Cushing syndrome
Reading File
Finding Sources
Searching PubMed
"Cushing syndrome"[MeSH Terms] AND diagnosis
Reading File
Reading File
Searching Images
Cushing syndrome clinical features moon face buffalo hump
Reading File
Reading File
Reading File
Excellent — I now have comprehensive content. Let me compile a thorough, well-structured response.
Cushing Syndrome
Definition
Cushing syndrome refers to the clinical state resulting from chronic excess of glucocorticoids, whether from endogenous overproduction or exogenous administration. When caused specifically by an ACTH-secreting pituitary adenoma, it is called Cushing disease (a subset of Cushing syndrome). The condition occurs in 2–5 per million people per year and has a peak onset in the 20s–30s, affecting women ~4× more than men in non-iatrogenic cases. — Campbell Walsh Wein Urology, p. 3133; Andrews' Diseases of the Skin, p. 578
Etiology & Classification
1. Exogenous (Iatrogenic) — Most Common Overall
- Exogenous glucocorticoid administration (oral, topical, inhaled, intra-articular, ocular, nasal, gingival)
- Serum cortisol is very low (adrenocortical suppression/hypoplasia)
2. ACTH-Dependent (~80–85% of endogenous cases)
| Cause | Frequency |
|---|---|
| Cushing disease (pituitary corticotroph microadenoma) | ~70% of endogenous |
| Ectopic ACTH syndrome (malignant tumors — small cell lung Ca, bronchial carcinoid, islet cell tumor, pheochromocytoma, medullary thyroid Ca) | ~10% |
| Ectopic CRH syndrome | <1% |
3. ACTH-Independent (~15–20% of endogenous cases)
| Cause | Frequency |
|---|---|
| Adrenal adenoma | ~8–10% |
| Adrenocortical carcinoma (ACC) | ~8% |
| ACTH-independent macronodular adrenal hyperplasia (AIMAH) | <1% |
| Primary pigmented nodular adrenocortical disease (PPNAD) — associated with Carney complex | <1% |
— Campbell Walsh Wein Urology, p. 3133–3136
Pathophysiology
The hypothalamic–pituitary–adrenal (HPA) axis normally maintains cortisol homeostasis via negative feedback:
- Hypothalamic CRH → anterior pituitary ACTH → adrenal cortex (zona fasciculata) produces up to 20 mg cortisol/day
- Cortisol feeds back to inhibit both CRH and ACTH
- Cortisol follows a circadian rhythm: peak in the morning, nadir ~11 PM
In Cushing syndrome, this feedback is lost or overwhelmed, leading to persistent, non-suppressible hypercortisolism with loss of diurnal variation. — Campbell Walsh Wein Urology, p. 3133–3134
Clinical Features
Classic Findings
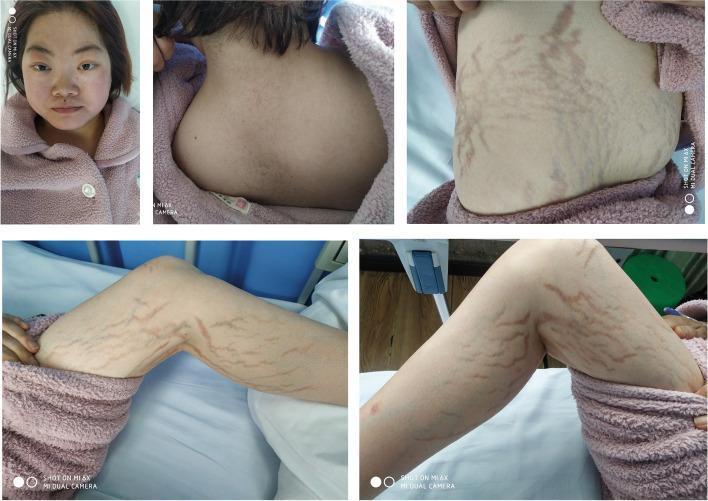
Classic features of Cushing syndrome in a 26-year-old female: moon facies, dorsocervical fat pad (buffalo hump), and wide violaceous striae across the abdomen and thighs — secondary to a left adrenal adenoma.
Characteristic signs (by system):
| System | Findings |
|---|---|
| Fat redistribution | Central/truncal obesity; moon facies (round, wide face); dorsocervical fat pad (buffalo hump); supraclavicular fat pads; limbs relatively spared |
| Skin | Violaceous abdominal striae (>1 cm wide); skin thinning/fragility; easy bruising; cigarette-paper wrinkling; Liddle sign (skin peels with tape removal); acne; hypertrichosis; facial plethora; facial lanugo in women |
| Musculoskeletal | Proximal muscle weakness/wasting; osteoporosis; pathologic fractures; kyphosis |
| Metabolic | Hypertension; dyslipidemia; insulin resistance; diabetes mellitus (~20%); hyperglycemia/glycosuria |
| Infections | Susceptibility to superficial dermatophyte, Pityrosporum, and opportunistic fungal infections |
| Reproductive | Oligomenorrhea/amenorrhea; hirsutism; loss of libido; impotence |
| Neuropsychiatric | Depression, cognitive impairment, psychosis |
| Renal | Hypokalemia (especially in ectopic ACTH), nephrolithiasis |
— Andrews' Diseases of the Skin, p. 577–578; Campbell Walsh Wein Urology, p. 3137
Ectopic ACTH syndrome often lacks classic Cushingoid fat redistribution (due to rapid onset), but presents with severe hypokalemic alkalosis, hyperpigmentation (from high ACTH stimulating melanocortin receptors), and cachexia from the underlying malignancy.
Diagnosis
Step 1 — Confirm Hypercortisolism (Screening Tests)
At least two of the following should be abnormal:
| Test | Threshold / Interpretation |
|---|---|
| 1 mg overnight dexamethasone suppression test (DST) | Cortisol < 50 nmol/L (1.8 µg/dL) at 8 AM = rules out (except iatrogenic) |
| 24-hour urinary free cortisol (UFC) | Elevated on ≥2 collections |
| Late-night salivary cortisol | Elevated (loss of diurnal nadir) |
| Low-dose DST (2 mg/day × 2 days) | 17-OHCS fail to suppress |
— Andrews' Diseases of the Skin, p. 578; Campbell Walsh Wein Urology, p. 3138
Step 2 — Determine ACTH Dependency
- Plasma ACTH (8 AM):
- ACTH suppressed (<5 pg/mL) → ACTH-independent (adrenal source)
- ACTH elevated/normal (>15–20 pg/mL) → ACTH-dependent
Step 3 — Localize the Source
If ACTH-dependent:
| Test | Purpose |
|---|---|
| Pituitary MRI | Identify corticotroph microadenoma |
| CRH stimulation test | Cushing disease: ACTH rises >35%; ectopic: minimal response |
| High-dose DST (8 mg overnight) | Cushing disease: cortisol suppresses >50%; ectopic: fails to suppress |
| Inferior petrosal sinus sampling (IPSS) | Gold standard to distinguish Cushing disease vs. ectopic ACTH |
| CT chest/abdomen/pelvis | Identify ectopic tumor (lung, pancreas) |
If ACTH-independent:
- CT/MRI adrenals to distinguish adenoma, carcinoma, AIMAH, PPNAD
Pseudo-Cushing Syndrome
Mimics true Cushing syndrome clinically and biochemically. Causes include:
- Alcohol abuse (most common) — resolves with abstinence
- Major depression
- Obesity
Treatment
1. Surgery (First-line for most causes)
- Cushing disease → Transsphenoidal pituitary adenomectomy (remission in ~65–90% when microadenoma found)
- Adrenal adenoma/carcinoma → Laparoscopic or open adrenalectomy
- Ectopic ACTH → Resection of primary tumor if possible
- Bilateral adrenal disease → Bilateral adrenalectomy (requires lifelong steroid replacement)
- Beware Nelson syndrome after bilateral adrenalectomy in Cushing disease (ACTH-secreting macroadenoma can expand)
2. Steroidogenesis Inhibitors (Medical)
Used pre-operatively, for inoperable disease, or while awaiting surgery:
- Metyrapone (11β-hydroxylase inhibitor) — most commonly used
- Ketoconazole (CYP450 inhibitor of adrenal enzymes)
- Mitotane (adrenolytic; mainly for ACC)
- Etomidate (IV; for rapid control in crisis)
- Osilodrostat, levoketoconazole (newer agents)
3. Pituitary-Directed (for Cushing disease)
- Pasireotide (somatostatin receptor ligand)
- Cabergoline (dopamine agonist)
- Mifepristone (GR antagonist; especially for diabetes in Cushing syndrome)
4. Radiation
- Stereotactic radiosurgery (Gamma Knife) for residual/recurrent pituitary tumor
Post-Treatment
- After successful surgery, contralateral normal adrenal tissue is suppressed — patients require glucocorticoid replacement for months (occasionally years) until the HPA axis recovers
- Cushingoid phenotype resolves in most patients within 7–9 months post-adrenalectomy — Campbell Walsh Wein Urology, p. 3136
Key Associations & Special Situations
| Condition | Associated Feature |
|---|---|
| Carney complex (PRKAR1A mutation) | PPNAD in 30%; spotty skin pigmentation, cardiac myxomas, Sertoli cell tumors |
| McCune-Albright | AIMAH; polyostotic fibrous dysplasia, café-au-lait spots |
| MEN type 1 | Rare feature |
| Adrenocortical carcinoma | Independent predictor of poor prognosis |
| Subclinical Cushing syndrome | Radiographically detected adrenal incidentaloma with mild biochemical hypercortisolism but no classic phenotype |
Recent Evidence (PubMed 2024)
- A 2024 systematic review on primary pigmented nodular adrenocortical disease (PPNAD) analyzed 210 patients, characterizing pathogenic variants and clinical features [PMID: 39006359].
- A 2024 meta-analysis on mild autonomous cortisol secretion (subclinical Cushing) found surgical adrenalectomy superior to conservative management for metabolic outcomes [PMID: 39086899].
- A 2024 meta-analysis characterized thyroid dysfunction as a common comorbidity in Cushing syndrome [PMID: 38840128].
Sources: Campbell Walsh Wein Urology (3-Volume Set), pp. 3133–3140 · Andrews' Diseases of the Skin, pp. 577–578 · Fitzpatrick's Dermatology, Vol. 1–2
Labour
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Searching Images
stages of labor cervical dilation uterine contractions cardinal movements fetal descent
Reading File
Reading File
Reading File
Reading File
I now have all the content needed. Let me compile the comprehensive response.
Labour (Parturition)
Definition
Labour is defined as the presence of sufficient uterine contractions in frequency, intensity, and duration to bring about effacement and dilation of the cervix, ultimately resulting in delivery of the fetus, placenta, and membranes. It is a continuous but clinically divisible process. — Textbook of Family Medicine 9e, p. 501
Physiology of Onset
The mechanisms initiating labour are complex and incompletely understood. Key mediators include:
- Prostaglandins (PGE₂ and PGF₂α) — evidence of central involvement in cervical ripening and myometrial activation
- Oxytocin — increases myometrial sensitivity near term via upregulation of oxytocin receptors
- Fetal adrenal maturation — cortisol surge contributes to the hormonal cascade
- Mechanical stretch of the uterus — activates stretch-sensitive pathways
Signs of Impending Labour
- Lightening: descent of the fetal head into the pelvis; may occur acutely or over weeks; relieved breathlessness but increased pelvic pressure
- Bloody show: passage of blood-tinged mucous plug from the cervix as early dilation begins; true labour usually follows within 3 days
- Braxton Hicks contractions: irregular, often painless preliminary contractions that help efface the cervix but do not produce regular dilation
True vs. False Labour
| Feature | True Labour | False Labour (Braxton Hicks) |
|---|---|---|
| Contraction rhythm | Regular | Irregular |
| Intervals | Gradually shorten | Unchanged |
| Intensity | Gradually increases | Unchanged |
| Location of pain | Back and abdomen | Lower abdomen only |
| Effect of sedation | None | Usually relieved |
| Cervical dilation | Yes | No |
— Tintinalli's Emergency Medicine, p. 679
Stages of Labour
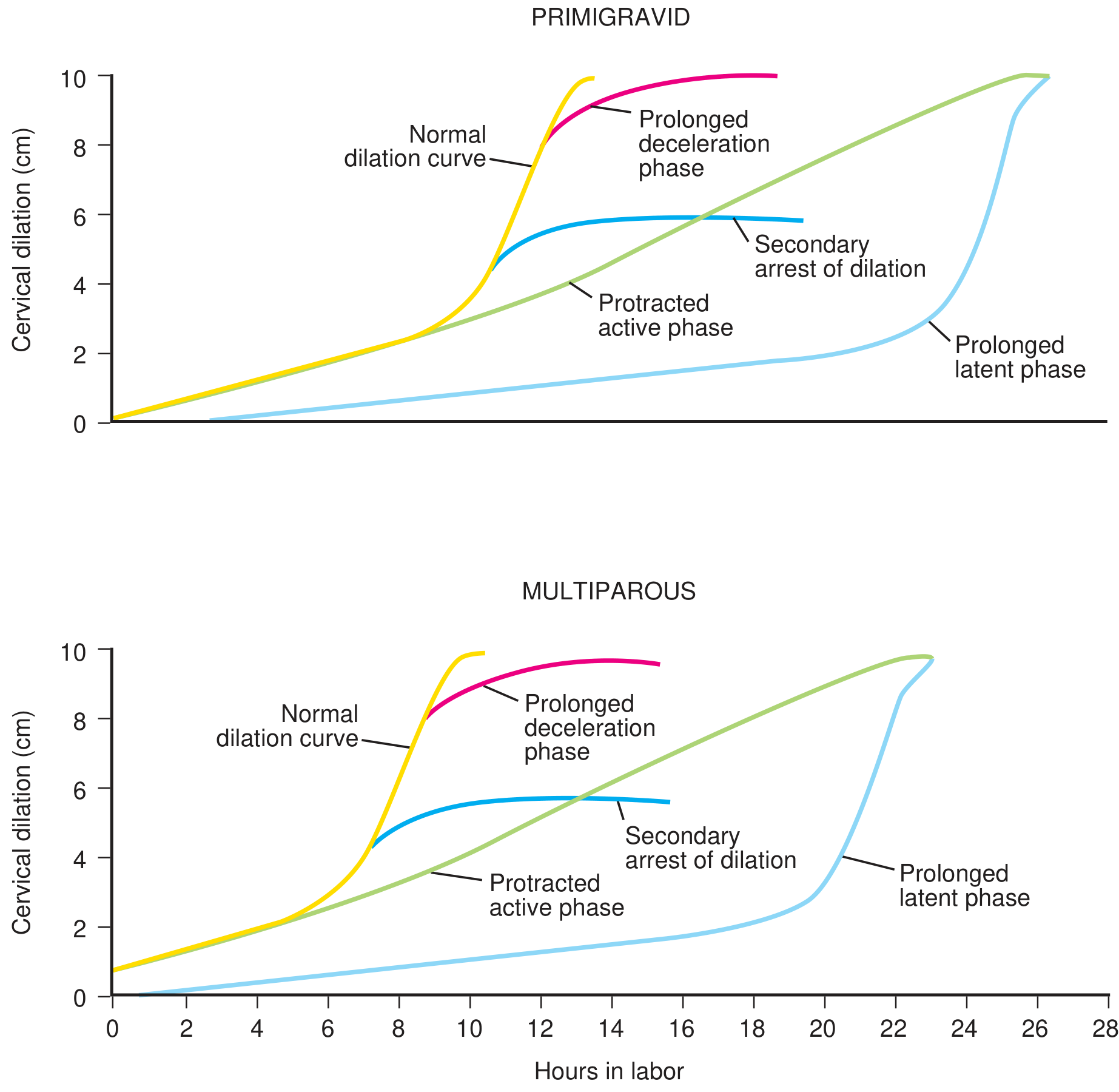
Composite curves of normal and abnormal labour progress — primigravid vs. multiparous. (From Textbook of Family Medicine 9e, p. 502)
First Stage — Onset of contractions → Full cervical dilation (10 cm)
Latent Phase
- Cervix effaces, softens, and moves anteriorly; dilation is slow (≤0.6 cm/hr)
- Duration: <20 hours (nullipara) / <14 hours (multipara)
- Contractions are mild, irregular
- Best managed at home; clear liquids encouraged
- Prolonged latent phase: treated conservatively (rest, hydration); therapeutic morphine may be used to allow rest and transition to active phase; amniotomy should be avoided (risk of chorioamnionitis)
Active Phase
- Begins at 3–4 cm dilation
- Rapid dilation: ≥1.2 cm/hr (nullipara) / ≥1.5 cm/hr (multipara)
- Contractions: every 2–3 minutes, lasting ~1 minute, ~40 mmHg intensity
- Fetal heart rate should be auscultated every 30 min (low risk) or every 15 min (higher risk); continuous EFM is an alternative
The 3 P's governing active phase progress:
- Power — strength and frequency of uterine contractions
- Passenger — size, position, and attitude of the fetal head
- Passage — size and shape of the bony pelvis
Second Stage — Full dilation → Delivery of fetus
- Duration: ~50 minutes (nullipara) / ~20 minutes (multipara)
- Maternal expulsive pushing during contractions aids fetal descent
- Fetal HR auscultated every 15 min (low risk) / every 10 min (high risk)
- Pushing >2 hours (without epidural) or >3 hours (with epidural) in a nullipara → evaluate for cephalopelvic disproportion (CPD)
Cardinal Movements of the Fetal Head
These sequential movements allow the fetal head to negotiate the changing diameters of the bony pelvis:
| Movement | Description |
|---|---|
| Engagement | Largest diameter of fetal head passes through pelvic inlet (station 0) |
| Descent | Continuous downward movement throughout labour |
| Flexion | Chin brought to chest → smallest diameter presented |
| Internal rotation | Head rotates to align occiput anteriorly (OA) at pelvic outlet |
| Extension | Head extends as it passes under pubic symphysis |
| External rotation (restitution) | Head returns to face obliquely as shoulders rotate internally |
| Expulsion | Delivery of shoulders and body |
— Textbook of Family Medicine 9e, p. 503
Delivery Technique
- Fetal head delivers by extension → suction mouth, pharynx, nose
- Nuchal cord: reduce if possible; clamp and cut if too tight
- Anterior shoulder: gentle downward traction + maternal pushing (avoid excess → brachial plexus injury)
- Posterior shoulder: gentle upward traction
- Cord clamping: timing is debated; delayed clamping is currently favoured
Third Stage — Delivery of fetus → Delivery of placenta
- Duration: usually <10 minutes; active management not required until 30 minutes unless haemorrhage occurs
- Retained placenta = not expelled within 60 minutes
Signs of placental separation:
- Lengthening (descent) of the cord
- Gush of blood
- Change in fundal contour (uterus becomes globular)
Mechanism: Uterine retraction reduces placental attachment area → haematoma forms deep to placenta → placenta shears off through the spongy layer of the decidua basalis → myometrial contractions constrict spiral arteries → haemostasis. — The Developing Human, p. 372
Active management of third stage (AMTSL):
- Oxytocin 10 units IM (or 20 units in 1L normal saline IV) immediately after delivery
- Controlled cord traction
- Uterine massage
Fourth Stage — Delivery of placenta → First hour postpartum
- Monitor for: excessive vaginal bleeding, boggy uterus, hypotension
- Uterine atony management:
- Fundal massage
- Oxytocin 20–40 units/L IV or 20 units IM
- Methylergonovine (Methergine) 0.2 mg IM q20 min (avoid in hypertensive patients)
- PGF₂α (Carboprost/Hemabate) 250 μg IM q15–20 min up to 3 doses
- Misoprostol 200–1000 μg (oral/sublingual/vaginal/rectal) — widely used off-label; use with caution in cardiovascular disease
— Textbook of Family Medicine 9e, pp. 503–504
Rupture of Membranes
- Spontaneous rupture (SROM) occurs during active labour in the majority; occurs before onset of labour in ~8% (PROM)
- 50% with PROM deliver within 5 hours; 95% within 28 hours
- Confirmed by:
- Nitrazine paper: amniotic fluid pH 7.0–7.4 → turns dark blue (vaginal fluid pH 4.5–5.5 = remains yellow); false positives with blood, semen, lubricant, Trichomonas
- Ferning: NaCl crystal pattern on microscopy slide as fluid dries
— Tintinalli's Emergency Medicine, p. 678
Fetal Monitoring
- Normal FHR: 120–160 bpm
- Bradycardia: <110 bpm; Tachycardia: >160 bpm
- Late decelerations: persistent drop in FHR during and >30 sec after contraction → sign of fetal distress
- Management of fetal distress: left lateral positioning, IV hydration, supplemental O₂, obstetric consultation
Dysfunctional Labour
| Disorder | Definition | Management |
|---|---|---|
| Prolonged latent phase | >20 hr (nullipara), >14 hr (multipara) | Rest, hydration, therapeutic morphine; avoid amniotomy |
| Protracted active phase | Dilation <1.2 cm/hr (nullipara) or <1.5 cm/hr (multipara) | Oxytocin augmentation; assess for malposition, CPD |
| Secondary arrest of dilation | No cervical change for ≥2 hr after previously normal dilation | Assess for CPD; intrauterine pressure catheter; amniotomy; oxytocin; consider operative delivery |
| Protracted descent (2nd stage) | <1 cm/hr descent (nullipara), <2 cm/hr (multipara) | Assess for CPD, macrosomia, inadequate pushing |
| Arrest of descent (failure of descent) | No change in station | High risk of CPD; consider operative delivery |
— Textbook of Family Medicine 9e, pp. 506–507
Pain Relief in Labour
| Method | Notes |
|---|---|
| Epidural analgesia | Most effective; may prolong active phase but does not affect outcome if contractions adequate |
| Systemic opioids | Pethidine/fentanyl; cause neonatal respiratory depression — have naloxone available |
| Nitrous oxide (Entonox) | Inhaled 50:50 N₂O/O₂; patient-controlled |
| Non-pharmacological | Hydrotherapy, ambulation, TENS, position changes, support persons |
Key Clinical Thresholds Summary
| Parameter | Nullipara | Multipara |
|---|---|---|
| Latent phase max duration | 20 hours | 14 hours |
| Active phase dilation rate | ≥1.2 cm/hr | ≥1.5 cm/hr |
| 2nd stage average | 50 minutes | 20 minutes |
| 2nd stage limit (no epidural) | 2 hours | — |
| 2nd stage limit (with epidural) | 3 hours | — |
| 3rd stage (normal) | <10–15 min | <10–15 min |
| Retained placenta threshold | 60 minutes | 60 minutes |
Sources: The Developing Human: Clinically Oriented Embryology, pp. 370–372 · Textbook of Family Medicine 9e, pp. 501–507 · Tintinalli's Emergency Medicine, pp. 678–680 · Creasy & Resnik's Maternal-Fetal Medicine, p. 969
This is a shared conversation. Sign in to Orris to start your own chat.