma (leading to unnecessary enucleation if misdiagnosed).
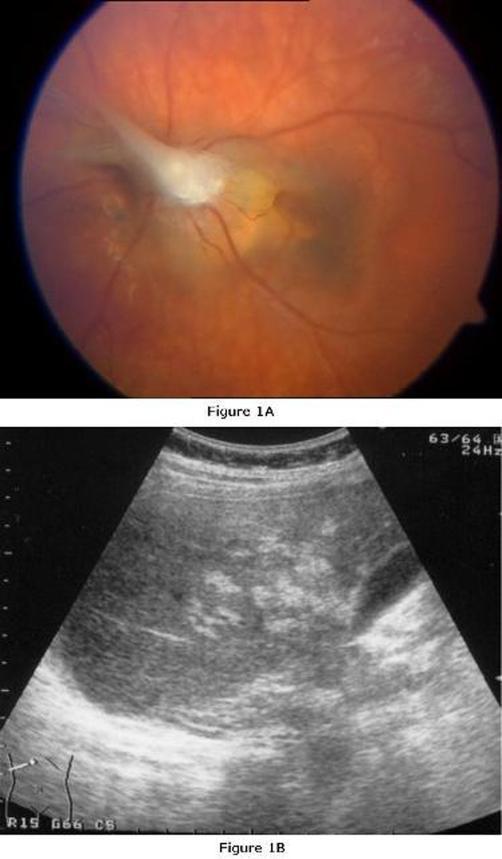
Top (Fig. 1A): Ocular larva migrans — whitish epiretinal granulomatous lesion near the optic disc with adjacent chorioretinal scarring. Bottom (Fig. 1B): Visceral larva migrans — multiple hyperechoic hepatic granulomas on ultrasound.
5. Laboratory and Serological Diagnostics
| Test | Notes |
|---|---|
| CBC | Leukocytosis with marked eosinophilia (can reach 70–90%); hypergammaglobulinemia; occasional anemia |
| Isohemagglutinins | Elevated anti-A and anti-B isohemagglutinin titers — characteristic of VLM |
| ELISA (anti-Toxocara) | Method of choice; detects IgG antibodies to larval-stage excretory-secretory antigens; CDC reference lab preferred over commercial labs; does not distinguish active from past infection |
| Immunoblot (Western blot) | Used to confirm positive ELISA; improves specificity |
| Recombinant antigen ELISA | Higher sensitivity and specificity; not yet widely available |
| ELISA on aqueous/vitreous humor | Elevated antibody titers relative to serum support OLM diagnosis (test less sensitive than for VLM) |
| Stool examination | Useless — no adult worms in humans, no eggs produced |
| PCR | Not currently available clinically |
| Liver biopsy | May show eosinophilic granulomas (larvae rarely identified); low sensitivity and not routinely recommended |
| Imaging — liver US/CT/MRI | Multiple hypodense/hypoechoic nodules <2 cm; resolve to calcifications after treatment |
| Chest X-ray | Transient pulmonary infiltrates in ~50% of symptomatic VLM |
| CT/fluorescein angiography (OLM) | Differentiates retinal granuloma from retinoblastoma |

CT abdomen: acute hepatic toxocariasis (A) — multiple small hypodense nodules; follow-up one year post-treatment (B) — residual calcific granulomas (arrow).
6. Treatment Principles
| Setting | Drug | Regimen |
|---|---|---|
| VLM (acute) | Albendazole | 400 mg PO twice daily × 5 days |
| Alternative (VLM) | Mebendazole | Per weight-based dosing |
| Severe VLM (CNS, cardiac, pulmonary) | Albendazole + corticosteroids | Prednisone 60 mg/day × 5 days (or per severity) added concurrently |
| OLM | Corticosteroids ± albendazole | Prednisone 30–60 mg/day × 2 weeks; albendazole as adjunct; with ophthalmologist supervision |
| OLM complicated | Vitrectomy | For vitreous traction, retinal detachment |
Most light toxocara infections are self-limited and resolve without treatment. Anthelmintics are primarily indicated in symptomatic patients or those with significant eosinophilia. Corticosteroids address the inflammatory component but do not kill larvae; combination therapy is used in severe cases.
7. Preventive and Hygienic Measures
Individual/household level:
- Regular deworming of dogs and cats, especially puppies and kittens (highest egg output) — reduces environmental contamination.
- Prevent children from eating soil (pica); supervise play in parks and sandboxes.
- Cover sandboxes when not in use to prevent animal defecation.
- Thorough handwashing after contact with soil, animals, or animal feces.
- Do not allow dogs to defecate in playgrounds or children's play areas.
Environmental/public health level:
- Proper disposal of dog and cat feces; municipal by-laws on leash and fecal disposal.
- Routine veterinary deworming programs reduce the community reservoir.
- Public health education about geophagia (pica) risk, especially in low-income communities.
- No vaccine is available.
FASCIOLIASIS
1. Etiology and Life Cycle
Causative agents: Fasciola hepatica (common liver fluke; adult 30 × 13 mm, leaf-shaped) and Fasciola gigantica (adult up to 75 × 20 mm). Both are trematodes (flatworms, Phylum Platyhelminthes). F. hepatica is the major human pathogen globally; F. gigantica is found mainly in Africa and Asia.
Life cycle:
Adult flukes in host bile ducts
↓ (eggs pass via bile → feces)
Unembryonated eggs deposited in water
↓ (9–14 days)
Miracidia hatch → invade freshwater snails (Lymnaea spp.)
↓ (4–7 weeks in snail)
Sporocysts → Rediae → Cercariae
↓ (cercariae exit snail, swim freely)
Metacercariae encyst on aquatic vegetation
(watercress, water lettuce, alfalfa, mint, parsley, khat)
↓ HUMAN INFECTION (ingestion of contaminated plants/water)
Metacercariae excyst in duodenum
↓
Larvae penetrate intestinal wall → peritoneal cavity
↓ (~4 weeks)
Penetrate Glisson's capsule → migrate through liver parenchyma (3–5 months)
↓
Reach bile ducts → mature into adult flukes
↓ (~2 months after initial infection)
Adults lay eggs → eggs pass via sphincter of Oddi → stool
(Adults may survive in bile ducts for up to 13 years)
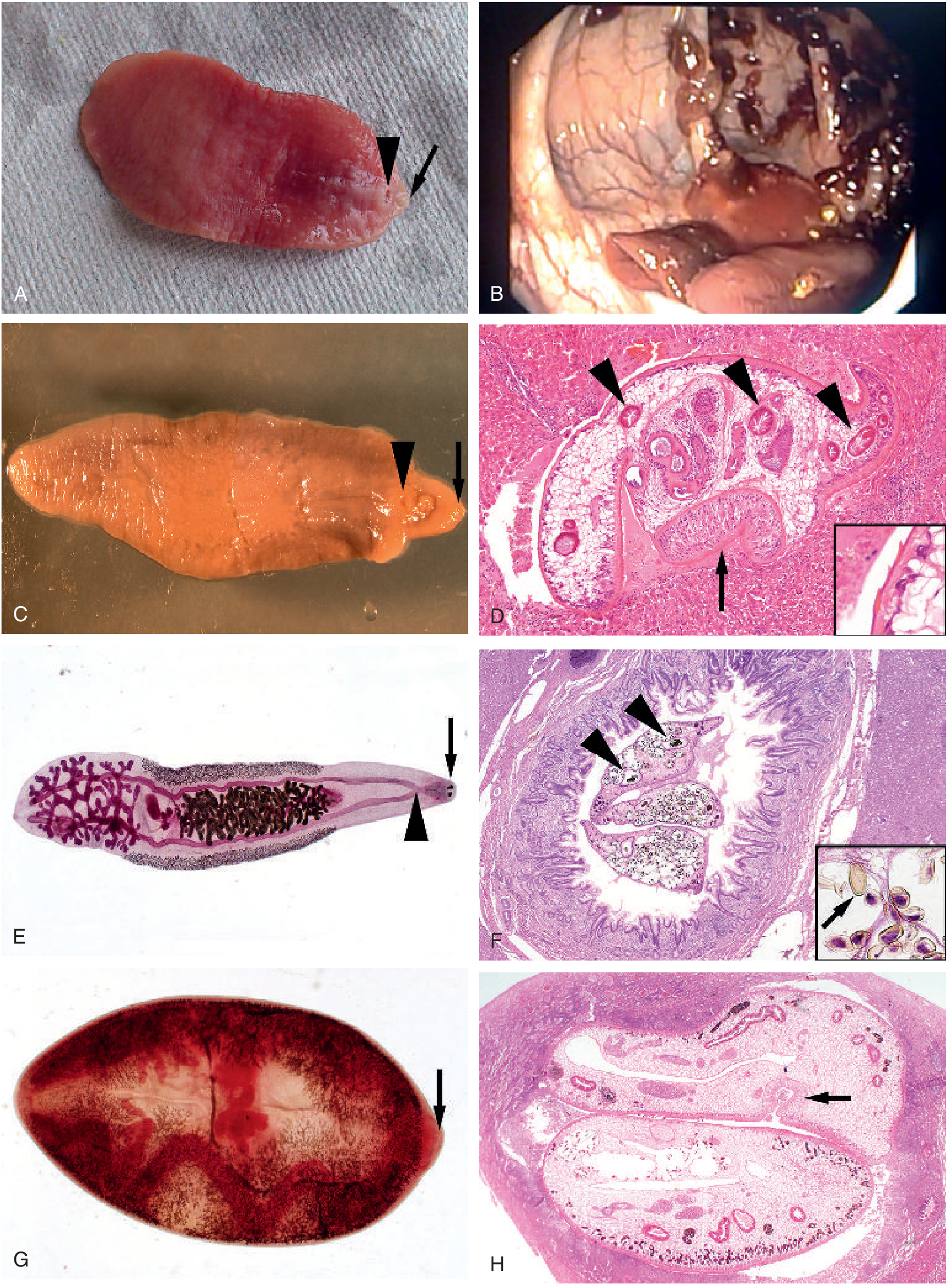
Trematode morphology panel. (C) Adult F. hepatica — leaf-shaped body with cone-shaped anterior, oral sucker (arrow), ventral sucker (arrowhead), ~3 cm in length. (D) F. hepatica migrating through liver parenchyma — multiple intestinal branches in cross-section (arrowheads), ventral sucker (arrow), cuticular spines on inset (H&E, 100×). — Tietz Textbook of Laboratory Medicine
2. Epidemiological Characteristics
| Feature | Detail |
|---|---|
| Distribution | Cosmopolitan (>70 countries); F. hepatica worldwide; F. gigantica in Africa and Asia only |
| Global burden | WHO estimates 2.4–17 million infected; 90,041 DALYs (2010 data) |
| Highest prevalence | Peru and Bolivia (>60%); also Egypt, Ethiopia, Islamic Republic of Iran, parts of Europe |
| Primary reservoir hosts | Cattle, sheep, goats |
| Intermediate host | Freshwater snails (Lymnaea spp.) |
| Route of infection | Ingestion of aquatic vegetation (especially watercress) or water contaminated with metacercariae |
| Sex differences | Women have higher incidence and more severe infections than men |
| At-risk populations | Rural populations in sheep/cattle-raising areas; travelers returning from endemic regions; immigrants |
| Seasonal variation | Linked to rainy season (increased snail habitat) and fresh vegetable availability |
3. Pathogenesis of Hepatic Involvement
Fascioliasis has two distinct pathological phases:
Acute (Invasive/Migratory) Phase — weeks to 3–5 months post-infection
- Intestinal phase: Metacercariae excyst in the duodenum. Acute diarrhea may occur for 2–5 days before liver invasion.
- Peritoneal phase: Larvae penetrate the bowel wall and traverse the peritoneal cavity.
- Hepatic migration: Larvae penetrate the liver capsule and burrow through the hepatic parenchyma for 3–5 months, creating inflammatory tracks. The ongoing inflammatory process is accompanied by:
- Eosinophilic infiltration at the migration front.
- Focal hemorrhage, necrosis, and abscess formation along larval tracks.
- Subcapsular liver hematoma in severe cases.
- Enhancement of Glisson's capsule on imaging.
- When larvae die during migration, they leave cavities filled with necrotic debris → replaced by scar tissue → eventually calcified granulomas.
- On CT: multiple hypodense lesions that change in position and shape over time (actively migrating parasites) — characteristic of acute fascioliasis. May mimic hepatic metastases or abscesses.
Chronic (Biliary) Phase — after flukes establish in bile ducts
- Mature adult flukes reside in hepatic and common bile ducts (and occasionally the gallbladder), consuming hepatocytes and duct epithelium.
- Bile ducts develop: dilatation, thick walls, hyperplastic epithelium, marked periductal fibrosis, calcareous deposits, and yellowish-brown bile.
- Biliary obstruction → elevated alkaline phosphatase, colicky pain.
- Bacterial superinfection → ascending cholangitis.
- Other complications: choledocholithiasis, hemobilia, liver abscess, liver fibrosis.
- Adults may survive in the biliary tree for up to 13 years.
- Ectopic migration (rare): subcutaneous tissue, pancreas, eye, brain, stomach wall — causing site-specific manifestations including ectopic pancreatitis (classic ERCP case: flukes extracted from the common bile duct causing acute pancreatitis).
4. Clinical Manifestations
Acute Phase
- Prolonged fever (weeks to months).
- Hepatomegaly with right upper quadrant / epigastric pain and tenderness.
- Marked eosinophilia (often the dominant laboratory finding).
- Anorexia, weight loss, nausea, vomiting.
- Cough (diaphragmatic irritation or pulmonary migration).
- Urticaria, arthralgia, lymphadenopathy.
- Mimics acute cholecystitis, but with significant eosinophilia and absent hyperbilirubinemia — a key distinguishing feature in the acute phase.
Chronic Phase
- Colicky RUQ pain (biliary colic pattern).
- Signs of biliary obstruction: jaundice, pale stools, dark urine.
- Elevated alkaline phosphatase; bilirubin rises (unlike acute phase).
- Eosinophilia may be absent in up to 50% of chronic cases.
- Recurrent bacterial cholangitis.
- Complications: choledocholithiasis, liver abscess, biliary cirrhosis, hemobilia.
Ectopic Fascioliasis
- Subcutaneous nodules (migratory), pancreatitis, ocular involvement, brain lesions.
5. Laboratory and Serological Diagnostics
| Test | Notes |
|---|---|
| Stool microscopy | Identification of large, yellow-brown, thin-shelled, operculate eggs: 130–150 × 63–90 µm (F. hepatica); up to 200 µm (F. gigantica). Stool is negative during the acute phase (adults not yet in bile ducts, no egg production). Available only 5–7 weeks after infection |
| Spurious passage | Eating infected raw sheep/cattle liver can result in false-positive stool eggs — exclude by dietary avoidance and repeat stool examination |
| Serology (Fas2 ELISA) | Method of choice in acute phase (before eggs appear in stool); uses excretory-secretory (ES) antigens; sensitivity >95%. Antibodies detectable 2–4 weeks after infection. Also useful for detecting ectopic infection and monitoring treatment response |
| Immunoblot | Used to confirm positive or equivocal EIA results; reduces cross-reactivity (main cross-reactant: schistosomiasis) |
| CDC reference laboratory | Serologic testing available in the USA only at the CDC |
| CBC | Eosinophilia (prominent in acute phase; absent in 50% chronic); leukocytosis; elevated ESR |
| LFTs | Elevated alkaline phosphatase (especially chronic phase); elevated ALT/AST; hyperbilirubinemia in chronic phase |
| Imaging | CT/MRI/US: tracklike hypodense lesions (2–10 mm, serpiginous/tortuous), subcapsular hematoma, multiple nodular hypodense lesions, enhancement of Glisson's capsule, cystic calcifications; bile duct thickening and dilatation in chronic phase |
| ERCP | Can visualize and extract adult flukes from the bile duct in chronic phase — simultaneously diagnostic and therapeutic |
| Liver biopsy | Rarely performed; flukes may be seen migrating through parenchyma (large size, cuticular spines, multiple intestinal branches on cross-section distinguish Fasciola from Clonorchis/Opisthorchis) |
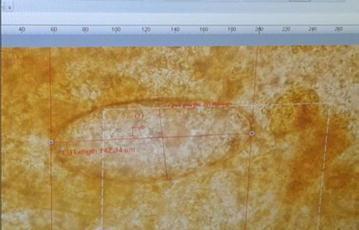
F. hepatica egg on stool microscopy — large (142 µm × 70 µm), oval, bile-stained, thin-shelled, with a small indistinct operculum at one end. Among the largest helminth eggs seen in humans. Morphologically indistinguishable from F. buski eggs.
6. Treatment Principles
Triclabendazole (TCZ) is the drug of choice for all forms of fascioliasis. Fasciola is uniquely resistant to praziquantel — a critical point that distinguishes it from all other liver, intestinal, and lung flukes.
| Setting | Drug | Dose |
|---|---|---|
| Fascioliasis (standard) | Triclabendazole | 10 mg/kg orally in a single dose (with food, to enhance bioavailability) |
| Severe/resistant infection | Triclabendazole | 10 mg/kg × 2 doses 12–24 hours apart |
| Biliary obstruction | ERCP with fluke extraction | Often combined with triclabendazole; may require sphincterotomy |
| Surgical | Rare; reserved for biliary stricture or unresolved obstruction |
- FDA approved triclabendazole for fascioliasis in patients aged ≥6 years.
- Serology is used to monitor treatment response (antibody titers should fall after successful treatment).
- Praziquantel is ineffective — must not be used for Fasciola.
7. Preventive and Hygienic Measures
Food safety:
- Do not eat raw watercress, water lettuce, or other aquatic plants from areas with grazing livestock.
- Wash and cook all aquatic vegetables thoroughly — metacercariae are killed by heat.
- Do not drink untreated water from streams or ponds in endemic areas; use filtered or boiled water.
- Avoid raw plant preparations (salads, juices) from endemic regions where contamination is plausible.
Agricultural and veterinary measures:
- Regular anthelmintic treatment of livestock (cattle, sheep, goats) reduces environmental egg contamination.
- Control of snail populations (Lymnaea spp.) in irrigation channels and water bodies (molluscicides, drainage management).
- Proper disposal of animal feces; prevent livestock from defecating near water sources or vegetable beds.
- Fencing of watercress cultivation sites to prevent livestock access.
Public health and surveillance:
- Health education in endemic communities about the risks of raw aquatic vegetable consumption.
- Surveillance and treatment programs in high-prevalence regions (Peru, Bolivia, Egypt).
- Travelers to endemic areas should be advised to avoid raw aquatic greens.
- WHO has included triclabendazole in its essential medicines list and preventive chemotherapy programs for fascioliasis.
Summary Comparison Table
| Feature | Toxocariasis | Fascioliasis |
|---|---|---|
| Pathogen | T. canis / T. cati (nematode) | F. hepatica / F. gigantica (trematode) |
| Infection route | Ingestion of embryonated eggs in soil | Ingestion of metacercariae on aquatic plants |
| Human host status | Dead-end — no adult worms, no eggs in stool | Definitive — adults in bile ducts, eggs in stool |
| Intermediate host | None (direct soil-to-human) | Freshwater snail (Lymnaea spp.) |
| Main target organ | Liver, lungs, CNS, eyes | Liver (parenchyma → bile ducts) |
| Eosinophilia | Marked; may reach 90% | Prominent in acute; often absent in chronic |
| Key clinical clue | VLM + eosinophilia in child; OLM mimics retinoblastoma | Acute hepatitis + eosinophilia + absent jaundice; chronic biliary disease |
| Stool examination | Useless | Diagnostic in chronic phase; negative in acute |
| Serology | ELISA + immunoblot (larval ES antigens) | Fas2 ELISA (>95% sensitivity); CDC only in USA |
| Treatment | Albendazole 400 mg bid × 5 days | Triclabendazole 10 mg/kg single dose |
| Praziquantel | N/A | Ineffective |
| Corticosteroids | Adjunct in severe VLM/OLM | Not typically indicated |
| Prevention | Deworm pets; cover sandboxes; handwashing | Cook aquatic vegetables; treated water; deworm livestock; snail control |
Sources: Harrison's Principles of Internal Medicine 22E; Goldman-Cecil Medicine International Edition; Red Book 2021 (AAP); Tietz Textbook of Laboratory Medicine 7th ed.; Henry's Clinical Diagnosis and Management; Medical Microbiology 9e.
This is a shared conversation. Sign in to Orris to start your own chat.