Aortic regurgitation
Reading File
Finding Sources
Searching PubMed
"aortic regurgitation"[MeSH Terms] AND management
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
aortic regurgitation echocardiography color Doppler
I now have comprehensive data from Braunwald's Heart Disease to deliver a thorough answer. Let me compile it.
Aortic Regurgitation (AR)
Definition & Pathophysiology
Aortic regurgitation is diastolic backflow of blood from the aorta into the left ventricle (LV) due to incompetent aortic valve leaflets or aortic root dilatation. Unlike mitral regurgitation — where the LV ejects into the low-pressure left atrium — in AR the entire stroke volume is ejected into the high-pressure aorta, imposing both increased preload (from regurgitant volume refilling the LV) and increased afterload (wall stress from LV dilation per Laplace's law).
The LV compensates via eccentric hypertrophy — sarcomeres replicate in series, myocytes elongate, and the LV dilates while maintaining wall thickness proportional to radius (normal end-diastolic wall stress). Over time, wall thickening fails to keep pace, end-systolic wall stress rises (afterload mismatch), and LVEF falls. LV mass in AR is often among the highest seen in any cardiac condition. — Braunwald's Heart Disease
Etiology
| Leaflet Abnormalities | Aortic Root Abnormalities |
|---|---|
| Rheumatic disease | Chronic hypertension |
| Bicuspid / unicuspid / quadricuspid valve | Marfan syndrome / annulo-aortic ectasia |
| Infective endocarditis | Aortic dissection |
| Myxomatous valve disease | Ehlers-Danlos / Osteogenesis imperfecta |
| Calcific valve disease | Ankylosing spondylitis / reactive arthritis |
| Post-TAVI paravalvular leak | Syphilitic aortitis |
| Leaflet fenestration, irradiation, trauma | Giant cell arteritis |
Root disease causes AR by distorting leaflet geometry even when leaflets themselves are normal — annular dilation reduces leaflet apposition.
Clinical Stages (ACC/AHA 2020)
| Stage | Definition | Echocardiographic Criteria |
|---|---|---|
| A | At risk | Bicuspid valve, aortic disease; no AR |
| B | Progressive (mild–mod) | Jet width <65% LVOT; vena contracta <0.6 cm; RVol <60 mL/beat; RF <50%; ERO <0.30 cm² |
| C1 | Asymptomatic severe, compensated | Jet width ≥65% LVOT; vena contracta >0.6 cm; RVol ≥60 mL; RF ≥50%; ERO ≥0.30 cm²; LVEF >55%; LVESD ≤50 mm |
| C2 | Asymptomatic severe, decompensated | As above + LVEF ≤55% or LVESD >50 mm |
| D | Symptomatic severe | Any of above + exertional dyspnea, angina, or HF symptoms |
— Braunwald's Heart Disease, Table 73.2
Clinical Features
Chronic AR
- Long asymptomatic period (sometimes decades)
- Symptoms: exertional dyspnea, orthopnea, angina (rare without CAD — due to reduced diastolic coronary perfusion + increased oxygen demand)
- Classic signs from widened pulse pressure (high systolic, low diastolic pressure):
- Corrigan's (water-hammer) pulse — bounding, rapidly collapsing
- de Musset's sign — head bobbing with each heartbeat
- Quincke's sign — pulsatile capillary pulsation in fingernails
- Duroziez's sign — systolic/diastolic bruit over femoral artery
- Hill's sign — popliteal SBP exceeds brachial SBP by >20 mmHg
- Traube's sign — "pistol-shot" femoral pulse sounds
Note: Widened pulse pressure is less helpful in older adults because age-related arterial stiffening produces it independently.
Auscultation
- High-pitched, blowing, decrescendo diastolic murmur heard best at lower left sternal border (valvular AR) or upper right sternal border (root AR), with patient sitting forward
- Austin Flint murmur: low-pitched mid-diastolic rumble at apex — from regurgitant jet impinging on anterior mitral leaflet, mimicking mitral stenosis
- Often accompanied by a systolic ejection murmur (due to increased forward stroke volume, not obstruction)
Diagnosis & Severity Assessment
Echocardiography (primary modality)
Structural assessment: identifies bicuspid valve, vegetations, leaflet prolapse, root dilation, annular dilation.
Color Doppler: regurgitant jet area in LVOT as % of LVOT width:
- Mild: <25% LVOT
- Moderate: 25–64%
- Severe: ≥65%
Quantitative parameters (severe AR):
- Vena contracta >0.6 cm
- Regurgitant volume ≥60 mL/beat
- Regurgitant fraction ≥50%
- ERO ≥0.30 cm²
- Holodiastolic flow reversal in the proximal abdominal aorta
- Short pressure half-time (<200 ms) on CW Doppler
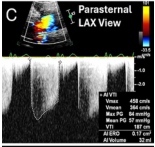
Parasternal LAX color Doppler and spectral Doppler of severe AR. ERO 0.17 cm², AI volume 32 mL, Vmax 458 cm/s.
LV response: LVESD, LVEDD, LVEF are tracked serially. LVEF ≤55% or LVESD >50 mm are surgical thresholds in asymptomatic patients.
CMR: Used when echo windows are suboptimal; accurately quantifies regurgitant fraction via phase-contrast flow measurement across the aortic root.
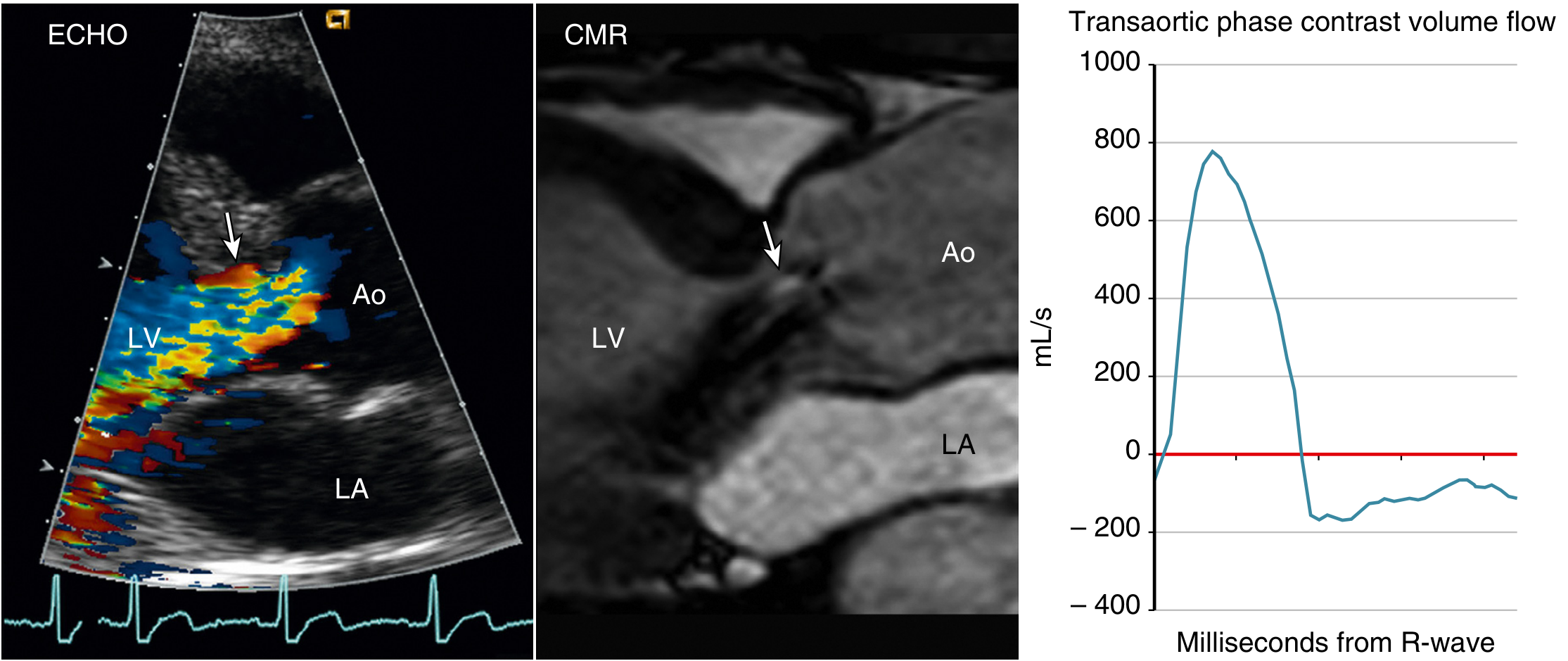
Fig. 12.17 — CMR quantification of severe AR from bicuspid valve. — Textbook of Clinical Echocardiography
Management
Chronic AR — Medical Therapy
- No disease-modifying drug therapy exists for AR; randomized trials of CCBs and ACE inhibitors have not shown consistent benefit in slowing LV dilation or delaying AVR.
- Treat hypertension (SBP >140 mm Hg) — vasodilators reduce regurgitant fraction by lowering diastolic arterial pressure.
- Manage CAD, arrhythmias, and comorbidities per guidelines.
- In inoperable symptomatic patients: evidence-based HF regimen — ACE inhibitors, diuretics, ± vasodilators; beta-blockers may help. Nitrates can be tried for angina.
- Pre-operative stabilization of decompensated LV: IV nitroprusside or vasodilators.
Chronic AR — Surgical Indications (AVR)
| Indication | Class |
|---|---|
| Any symptoms (NYHA I–IV) with severe chronic AR | I (mandatory) |
| Asymptomatic severe AR + LVEF ≤55% (C2) | I |
| Asymptomatic severe AR + LVESD >50 mm (or indexed >25 mm/m²) (C2) | I |
| Asymptomatic severe AR + undergoing other cardiac surgery | I |
| Asymptomatic severe AR + LVEF 55–60% or LVESD 50–60 mm on serial echo | IIa (reasonable) |
Key threshold update in 2020 ACC/AHA guidelines: LVEF <55% (previously <50%) is now the cut-off for surgery, as data show higher long-term mortality when operating below 55%. — Braunwald's Heart Disease
Operative mortality: <3% at experienced centers with normal LV; rises to ~14% when LVEF <35%.
Aortic valve repair (instead of replacement) is feasible in select patients — especially those with root dilation but preserved leaflets (Marfan syndrome), or bicuspid valve with isolated prolapse.
TAVR for AR: Increasingly reported in high-risk patients; standard TAVR is challenging because AR lacks the calcification that anchors transcatheter valves. Dedicated devices (e.g., JenaValve) are under investigation.
Acute Aortic Regurgitation
Causes
- Infective endocarditis (leaflet perforation/destruction)
- Aortic dissection (Type A)
- Trauma
Pathophysiology (distinct from chronic)
The LV is normal-sized and cannot accommodate the sudden large regurgitant volume. LV diastolic pressure rises rapidly above left atrial pressure → premature mitral valve closure → tachycardia (compensatory). Unlike chronic AR, there is no time for eccentric remodeling → severely reduced forward stroke volume → cardiogenic shock rapidly.
Presentation
- No widened pulse pressure (LV hasn't dilated yet)
- Soft or absent diastolic murmur (rapid equalization of aortic/LV pressures)
- Pulmonary edema on CXR; normal-sized cardiac silhouette (no cardiomegaly yet)
- Tachycardia, hemodynamic instability
Management — Surgical Emergency
- Prompt AVR is the primary treatment — acute severe AR is nearly always fatal without surgery
- Bridge therapy: IV dopamine or dobutamine (inotrope) + IV nitroprusside (vasodilator to reduce afterload)
- Beta-blockers are CONTRAINDICATED (tachycardia is compensatory; slowing HR causes decompensation)
- Intra-aortic balloon pump is CONTRAINDICATED (augments diastolic pressure → worsens regurgitation)
- Endocarditis: 5–7 days of IV antibiotics if hemodynamically stable, then surgery; operate immediately at any hemodynamic instability or abscess formation
- Aortic dissection causing AR: aortic repair/replacement combined with AVR
Surveillance (Asymptomatic Patients)
| Severity | Follow-up interval |
|---|---|
| Mild AR, normal LV | Every 3–5 years |
| Moderate AR | Every 1–2 years |
| Severe AR, compensated (C1) | Every 6–12 months; serial echo |
Exercise testing is reasonable in Stage C1 to unmask symptoms.
Key take-aways:
- Chronic AR causes a massive eccentric LV — the "cor bovinum." Compensation can last decades.
- Operate when symptomatic (any symptom), LVEF ≤55%, or LVESD >50 mm — don't wait for severe dysfunction.
- Acute AR is a surgical emergency; pulse pressure may be normal and the heart not enlarged — diagnosis is clinical + echo.
- No vasodilator drug therapy has proven benefit for delaying surgery in asymptomatic AR; treat hypertension and comorbidities.
— Braunwald's Heart Disease (9780323722193), Textbook of Clinical Echocardiography (9780323882088)
This is a shared conversation. Sign in to Orris to start your own chat.