Lipids biochemistry and metabolism
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Loading Image
Loading Image
I now have comprehensive material from multiple authoritative textbooks. Let me compile the full answer.
Lipids: Biochemistry and Metabolism
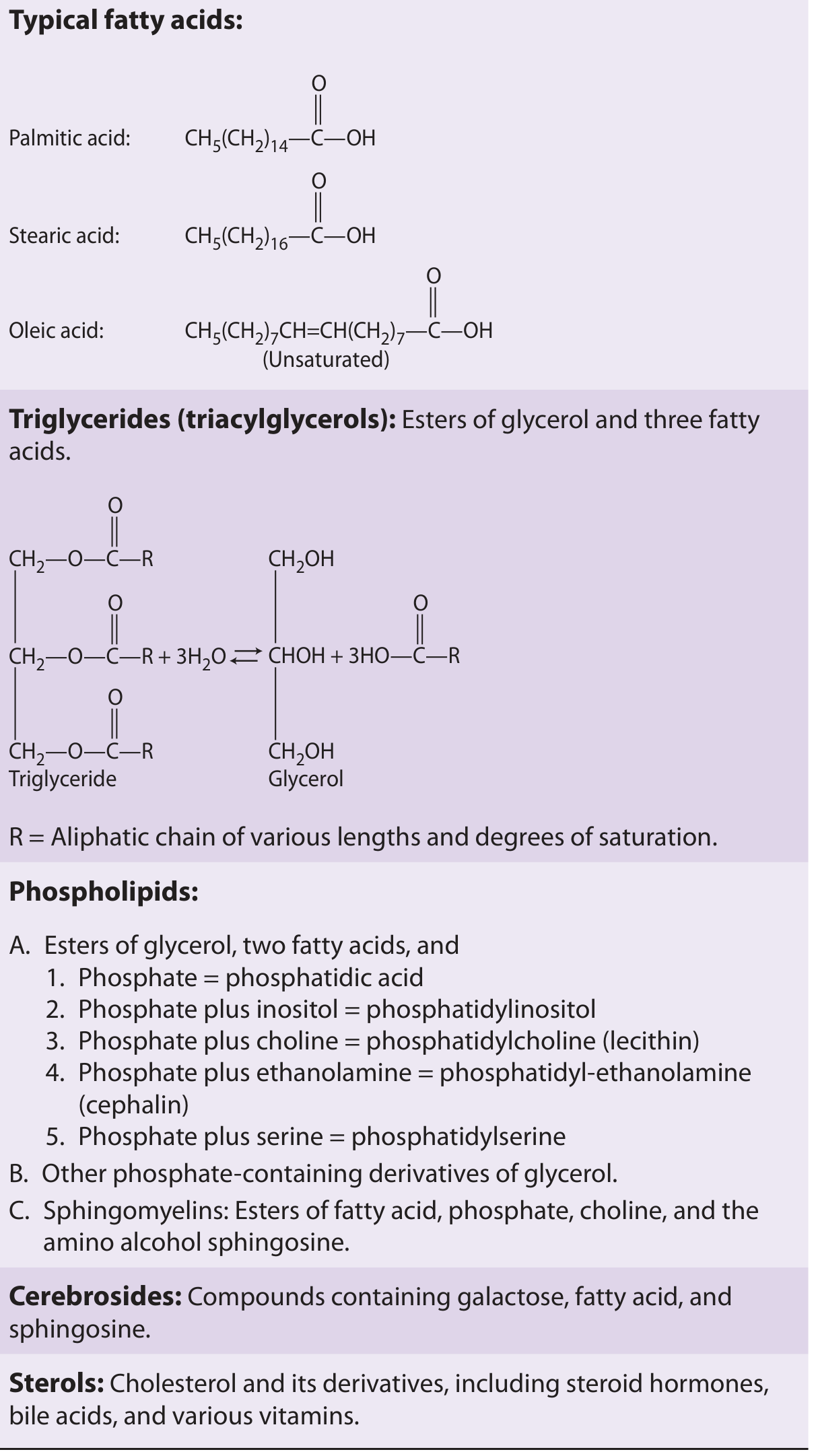
1. Classification of Lipids
Lipids are a chemically diverse group of hydrophobic or amphipathic molecules. The major classes are:
| Class | Structure | Examples |
|---|---|---|
| Fatty acids | Long hydrocarbon chain + carboxyl group | Palmitic (C16:0), Stearic (C18:0), Oleic (C18:1) |
| Triglycerides (triacylglycerols) | Glycerol + 3 fatty acids | Storage fat |
| Phospholipids | Glycerol + 2 FA + phosphate + polar head | Phosphatidylcholine (lecithin), phosphatidylserine |
| Sphingolipids | Sphingosine backbone | Sphingomyelin, cerebrosides |
| Sterols | Steroid nucleus | Cholesterol, steroid hormones, bile acids, vitamin D |
- Saturated fatty acids have no double bonds (e.g., palmitic acid).
- Unsaturated fatty acids have one or more double bonds (e.g., oleic acid has one; linoleic acid has two — an essential fatty acid).
2. Dietary Lipid Digestion & Absorption
Dietary fat is predominantly triglycerides. Key steps:
- Lingual/gastric lipase begins hydrolysis in the stomach.
- Pancreatic lipase (with colipase) cleaves triglycerides to 2-monoglycerides + free fatty acids in the small intestine.
- Bile salts emulsify lipids, forming micelles that allow uptake by enterocytes.
- Inside enterocytes, monoglycerides and fatty acids are resynthesized into triglycerides → packaged into chylomicrons with apolipoprotein B-48.
- Chylomicrons enter the lymphatics (lacteals) → thoracic duct → venous blood at the jugular-subclavian junction.
- Guyton and Hall Textbook of Medical Physiology, p. 842
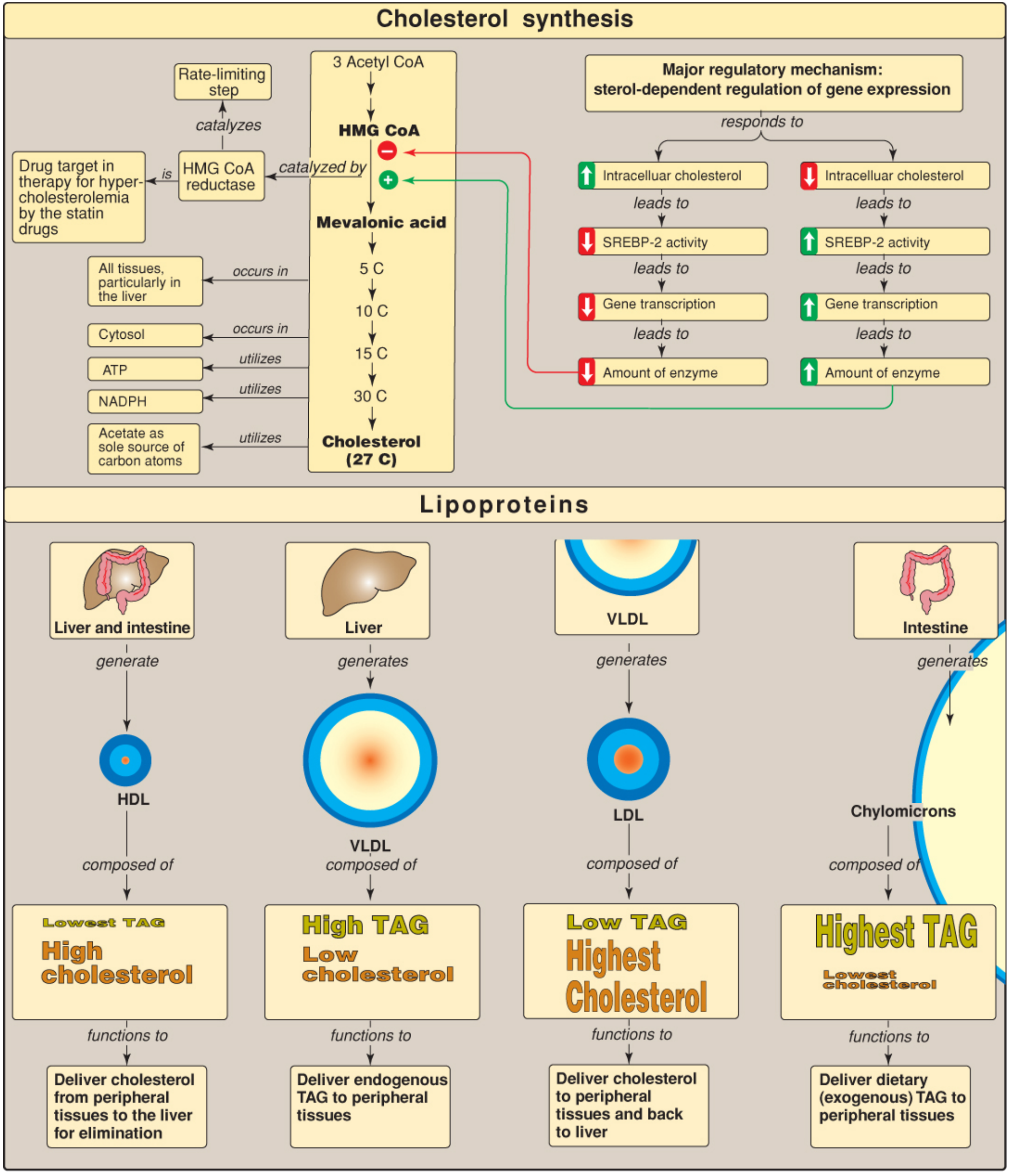
3. Lipid Transport: Lipoproteins
Lipids are insoluble in plasma and are transported as lipoproteins — spherical particles with a hydrophobic core (triglycerides, cholesterol esters) surrounded by an amphipathic shell (phospholipids, free cholesterol, apolipoproteins).
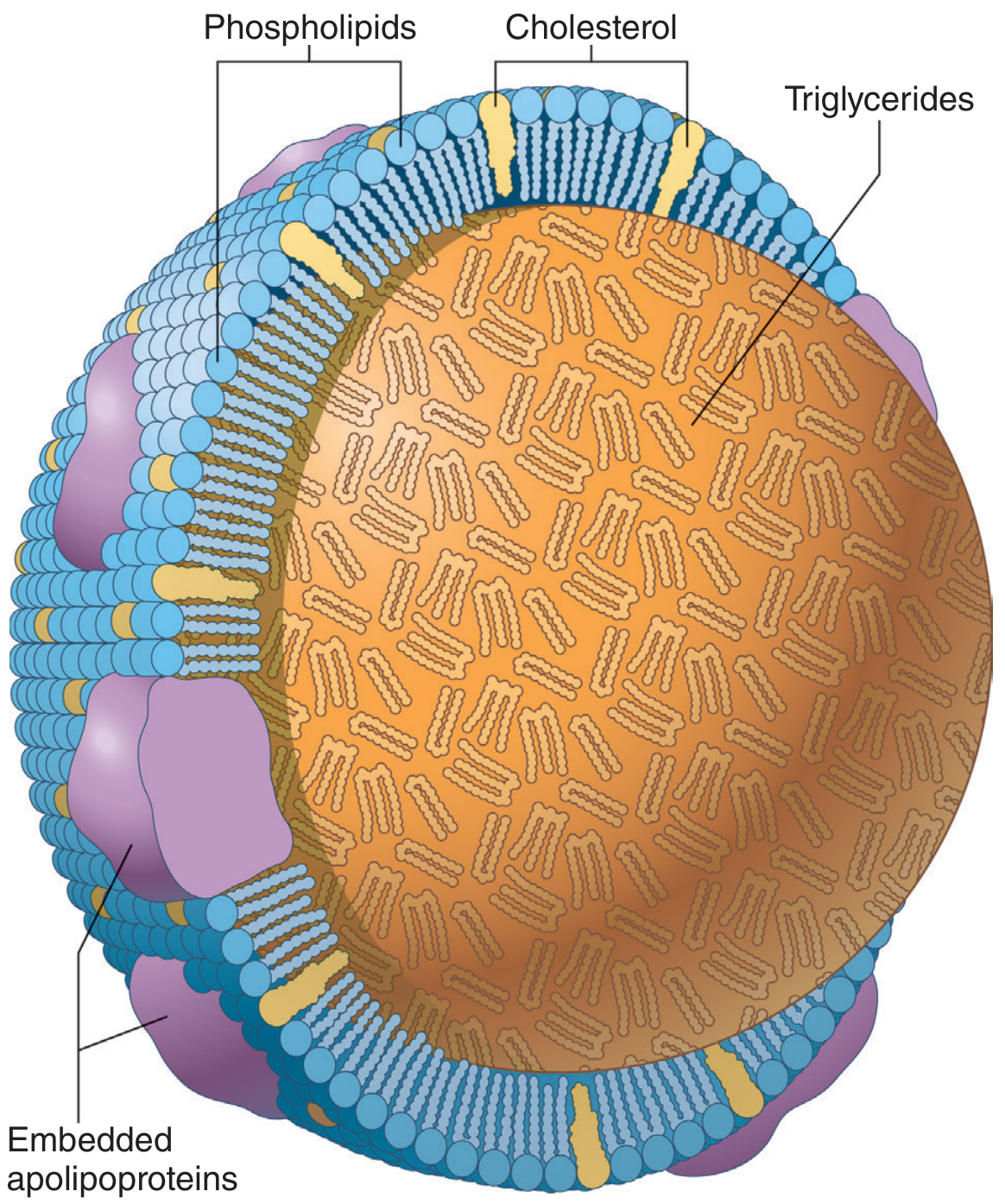
Lipoprotein Classes
| Lipoprotein | Origin | Main Cargo | Key Apolipoprotein | Function |
|---|---|---|---|---|
| Chylomicron | Intestine | Dietary TG (highest) | Apo B-48, Apo C-II, Apo E | Deliver exogenous TG to periphery |
| VLDL | Liver | Endogenous TG (high) | Apo B-100 | Deliver hepatic TG to periphery |
| IDL | From VLDL | TG + cholesterol | Apo B-100, Apo E | Intermediate; cleared by liver or → LDL |
| LDL | From IDL | Cholesterol (highest) | Apo B-100 | Deliver cholesterol to tissues |
| HDL | Liver + intestine | Cholesterol (high protein) | Apo A-I | Reverse cholesterol transport |
- Guyton and Hall Textbook of Medical Physiology, p. 843
- Lippincott's Biochemistry, p. 673
Key Enzymes in Lipoprotein Metabolism
- Lipoprotein lipase (LPL): On capillary endothelium; activated by Apo C-II; hydrolyzes TG in chylomicrons and VLDL → releases fatty acids for storage or energy.
- Hepatic lipase: Converts IDL → LDL.
- LCAT (Lecithin-Cholesterol Acyltransferase): Activated by Apo A-I; esterifies free cholesterol in HDL (core of HDL grows).
- CETP (Cholesterol Ester Transfer Protein): Transfers cholesterol esters from HDL to VLDL/LDL.
Reverse Cholesterol Transport (RCT)
HDL is assembled from lipid-poor Apo A-I secreted by the liver and intestine. Peripheral cells export cholesterol via the ABCA1 transporter → loaded onto HDL → LCAT esterifies it → HDL delivers cholesterol esters to the liver via SR-B1 receptor for elimination as bile acids.
4. Fatty Acid Oxidation (β-Oxidation)
β-Oxidation is the primary pathway for catabolizing fatty acids, occurring in the mitochondrial matrix.
Step 1 — Activation: Fatty acid + CoA → Fatty acyl-CoA (uses ATP; occurs in cytoplasm/outer mitochondrial membrane).
Step 2 — Transport into mitochondria:
- Short- and medium-chain FAs enter freely.
- Long-chain FAs require carnitine shuttle:
- Acyl-CoA + Carnitine → Acylcarnitine (catalyzed by CPT-I on outer membrane)
- Translocase shuttles acylcarnitine across inner membrane
- CPT-II regenerates Acyl-CoA in the matrix
- Carnitine is synthesized from lysine + methionine.
Step 3 — β-Oxidation spiral (repeating 4-step cycle):
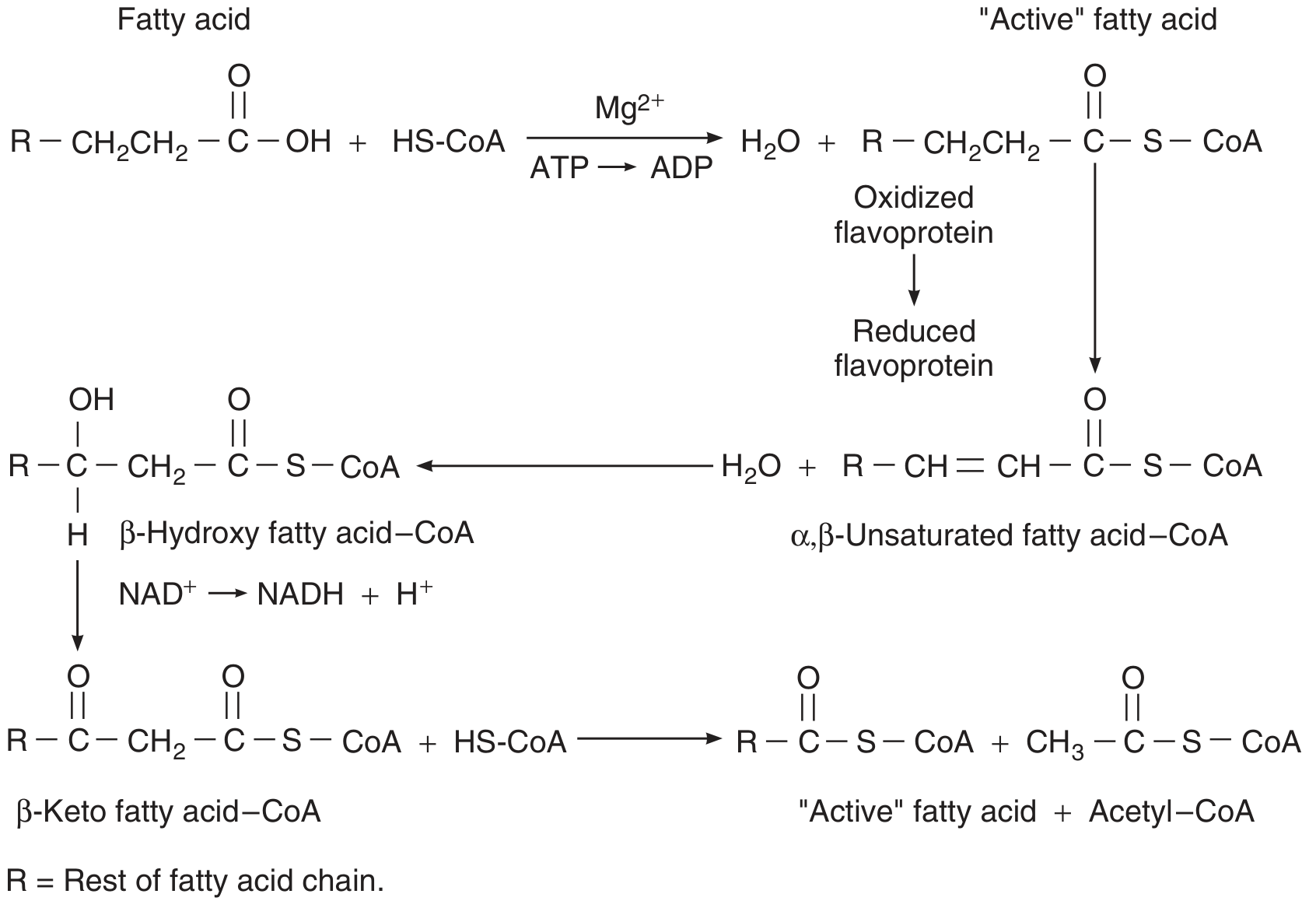
| Step | Reaction | Cofactor |
|---|---|---|
| 1. Oxidation | Acyl-CoA → 2,3-Enoyl-CoA | FAD → FADH₂ |
| 2. Hydration | Enoyl-CoA → 3-Hydroxyacyl-CoA | — |
| 3. Oxidation | 3-Hydroxyacyl-CoA → 3-Ketoacyl-CoA | NAD⁺ → NADH |
| 4. Thiolysis | 3-Ketoacyl-CoA + CoA → Acyl-CoA (–2C) + Acetyl-CoA | — |
Each cycle shortens the chain by 2 carbons, releasing one acetyl-CoA, one FADH₂, and one NADH. Acetyl-CoA enters the TCA cycle.
Energy yield: Catabolism of 1 mol of a 6-carbon fatty acid yields 44 mol ATP vs. 38 mol ATP from glucose — fatty acids are far more energy-dense than carbohydrates.
- Ganong's Review of Medical Physiology, p. 37
5. Ketone Body Formation & Metabolism
When acetyl-CoA production exceeds TCA cycle capacity (starvation, diabetes, high-fat diet), the liver diverts acetyl-CoA to ketone body synthesis:
Pathway (in liver mitochondria):
- 2 Acetyl-CoA → Acetoacetyl-CoA
- Acetoacetyl-CoA + Acetyl-CoA → HMG-CoA (mitochondrial HMG-CoA synthase)
- HMG-CoA → Acetoacetate + Acetyl-CoA (HMG-CoA lyase)
- Acetoacetate → β-Hydroxybutyrate (reversible) or Acetone (irreversible, volatile)
Ketone bodies (acetoacetate, β-hydroxybutyrate, acetone) are exported to peripheral tissues (brain, heart, muscle), where they are converted back to acetyl-CoA → TCA cycle.
Ketoacidosis occurs when ketone body production exceeds peripheral utilization — pH falls, characteristic acetone breath develops. Triggered by: DKA, starvation, alcoholism.
- Guyton and Hall Textbook of Medical Physiology, p. 846
6. Fatty Acid Synthesis (De Novo Lipogenesis)
Fatty acid synthesis is the reverse of β-oxidation in direction but uses a different set of enzymes, location, and cofactors.
| Feature | β-Oxidation | De Novo Synthesis |
|---|---|---|
| Location | Mitochondria | Cytoplasm |
| Carrier | CoA | ACP (Acyl Carrier Protein) |
| Reducing agent | FAD, NAD⁺ (oxidized) | NADPH (consumed) |
| Key enzyme | Thiolase | Fatty Acid Synthase (FAS) |
| Key intermediate | Acetyl-CoA | Malonyl-CoA |
Key steps:
- Acetyl-CoA (mitochondrial) → exported to cytoplasm as citrate via the citrate shuttle
- Acetyl-CoA + CO₂ → Malonyl-CoA (catalyzed by Acetyl-CoA Carboxylase, ACC; requires biotin) — the committed, rate-limiting step
- Fatty Acid Synthase (FAS) elongates the chain by 2 carbons per cycle using malonyl-CoA as donor
- NADPH is supplied by the pentose phosphate pathway (HMP shunt)
- The primary product is Palmitate (C16:0); further elongation/desaturation occurs in the ER
Regulation:
- Insulin activates ACC (stimulates lipogenesis)
- Glucagon/epinephrine inactivate ACC via PKA phosphorylation
- AMPK phosphorylates and inactivates ACC when energy is low
- Malonyl-CoA inhibits CPT-I, preventing simultaneous synthesis and oxidation
7. Cholesterol Biosynthesis
All 27 carbons of cholesterol come from acetyl-CoA. Synthesis occurs primarily in the liver, also in the intestine, adrenal cortex, and gonads. The pathway is cytoplasmic (ER).
4 Stages:
Stage 1: Acetyl-CoA → Mevalonate
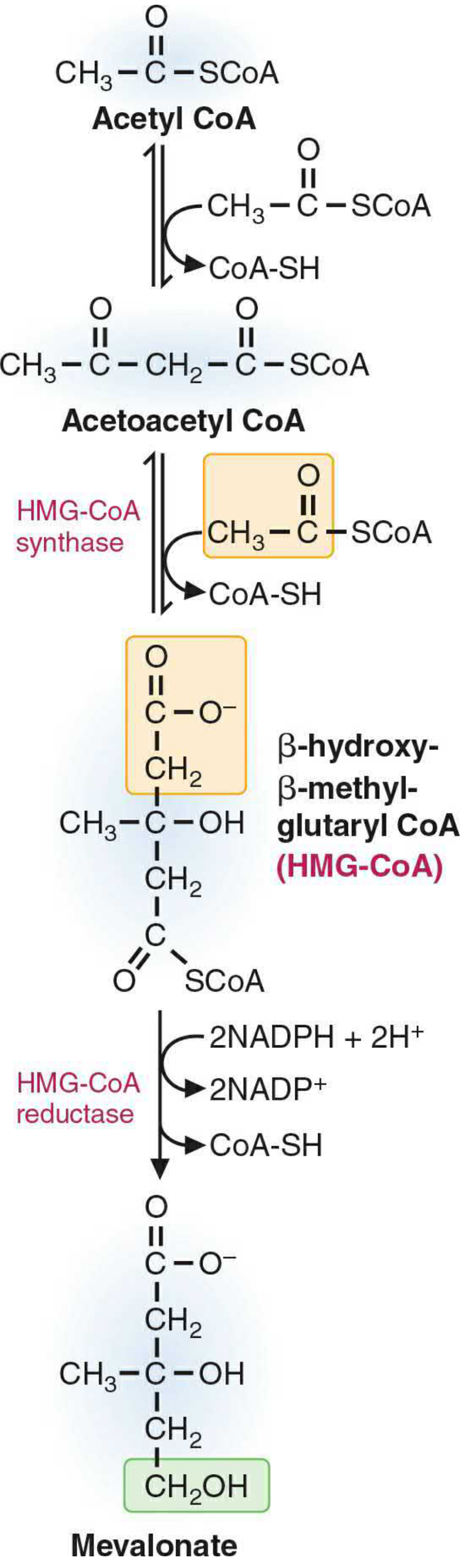
- 2 Acetyl-CoA → Acetoacetyl-CoA → HMG-CoA (cytosolic HMG-CoA synthase)
- HMG-CoA + 2 NADPH → Mevalonate (HMG-CoA reductase) ← rate-limiting step; target of statins
Stage 2: Mevalonate → Activated isoprene (IPP)
- 3 ATP phosphorylate mevalonate → decarboxylation → isopentenyl pyrophosphate (IPP, Δ³-isopentenyl-PP)
Stage 3: IPP → Squalene (C30)
- 6 isoprene units condense (via geranyl-PP, farnesyl-PP) → Squalene (requires NADPH)
Stage 4: Squalene → Cholesterol (C27)
- Squalene cyclizes → lanosterol → multiple steps → cholesterol
Regulation of HMG-CoA Reductase
- SREBP-2 (Sterol Regulatory Element-Binding Protein 2): When intracellular cholesterol is low, SREBP-2 is activated → increases HMG-CoA reductase gene transcription.
- Accelerated protein degradation: High cholesterol promotes enzyme degradation.
- AMPK phosphorylation: Inactivates the enzyme when energy is low.
- Insulin activates; glucagon inhibits.
- Statins: Competitive inhibitors of HMG-CoA reductase → reduce hepatic cholesterol → upregulate LDL receptors → lower plasma LDL.
- Lippincott's Biochemistry, p. 672; Basic Medical Biochemistry 6e
8. Cholesterol Utilization & Elimination
Cholesterol cannot be fully catabolized in humans — its ring structure is intact. It is eliminated by:
- Bile acid synthesis: Rate-limiting step = cholesterol-7α-hydroxylase (inhibited by bile acids — feedback)
- Steroid hormone synthesis: Cholesterol → Pregnenolone (P450scc, rate-limiting) → cortisol, aldosterone, sex hormones
- Vitamin D synthesis: Skin + liver + kidney
- Direct secretion into bile (risk: gallstones / cholelithiasis if bile salts insufficient)
Enterohepatic circulation: >95% of bile acids are reabsorbed in the terminal ileum and returned to the liver. Bile acid sequestrants (cholestyramine) interrupt this cycle.
9. Regulation Summary: Fed vs. Fasted State
| State | Dominant Pathway | Key Hormones |
|---|---|---|
| Fed / Insulin ↑ | FA synthesis, TG storage, cholesterol synthesis | Insulin activates ACC, FAS, HMG-CoA reductase |
| Fasted / Glucagon ↑ | β-Oxidation, ketogenesis, lipolysis | Glucagon/epinephrine activate HSL, inhibit ACC |
| Starvation / DM | Maximal β-oxidation + ketogenesis | Cortisol ↑, glucagon ↑ → FFA release from adipose |
Hormone-sensitive lipase (HSL) in adipocytes is activated by glucagon/epinephrine via cAMP-PKA → hydrolyzes stored TG → releases FFA bound to albumin for transport to other tissues.
10. Clinical Correlations
| Condition | Defect | Consequence |
|---|---|---|
| Familial hypercholesterolemia (FH) | Defective LDL receptor | ↑ LDL-C; premature atherosclerosis |
| Type I hyperlipoproteinemia | LPL or Apo C-II deficiency | Massive chylomicronemia; pancreatitis |
| Type III hyperlipoproteinemia | Apo E2/E2 homozygosity | IDL & chylomicron remnant accumulation |
| Diabetic ketoacidosis | No insulin → no glucose utilization | Massive ketone body production → acidosis |
| MCAD deficiency | Medium-chain acyl-CoA dehydrogenase deficiency | β-Oxidation block → hypoglycemia, hypoketosis |
| Abetalipoproteinemia | No Apo B synthesis | Cannot form chylomicrons/VLDL → fat malabsorption |
| Atherosclerosis | LDL oxidation + macrophage uptake | Foam cells → plaques |
Sources: Guyton and Hall Textbook of Medical Physiology (Chap. 69); Ganong's Review of Medical Physiology, 26e; Lippincott Illustrated Reviews: Biochemistry, 8e; Basic Medical Biochemistry: A Clinical Approach, 6e
This is a shared conversation. Sign in to Orris to start your own chat.