Give about antiretrovirals from Pharma
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Reading File
Searching PubMed
antiretroviral therapy HIV treatment
I now have comprehensive material from Katzung's pharmacology textbook. Let me compile the complete answer.
Antiretroviral Agents
— Katzung's Basic and Clinical Pharmacology, 16th Edition; Goodman & Gilman's Pharmacological Basis of Therapeutics
HIV Life Cycle & Sites of Drug Action
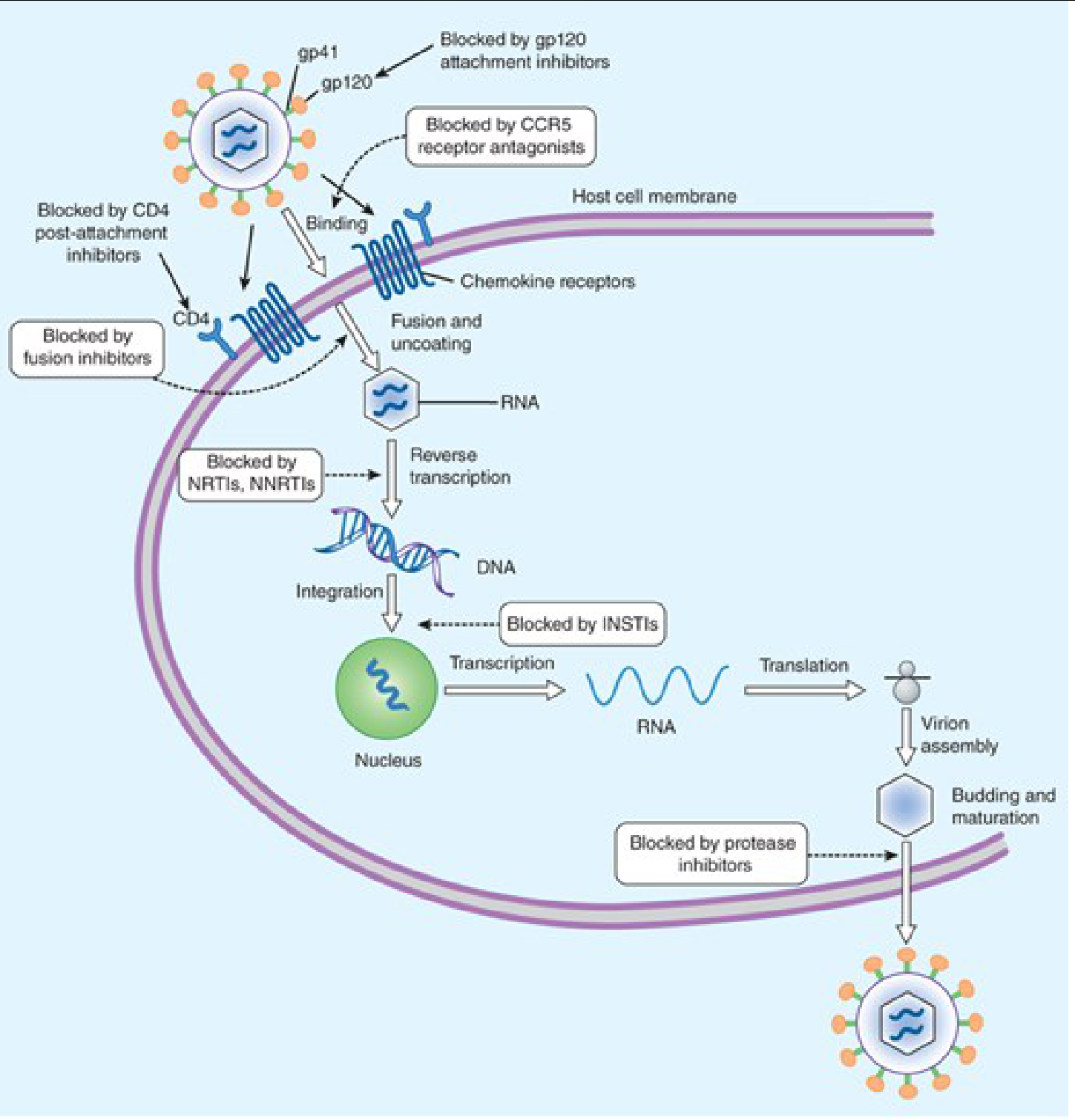
Each class of antiretroviral agent blocks a distinct step in the HIV replication cycle:
- gp120 attachment inhibitors → block initial attachment to CD4
- CCR5 antagonists → block co-receptor binding
- CD4 post-attachment inhibitors → block post-binding entry
- Fusion inhibitors → block membrane fusion via gp41
- NRTIs / NNRTIs → inhibit reverse transcription (RNA → DNA)
- INSTIs → block viral DNA integration into host chromosome
- Protease inhibitors (PIs) → block virion maturation (budding step)
- Capsid inhibitors → block capsid assembly/disassembly
Class 1: Nucleoside/Nucleotide Reverse Transcriptase Inhibitors (NRTIs)
Mechanism: Analogues of natural nucleosides → phosphorylated intracellularly to active triphosphates → competitively inhibit HIV reverse transcriptase → cause chain termination.
Class toxicity: Mitochondrial toxicity (via inhibition of mitochondrial DNA polymerase γ) → lactic acidosis, hepatic steatosis, lipoatrophy, peripheral neuropathy.
| Drug | Key Features | Toxicities |
|---|---|---|
| Zidovudine (ZDV/AZT) | First ARV (1987); thymidine analog; CSF penetration good | Bone marrow suppression (anemia, neutropenia), myopathy, macrocytosis |
| Abacavir (ABC) | Test HLA-B*5701 before use; avoid alcohol | Fatal hypersensitivity reaction (rash, fever, GI); possible ↑ MI risk |
| Emtricitabine (FTC) | Fluorinated lamivudine analog; intracellular t½ ≥24 h; only FDA-approved PrEP backbone (with TDF) | Headache, rash, hyperpigmentation of palms/soles (up to 13% in African Americans) |
| Lamivudine (3TC) | Active vs. HIV-1 & HBV; oral bioavailability >80% | Headache, GI discomfort; pancreatitis rare |
| Tenofovir DF (TDF) | Nucleotide prodrug; requires only 2 phosphorylation steps; active vs. HIV + HBV | Nephrotoxicity (proximal tubular dysfunction, Fanconi syndrome), ↓ bone density |
| Tenofovir AF (TAF) | Newer prodrug; better intracellular delivery → lower plasma levels → less renal/bone toxicity | Weight gain; lipid changes |
| Stavudine (d4T) | No longer preferred; thymidine analog | High mitochondrial toxicity, lipoatrophy, peripheral neuropathy |
| Didanosine (ddI) | Must fast (give on empty stomach) | Peripheral neuropathy, pancreatitis, retinal changes |
Note: Emtricitabine + lamivudine must not be combined — they compete for intracellular phosphorylation and both select for the M184V/I resistance mutation.
HBV co-infection: Stopping drugs active against HBV (3TC, FTC, TDF, TAF) can precipitate severe HBV flares.
Class 2: Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs)
Mechanism: Bind allosterically to a hydrophobic pocket near the active site of reverse transcriptase → conformational change → inhibit catalytic activity. Not phosphorylated; active against HIV-1 only (not HIV-2).
| Drug | Key Features | Toxicities / Drug Interactions |
|---|---|---|
| Efavirenz (EFV) | Once daily; CNS penetration excellent; teratogenic (avoid in 1st trimester) | Vivid dreams, dizziness, CNS disturbances, rash; CYP3A4 inducer |
| Nevirapine (NVP) | CYP3A4 inducer; risk of severe hepatotoxicity especially in women with CD4 >250 | Severe rash (Steven–Johnson syndrome), hepatotoxicity; must lead-in with low dose for 14 days |
| Rilpivirine (RPV) | Must be taken with a ≥500 kcal meal; contraindicated with PPIs; also available as LA injection (monthly) | Depression, insomnia, rash; lower barrier to resistance than EFV |
| Etravirine (ETR) | Active against some NNRTI-resistant strains; twice daily | Rash, nausea; multiple drug interactions (CYP3A4, CYP2C9 inhibitor/inducer) |
| Doravirine (DOR) | Once daily; no food restriction; minimal drug interactions | Dizziness, nausea, abnormal dreams |
Class 3: Protease Inhibitors (PIs)
Mechanism: Inhibit HIV aspartyl protease → prevent cleavage of the Gag-Pol polyprotein → virions remain immature and non-infectious.
Class toxicities: GI intolerance, hyperlipidemia (↑ triglycerides, ↑ LDL), insulin resistance, lipodystrophy, elevated liver enzymes.
Boosting: All modern PIs require co-administration with ritonavir (100 mg) or cobicistat (150 mg) as pharmacokinetic boosters. Ritonavir itself is a potent CYP3A4 inhibitor.
| Drug | Key Features | Toxicities |
|---|---|---|
| Atazanavir (ATV) | Once daily; no insulin resistance or ↑ CV risk; needs acid for absorption — separate from antacids/PPIs | Indirect hyperbilirubinemia/jaundice (~10%), PR prolongation, renal stones, rash |
| Darunavir (DRV) | High genetic barrier to resistance; sulfonamide moiety → sulfa allergy risk; monitor LFTs | Diarrhea, rash, hepatotoxicity |
| Lopinavir/ritonavir (LPV/r) | Fixed-dose; no additional booster needed | GI toxicity, QTc prolongation, lipid effects |
Class 4: Integrase Strand Transfer Inhibitors (INSTIs)
Mechanism: Inhibit HIV integrase → block insertion of viral dsDNA into host genome. Highest barrier to resistance among all ARV classes (especially dolutegravir, bictegravir).
| Drug | Key Features | Toxicities |
|---|---|---|
| Raltegravir (RAL) | First INSTI approved; twice daily; no CYP3A4 interactions (glucuronidated via UGT1A1) | Well tolerated; creatine kinase elevation, myopathy rare |
| Elvitegravir (EVG) | Requires cobicistat boosting; once daily; part of single-tablet regimens (Stribild, Genvoya) | GI effects; multiple drug interactions via cobicistat |
| Dolutegravir (DTG) | No boosting needed; high barrier to resistance; minimal drug interactions; preferred in pregnancy | Weight gain, insomnia, headache; neural tube defect signal with periconceptional use (controversial but label caution maintained) |
| Bictegravir (BIC) | Only available in fixed-dose tablet (BIC/TAF/FTC = Biktarvy); once daily; high genetic barrier | Nausea, diarrhea; separate from antacids by ≥2 h |
| Cabotegravir (CAB) | Available as LA IM injection (monthly or every 2 months) with rilpivirine; also oral for PrEP | Injection-site reactions; tail pharmacokinetics — drug detectable >1 year after last injection |
Class 5: Entry/Fusion Inhibitors
Fusion Inhibitor
- Enfuvirtide (T-20): Peptide that mimics the HR2 domain of gp41 → prevents membrane fusion. Subcutaneous injection twice daily. Major drawback: injection-site reactions in nearly 100% of patients. Reserved for salvage therapy in multidrug-resistant HIV.
CCR5 Co-receptor Antagonist
- Maraviroc: Binds host CCR5 → prevents HIV-CCR5 interaction. Requires tropism testing first — only active against CCR5-tropic virus (R5), NOT against CXCR4- or dual/mixed-tropic virus. Substrate for CYP3A4 and P-glycoprotein (dose must be adjusted with CYP3A4 inhibitors/inducers). Adverse effects: hepatotoxicity (preceded by allergic reaction), myocardial ischemia, postural hypotension.
CD4 Post-Attachment Inhibitor
- Ibalizumab: Monoclonal antibody binding CD4 domain 2 → blocks post-attachment conformational changes needed for HIV entry. Given as IV infusion every 2 weeks. Indicated for heavily treatment-experienced adults with multidrug-resistant HIV-1.
gp120 Attachment Inhibitor
- Fostemsavir: Prodrug converted to temsavir → binds HIV-1 gp120 directly, preventing interaction with CD4. For multidrug-resistant HIV. Adverse effects: nausea, diarrhea, elevated LFTs.
Capsid Inhibitor
- Lenacapavir: Binds HIV-1 capsid protein → interferes with multiple stages (nuclear import, capsid assembly, uncoating). Given as subcutaneous injection every 6 months — the longest-acting ARV available. For treatment-experienced adults with multidrug-resistant HIV.
Pharmacokinetic Boosters
| Drug | Role | Mechanism |
|---|---|---|
| Ritonavir | Booster for PIs (and nirmatrelvir in COVID-19 Paxlovid) | Potent CYP3A4 inhibitor → raises trough levels of co-administered drug |
| Cobicistat | Booster for PIs, TAF, elvitegravir | Selective CYP3A4 inhibitor; no anti-HIV activity itself |
Long-Acting (LA) Formulations
A major advance aimed at improving adherence:
| Regimen | Route | Frequency | Indication |
|---|---|---|---|
| Cabotegravir + Rilpivirine (Cabenuva) | IM injection | Monthly or every 2 months | HIV treatment (virologically suppressed) |
| Cabotegravir (Apretude) | IM injection | Every 8 weeks | HIV PrEP |
| Lenacapavir | SC injection | Every 6 months | Multidrug-resistant HIV |
LA cabotegravir for PrEP was shown to reduce HIV infection by 68% in men and 89% in women compared to daily oral TDF/FTC in large phase III trials.
Preferred First-Line Regimens (Current Guidelines)
A complete ART regimen typically contains 2 NRTIs + 1 "third agent" (INSTI preferred). Preferred initial regimens include:
- Bictegravir/TAF/FTC (Biktarvy) — single tablet, once daily
- Dolutegravir + TAF/FTC or Dolutegravir/abacavir/3TC (Triumeq, if HLA-B*5701 negative)
- Dolutegravir + 3TC (two-drug regimen, Dovato) — for treatment-naive patients with no HBV co-infection
Drug Interactions — Key Points
- CYP3A4 inducers (rifampin, efavirenz, etravirine, carbamazepine, St. John's Wort): ↓ levels of PIs, NNRTIs, INSTIs → treatment failure
- CYP3A4 inhibitors (ritonavir, cobicistat, azole antifungals, macrolides): ↑ levels of PIs → toxicity
- Antacids/PPIs: ↓ absorption of atazanavir and rilpivirine (chelation/pH effect)
- Metformin: cobicistat inhibits organic cation transporters → ↑ metformin levels
Special Populations
| Situation | Preferred Agents |
|---|---|
| Pregnancy | Dolutegravir (preferred), boosted atazanavir or darunavir; emtricitabine + TDF (backbone) |
| HBV co-infection | Must include TDF or TAF (dual HIV/HBV activity); stopping these can cause severe HBV flare |
| Renal impairment | TAF preferred over TDF (less nephrotoxic); avoid TDF if GFR <30 |
| Multidrug-resistant HIV | Lenacapavir, fostemsavir, ibalizumab (salvage agents) |
Sources: Katzung's Basic and Clinical Pharmacology, 16th Edition, Chapter 49; Goodman & Gilman's Pharmacological Basis of Therapeutics, 14th Edition.
This is a shared conversation. Sign in to Orris to start your own chat.