Post prandial alkaline tide mbbs 1st year physiology exam notes
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
parietal cell HCl secretion alkaline tide mechanism diagram
I now have comprehensive information from multiple authoritative textbooks. Let me compile the complete MBBS 1st year physiology exam notes on the postprandial alkaline tide.
Postprandial Alkaline Tide — MBBS 1st Year Physiology Notes
Definition
The alkaline tide (postprandial alkaline tide) refers to the transient rise in blood pH and plasma bicarbonate (HCO₃⁻) — and a corresponding rise in urine pH — that occurs after a meal, as a direct consequence of gastric parietal cell acid secretion.
"For each H₃O⁺ ion secreted into the gastric lumen, a HCO₃⁻ ion is released from the basal side of the epithelium into the circulation, a process that has been named the 'alkaline tide.'" — Yamada's Textbook of Gastroenterology
Cellular Mechanism — Step by Step
The mechanism occurs within gastric parietal cells (oxyntic cells):
Key membrane equipment
| Membrane | Transporters/Enzymes |
|---|---|
| Apical (luminal) | H⁺-K⁺ ATPase (proton pump), Cl⁻ channels |
| Basolateral (blood-side) | Cl⁻-HCO₃⁻ exchanger (AE2/SLC4A2), Na⁺-K⁺ ATPase |
| Intracellular | Carbonic anhydrase |
Step-by-step sequence
Step 1 — CO₂ hydration:
CO₂ (from aerobic metabolism) + H₂O → H₂CO₃ (catalyzed by carbonic anhydrase)
H₂CO₃ → H⁺ + HCO₃⁻
Step 2 — H⁺ secretion into stomach lumen (apical side):
H⁺ is actively pumped into the stomach lumen via H⁺-K⁺ ATPase (proton pump) — against a massive concentration gradient (intracellular pH ~7.4 vs luminal pH ~0.8, a gradient of ~4 million-fold).
Cl⁻ follows via apical Cl⁻ channels → net HCl secretion into lumen.
Step 3 — HCO₃⁻ absorption into blood (basolateral side):
HCO₃⁻ exits the parietal cell into the bloodstream via the Cl⁻-HCO₃⁻ exchanger (AE2) on the basolateral membrane.
→ This raises plasma HCO₃⁻ → alkaline tide in gastric venous blood
Step 4 — Net result:
- Stomach lumen: net secretion of HCl (acidification to pH 1–2)
- Blood (gastric venous): net absorption of HCO₃⁻ → transient metabolic alkalosis
"The absorbed HCO₃⁻ is responsible for the 'alkaline tide' (high pH) that can be observed in gastric venous blood after a meal." — Costanzo Physiology, 7th Ed.
Diagram of Parietal Cell Mechanism
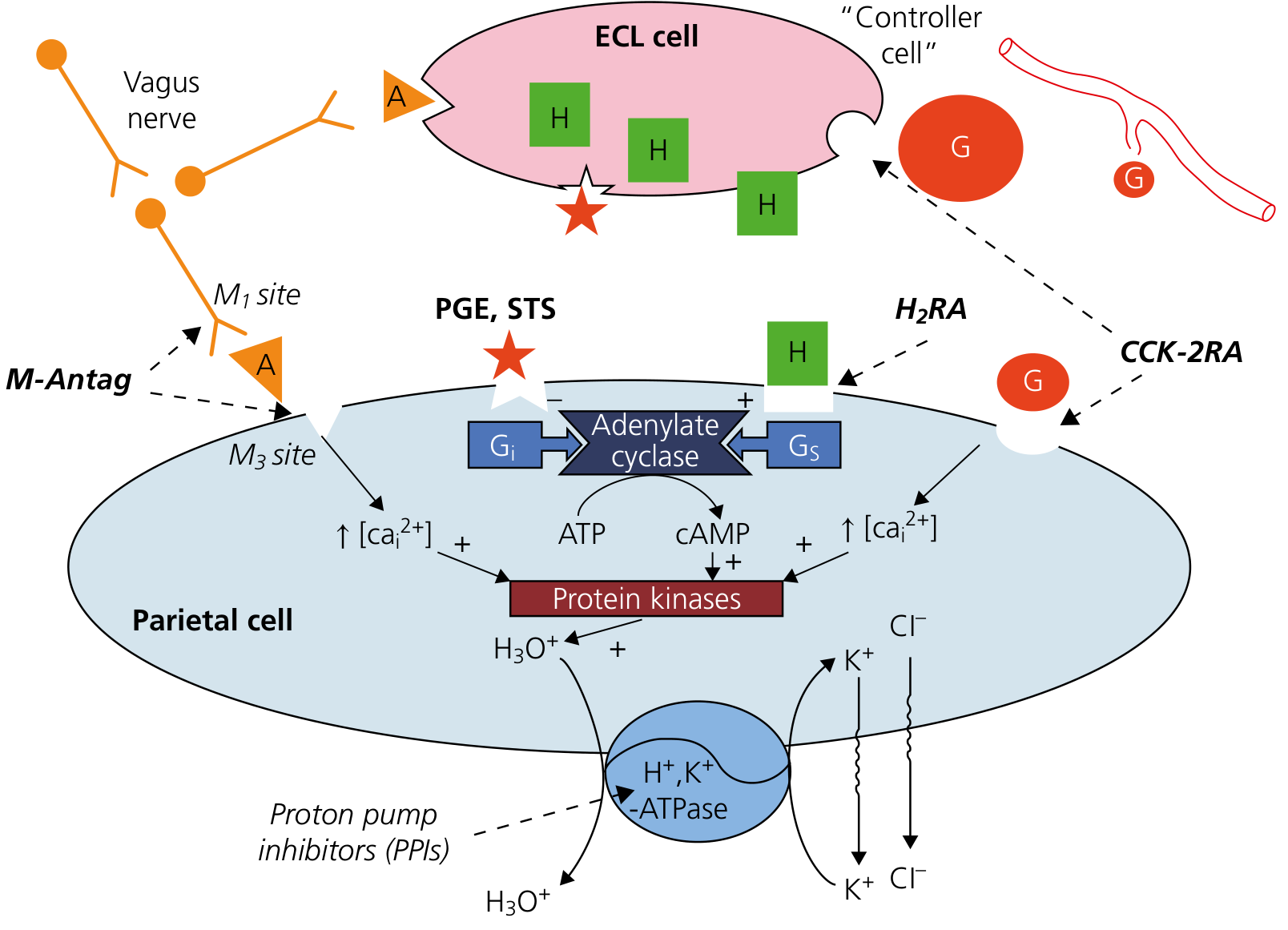
Parietal cell secretion pathways — gastrin (G cell), histamine (ECL cell), and acetylcholine (vagus) all converge on protein kinases to activate H⁺,K⁺-ATPase. — Yamada's Textbook of Gastroenterology
Chemical Equation Summary
CO₂ + H₂O →(carbonic anhydrase)→ H₂CO₃ → H⁺ + HCO₃⁻
Apical: H⁺ → [H⁺-K⁺ ATPase] → Gastric lumen (secreted as HCl)
Basolateral: HCO₃⁻ → [Cl⁻/HCO₃⁻ exchanger] → Blood (= alkaline tide)
Manifestations of the Alkaline Tide
| Site | Change | Mechanism |
|---|---|---|
| Gastric venous blood | ↑ pH, ↑ plasma HCO₃⁻ | HCO₃⁻ absorbed from parietal cells |
| Arterial blood | Mild transient metabolic alkalosis | HCO₃⁻ distributes systemically |
| Urine | ↑ pH (becomes alkaline after meals) | Excess HCO₃⁻ filtered and excreted by kidneys |
| pCO₂ | Slight ↑ | Respiratory compensation for alkalosis |
"Venous blood leaving the stomach is enriched with bicarbonates, and this phenomenon is responsible for postprandial metabolic alkalosis (i.e., the alkaline tide) with concomitant increase of pCO₂..." — Tietz Textbook of Laboratory Medicine
Urinary Alkaline Tide
- Urine pH rises after meals (typically from ~5–6 in the morning to ~7–8 postprandially).
- Kidneys excrete excess filtered HCO₃⁻ to restore acid-base balance.
- This is the basis of the historically observed "alkaline urine after meals."
"An alkaline pH often is noted after meals, when an 'alkaline tide' to balance gastric acid excretion increases urine pH." — Goldman-Cecil Medicine
Fate of the Alkaline Tide HCO₃⁻
The HCO₃⁻ released into the blood is not "lost" — it is eventually recycled back into the GI tract:
- Taken up by pancreatic ductal cells → secreted as pancreatic bicarbonate into the duodenum (to neutralize gastric acid entering the duodenum)
- Some is also taken up by gastric surface epithelial cells to aid mucosal protection
"Eventually this HCO₃⁻ will be secreted back into the gastrointestinal tract in pancreatic secretions." — Costanzo Physiology
Stimulants of Gastric Acid Secretion (= Triggers of Alkaline Tide)
Three main secretagogues stimulate parietal cells to secrete HCl (and thus generate the alkaline tide):
| Secretagogue | Type | Receptor on Parietal Cell | Second Messenger |
|---|---|---|---|
| Histamine | Paracrine (from ECL cells) | H₂ receptor | cAMP → PKA |
| Acetylcholine (ACh) | Neurocrine (vagus nerve) | M₃ muscarinic receptor | IP₃/Ca²⁺ → PKC |
| Gastrin | Hormone (from G cells, antrum) | CCK-2 (gastrin) receptor | IP₃/Ca²⁺ → PKC |
All three pathways converge to activate H⁺-K⁺ ATPase and stimulate HCl secretion.
Phases of Gastric Secretion (Meal Context)
The alkaline tide is most prominent during:
| Phase | Trigger | % of Total Secretion |
|---|---|---|
| Cephalic | Sight/smell/taste of food → vagus | ~30% |
| Gastric | Food in stomach → distension + gastrin | ~60% — peak alkaline tide |
| Intestinal | Chyme in duodenum | ~10% |
Clinical Correlations (Exam High-Yield)
| Condition | Relevance to Alkaline Tide |
|---|---|
| Vomiting | Loss of HCl → no HCO₃⁻ generated → metabolic alkalosis (hypochloremic, hypokalemic) — the "alkaline tide" analogy reversed |
| PPI use (omeprazole) | Blocks H⁺-K⁺ ATPase → ↓ HCl secretion → ↓ alkaline tide |
| H₂ blockers (cimetidine, ranitidine) | Block H₂ receptors → ↓ histamine-stimulated HCl → ↓ alkaline tide |
| Peptic ulcer disease | Excess HCl secretion → exaggerated alkaline tide |
| Zollinger-Ellison syndrome | Gastrin-secreting tumour → massive HCl and thus massive alkaline tide + metabolic alkalosis |
| Urine pH testing | Postprandial alkaline urine (pH 7–8) is normal; misinterpreting it as pathological alkalosis is an error |
Key Points to Remember for Exams
- Alkaline tide = HCO₃⁻ entering blood for every H⁺ secreted into the stomach.
- The exchanger involved is Cl⁻/HCO₃⁻ exchanger (AE2) on the basolateral membrane of parietal cells.
- The enzyme carbonic anhydrase is essential — it generates H⁺ and HCO₃⁻ from CO₂ + H₂O inside the parietal cell.
- The proton pump is H⁺-K⁺ ATPase — inhibited by PPIs (omeprazole, pantoprazole).
- The alkaline tide produces transient postprandial metabolic alkalosis — detected in gastric venous blood and urine.
- HCO₃⁻ from the alkaline tide is eventually recycled as pancreatic bicarbonate to neutralize duodenal acid.
- The alkaline tide is physiologically normal — not pathological.
Sources:
- Costanzo Physiology, 7th Ed., p. 366
- Yamada's Textbook of Gastroenterology, 7th Ed.
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease
- Tietz Textbook of Laboratory Medicine, 7th Ed.
- Goldman-Cecil Medicine
- Henry's Clinical Diagnosis and Management by Laboratory Methods
Short note
Postprandial Alkaline Tide — Short Note
Definition
A transient rise in blood and urine pH occurring after a meal, due to absorption of HCO₃⁻ into the blood as a by-product of gastric HCl secretion by parietal cells.
Mechanism
Inside the parietal cell:
CO₂ + H₂O →(carbonic anhydrase)→ H⁺ + HCO₃⁻
| Direction | Event |
|---|---|
| Apical → Lumen | H⁺ pumped out via H⁺-K⁺ ATPase + Cl⁻ → forms HCl |
| Basolateral → Blood | HCO₃⁻ exchanged for Cl⁻ via Cl⁻/HCO₃⁻ exchanger → alkaline tide |
Effects
- Gastric venous blood — ↑ HCO₃⁻, ↑ pH (transient metabolic alkalosis)
- Urine — becomes alkaline (pH 7–8) after meals
- pCO₂ — slightly raised (respiratory compensation)
Fate of HCO₃⁻
Recycled back into the gut via pancreatic ductal secretions to neutralize acid chyme in the duodenum.
Clinical Points
| Drug/Condition | Effect |
|---|---|
| PPIs (omeprazole) | Block H⁺-K⁺ ATPase → ↓ alkaline tide |
| H₂ blockers (cimetidine) | ↓ histamine-stimulated HCl → ↓ alkaline tide |
| Persistent vomiting | Loss of HCl → hypochloremic metabolic alkalosis |
| Zollinger-Ellison syndrome | Excess gastrin → exaggerated alkaline tide |
Key fact: For every 1 H⁺ secreted into the stomach, 1 HCO₃⁻ enters the blood — this 1:1 stoichiometry is the core of the alkaline tide.
This is a shared conversation. Sign in to Orris to start your own chat.