Hey lets study wilms tumor
Reading File
Finding Sources
Searching PubMed
"Wilms tumor"[MeSH Terms] AND ("nephroblastoma"[MeSH Terms] OR treatment[MeSH Terms])
Reading File
Reading File
Reading File
Searching Images
Wilms tumor nephroblastoma histology gross pathology
Reading File
Reading File
Loading Image
Loading Image
I have everything needed for a comprehensive study guide. Here it is:
Wilms Tumor (Nephroblastoma) — Complete Study Guide
1. Overview
Wilms tumor (nephroblastoma) is the most common primary renal malignancy in children and the third most common solid pediatric cancer overall. It accounts for ~85% of pediatric renal tumors, with ~500 new cases/year in the US. Peak incidence is ages 2–5, and the overall cure rate approaches 90% even with metastatic disease — one of the great success stories in pediatric oncology.
2. Associated Syndromes & Genetics
| Syndrome | Features | Genetic Locus | Risk of WT |
|---|---|---|---|
| WAGR | Wilms, Aniridia, Genital anomalies, mental Retardation | WT1 deletion (11p13) + PAX6 | ~33% |
| Denys-Drash (DDS) | Gonadal dysgenesis, early nephropathy | WT1 dominant-negative mutation (11p13) | ~90% |
| Beckwith-Wiedemann (BWS) | Macroglossia, macrosomia, visceromegaly, hemihypertrophy, neonatal hypoglycemia | WT2 locus (11p15.5) — loss of IGF2 imprinting | 4–10% |
| Li-Fraumeni | Predisposition to multiple cancers | TP53 germline | Increased |
| Neurofibromatosis | Café-au-lait spots, neurofibromas | NF1 | Increased |
Key molecular points:
- WT1 (11p13): transcription factor critical for renal and gonadal development. In WAGR → deletion; in DDS → dominant-negative point mutation
- WT2/IGF2 (11p15.5): IGF-2 is normally expressed from paternal allele; loss of maternal imprinting → overexpression → tumorigenesis (BWS)
- Sporadic tumors (90% of cases): mutations in β-catenin (~10%), or genes encoding microRNA processing proteins (15–20%) — leads to failure of mesenchymal-to-epithelial transformation and persistent blastemal rests
- TP53 mutations → anaplastic histology → especially poor prognosis
3. Gross Pathology
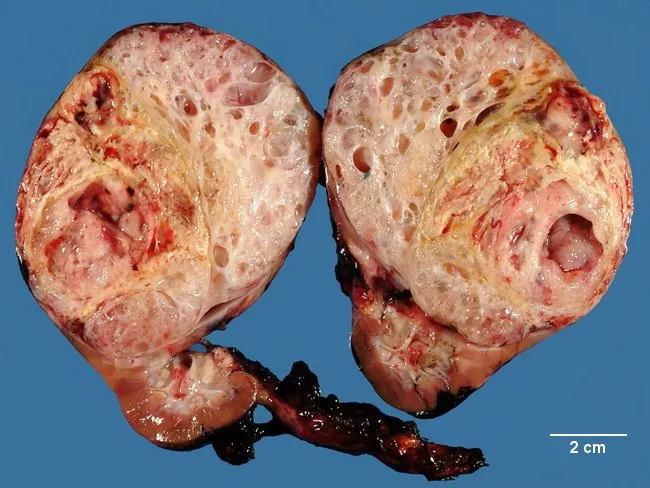
- Large, solitary, well-circumscribed mass replacing renal parenchyma
- Soft, homogeneous, tan to gray on cut section
- Foci of hemorrhage, cystic degeneration, and necrosis common
- 10% are bilateral or multicentric at diagnosis
- 13% bilateral in some series
4. Histology — Classic Triphasic Pattern
The hallmark is recapitulation of developing nephrons:
| Component | Appearance |
|---|---|
| Blastemal | Sheets of small, primitive blue cells — aggressive, associated with advanced disease |
| Epithelial | Abortive tubules or glomeruli |
| Stromal | Spindle cells, sometimes with skeletal muscle differentiation |
All three components must be present for favorable histology.
Histologic classification:
- Favorable histology (FH): classic triphasic OR epithelial-predominant (less aggressive, often stage I)
- Unfavorable histology (UFH): anaplasia (focal or diffuse), clear cell sarcoma, rhabdoid tumor
- Anaplastic WT: TP53 mutated, chemoresistant, high recurrence risk
- Blastemal-predominant: clinically aggressive, associated with advanced stage
Nephrogenic rests: precursor lesions found in 25–40% of kidneys with WT. They can differentiate and spontaneously regress — they do NOT have independent oncologic potential but mark increased risk.
5. Clinical Presentation
- Asymptomatic abdominal/flank mass — usually noticed by parents while bathing/dressing (most common presentation)
- Hypertension in ~25% (secondary to renin-angiotensin axis disruption)
- Hematuria (gross or microscopic)
- Vague abdominal discomfort
- Rarely: varicocele, hepatomegaly (hepatic vein obstruction), ascites, heart failure, obstipation, weight loss
- Sometimes discovered after blunt abdominal trauma
6. Imaging & Workup
| Modality | Role |
|---|---|
| Abdominal + chest CT | First-line — characterizes mass, staging, contralateral kidney, regional nodes, metastases |
| Abdominal ultrasound | Evaluate renal vein/IVC tumor thrombus |
| CT | Detects nephrogenic rests, assesses for bilateral disease |
| Lung mets | Present in ~8% at diagnosis |
7. Staging — NWTSG System
| Stage | Definition |
|---|---|
| I | Tumor limited to kidney, completely excised, capsule intact, no rupture/biopsy |
| II | Extends through capsule but completely removed; no residual at margins; vessels outside kidney may contain tumor; or local spillage limited to tumor bed |
| III | Residual non-hematogenous tumor confined to abdomen (positive nodes, peritoneal implants, incomplete resection, tumor spillage) |
| IV | Hematogenous metastases (lungs, liver, bone, brain) |
| V | Bilateral renal involvement |
8. Treatment
Two major protocols exist — differences are clinically important:
| NWTSG/COG (North American) | SIOP (European) | |
|---|---|---|
| Approach | Surgery → chemotherapy | Preoperative chemo → surgery |
| Advantage | Preserves staging info; avoids overtreatment | Tumor shrinkage, easier resection |
| Disadvantage | Larger initial surgery | Loss of pre-treatment staging data |
| Outcome | Overall survival equivalent |
Both groups agree on preoperative chemotherapy when:
- Bilateral disease (Stage V)
- IVC involvement above the hepatic veins
- Involvement of a solitary kidney
Surgery
- Radical nephrectomy is standard
- Explore contralateral kidney
- Lymph node sampling mandatory for staging
Chemotherapy
- Stage I/II FH: Vincristine + Actinomycin D (2-drug)
- Stage III/IV FH: Vincristine + Actinomycin D + Doxorubicin (3-drug)
- Anaplastic/UFH: Vincristine + Actinomycin D + Doxorubicin ± Cyclophosphamide + Etoposide
Radiation
- Stage III and IV with FH → whole abdomen/flank radiation
- All diffuse anaplastic stages → radiation
- Pulmonary mets: whole-lung radiation if present
9. Prognosis
| Stage | 4-year OS (FH) |
|---|---|
| I | >95% |
| II | ~90% |
| III | ~85% |
| IV | ~75–80% |
| Anaplastic/UFH | Significantly worse (~55–70% depending on stage) |
Key prognostic factors: stage, histology (favorable vs. unfavorable), blastemal predominance, TP53 status, and response to therapy.
10. High-Yield Summary for Exams
- Most common renal tumor in children (peak 2–5 years)
- Always think WT1 (11p13) with aniridia → WAGR
- BWS → IGF-2 overexpression → WT2 locus (11p15)
- Triphasic histology: blastemal + epithelial + stromal
- Anaplasia = unfavorable, TP53, chemoresistant
- Presents as painless abdominal mass; BP ↑ in 25%
- Do NOT rupture the tumor during surgery — causes upstaging to III
- Bilateral = Stage V → preoperative chemo, nephron-sparing surgery goal
- Overall cure ~90% — paradigm for multimodal pediatric oncology
Recent Evidence
Two 2025-2026 systematic reviews are worth noting:
- PMID 41459843 (2025): Compared SIOP vs. NWTSG/COG protocols — confirms equivalent survival outcomes between the two approaches
- PMID 41219039 (2026): Minimally invasive vs. open nephrectomy under SIOP guidelines — MIS appears feasible in selected patients, though long-term data are still emerging
Sources: Robbins & Kumar Basic Pathology; Robbins, Cotran & Kumar Pathologic Basis of Disease; Schwartz's Principles of Surgery 11e; Sabiston Textbook of Surgery; Campbell-Walsh-Wein Urology
Lets study mitral stenosis
Finding Sources
Searching PubMed
"mitral stenosis"[MeSH Terms] AND management[MeSH Terms]
Reading File
Reading File
Searching Images
mitral stenosis echocardiography pathology rheumatic heart disease
Reading File
Reading File
Loading Image
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
I have a comprehensive picture of mitral stenosis. Let me now compile the complete study guide.
Mitral Stenosis — Complete Study Guide
1. Overview & Epidemiology
Mitral stenosis (MS) is obstruction of the mitral valve orifice during diastole, impairing LV filling. The normal mitral valve area (MVA) is 4–6 cm²; gradients appear when MVA falls below 2 cm², and symptoms emerge at ≤1.5 cm².
- Most common cause worldwide: Rheumatic fever (rheumatic heart disease)
- Occurs in 40% of all rheumatic heart disease patients
- 2:1 female predominance for isolated MS
- ~60% of pure MS patients recall a prior episode of rheumatic fever
- In developed countries, MS is now rare and mostly seen in elderly foreign-born women, often post-commissurotomy
Rare non-rheumatic causes: Congenital anomalies, chest radiation, mucopolysaccharidosis, severe mitral annular calcification (MAC), ball-valve thrombus, left atrial myxoma, cor triatriatum (membrane divides LA — mimics MS)
2. Pathology & Pathophysiology
Structural Changes (Rheumatic)
- Leaflet thickening and nodularity
- Commissural fusion (anterior + posterior leaflets fuse at edges)
- Subvalvular involvement — chordal thickening, fusion, and shortening
- Calcification of leaflets
- Classic appearance: "fish-mouth" orifice (short axis); "hockey stick" deformity of anterior leaflet (long axis — doming due to restricted tip mobility with pliable body)
Hemodynamic Cascade
MVA↓ → LA pressure↑ → LA dilation → AF
↓
Pulmonary venous HTN → dyspnea, pulmonary edema, hemoptysis
↓
Pulmonary arterial HTN (chronic) → RV failure, TR
- Symptoms begin when MVA ≤1.5 cm² (mean gradient 5–10 mmHg)
- Severe MS = MVA <1.0 cm², gradient >10 mmHg
- AF develops from LA dilation → loss of atrial kick → acute symptom worsening + thromboembolic risk
- LA thrombus (especially LAA) → systemic embolism, stroke
- Death primarily from heart failure or systemic embolism
3. Clinical Presentation
Symptoms
- Dyspnea on exertion (most common) → orthopnea, PND, pulmonary edema
- Hemoptysis (pulmonary venous hypertension → rupture of bronchopulmonary anastomoses)
- Palpitations (atrial fibrillation)
- Systemic embolism / stroke
- Fatigue, exercise intolerance
- Hoarseness — Ortner's syndrome (LA compression of left recurrent laryngeal nerve)
- Right heart failure symptoms in advanced disease (edema, ascites)
Physical Examination
| Finding | Mechanism |
|---|---|
| Loud S1 | Mitral leaflets wide open at onset of systole (due to high LA pressure), then snap shut |
| Opening snap (OS) | Sudden tensing of leaflets at end of opening — heard after S2 |
| Short S2–OS interval | Reflects severity — the higher the LA pressure, the shorter the S2–OS gap; <70 ms = severe MS |
| Low-pitched rumbling mid-diastolic murmur | Heard best at apex with bell, in left lateral decubitus |
| Pre-systolic accentuation | From atrial contraction — absent in AF |
| Malar flush (mitral facies) | Peripheral cyanosis from low cardiac output |
| Signs of PH | Loud P2, RV heave, TR murmur |
In elderly patients with calcified valves: S1 may be soft, OS may be absent, murmur may be inaudible — echocardiography is essential.
4. Investigations
ECG
- P mitrale (bifid P wave in II, negative component in V1) — LA enlargement
- Atrial fibrillation — common in advanced disease
- RV hypertrophy pattern if pulmonary hypertension develops
Chest X-Ray
- LA enlargement — double density at right heart border, elevation of left main bronchus, splaying of carina
- Pulmonary venous congestion — upper lobe diversion, Kerley B lines
- Mitral valve calcification (lateral CXR)
- Straightening of left heart border (LAA prominence)
Echocardiography — Gold Standard
Echocardiographic image of rheumatic MS:
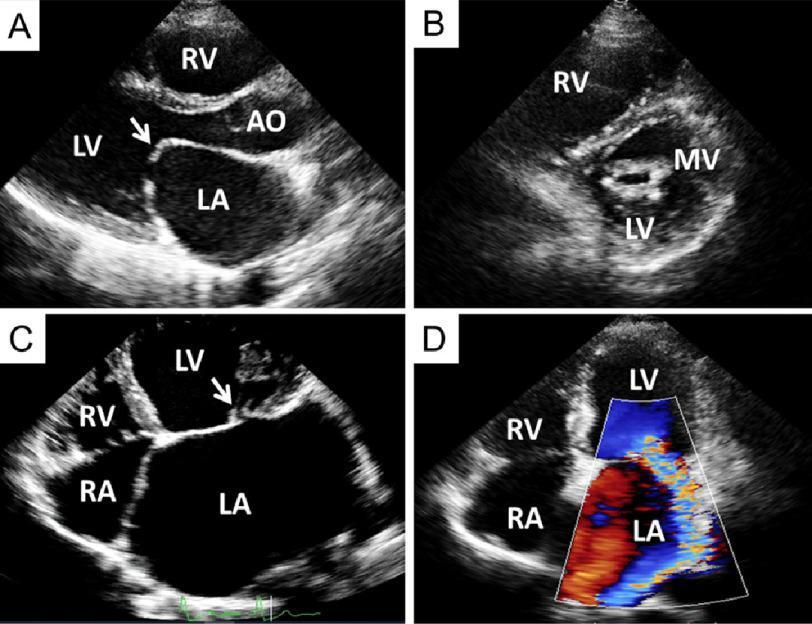
Key 2D findings:
- Leaflet thickening, calcification, restricted mobility
- Commissural fusion
- "Hockey stick" / "doming" AMVL on parasternal long axis
- "Fish-mouth" orifice on short axis
- LA enlargement, LA thrombus assessment
Severity quantification:
| Severity | MVA (cm²) | Mean Gradient (mmHg) | PHT (ms) |
|---|---|---|---|
| Mild | >1.5 | <5 | <100 |
| Moderate | 1.0–1.5 | 5–10 | 100–220 |
| Severe | <1.0 | >10 | >220 |
MVA calculation methods:
- Planimetry — direct tracing of orifice in PSAX (most reliable; 3D echo = highest accuracy)
- Pressure Half-Time (PHT): MVA = 220 ÷ T½ (T½ = DT × 0.29)
- Continuity equation: MVA = (LVOT TVI × LVOT area) ÷ MV TVI — used when PHT is unreliable (e.g., after valvuloplasty, significant AR, abnormal LV compliance)
TEE: Mandatory before balloon valvuloplasty to exclude LA thrombus and assess MR severity
5. Wilkins Echocardiographic Score
Used to determine suitability for percutaneous mitral balloon valvuloplasty (PMBV). Each of 4 parameters scored 1–4:
| Parameter | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| Leaflet mobility | Highly mobile, restricted only at tips | Mid and base portion reduced mobility | Valve moves forward only at base | No forward movement |
| Leaflet thickening | Near-normal (4–5 mm) | Mid-leaflets normal, tips thickened (5–8 mm) | Entire leaflet thickened (5–8 mm) | Marked thickening (>8–10 mm) |
| Calcification | Single area of brightness | Scattered areas at margins | Brightness extends to mid-leaflet | Extensive brightness all tissue |
| Subvalvular thickening | Minimal | Chordal thickening up to 1/3 length | Thickening to distal third | Extensive to papillary muscles |
Total score: 0–16
- Score ≤8: Favorable for PMBV — excellent outcomes
- Score >8: Increasing risk of suboptimal result, restenosis, and complications
- Score >12: Generally unsuitable for PMBV; consider surgery
French 3-group classification also used:
- Group 1: Pliable, noncalcified AMVL + mild subvalvular disease
- Group 2: Pliable AMVL + severe subvalvular disease
- Group 3: Any calcification (fluoroscopy) — worst outcomes with PMBV
6. Treatment
Medical Management
| Drug | Indication |
|---|---|
| Diuretics | Pulmonary congestion, volume overload |
| Beta-blockers / rate-limiting CCBs (diltiazem, verapamil) | Heart rate control (especially AF) — prolong diastole → more time for LV filling |
| Digoxin | Rate control in persistent AF |
| Anticoagulation (warfarin, target INR 2–3) | AF; prior embolism; LA thrombus; MS + severe LA enlargement |
| Antibiotics | Rheumatic fever prophylaxis (penicillin) |
| Avoid vasodilators | Can drop cardiac output precipitously |
Note: DOACs are not approved for rheumatic MS — warfarin remains standard.
Percutaneous Mitral Balloon Valvuloplasty (PMBV) — Inoue Technique
Mechanism: Balloon catheter inflated across fused commissures → separates them → increases MVA
Indications (AHA/ACC):
- Symptomatic MS with MVA ≤1.5 cm², favorable morphology (Wilkins ≤8), no LA thrombus, MR <moderate (Class I)
- Asymptomatic severe MS + new-onset AF (Class IIb, after excluding LA thrombus)
- Symptoms with mild MS (MVA >1.5 cm²) if exercise testing shows significant obstruction (Class IIb)
- High surgical risk patients with calcified valves — PMBV as palliation
Access: Transseptal (transfemoral venous → transseptal puncture → LA)
Contraindications: LA thrombus, MR ≥ moderate, Wilkins >12, heavy valve calcification (especially commissural)
Complications: Cardiac tamponade (~1–5%), severe MR (~2–10%), systemic embolism (~1–3%), death (~1–3%); success rates <50% in elderly
Surgical Options
| Procedure | Indication |
|---|---|
| Open mitral commissurotomy | Favorable anatomy, not suitable for PMBV (e.g., LA thrombus removable at surgery) |
| Mitral valve replacement (MVR) | Calcified, fibrotic, heavily distorted valve; significant MR coexists; failed PMBV; Wilkins >12 |
- Mechanical valves: Better durability; require lifelong anticoagulation
- Bioprosthetic valves: Avoid anticoagulation (if sinus rhythm); wear out in 10–15 years
7. MS in Special Populations
MS in Pregnancy
- Hemodynamic stress of pregnancy (↑HR, ↑CO, ↑blood volume) → acute decompensation
- Symptoms may worsen precipitously in 2nd trimester
- Management: beta-blockers for rate control; diuretics cautiously; PMBV in 2nd trimester if refractory; avoid MVR if possible (fetal risks of cardiopulmonary bypass + anticoagulation)
MS in the Elderly
- Often calcific rather than rheumatic; loud S1 and OS may be absent
- Higher procedural risk with both PMBV and MVR (periop mortality ≥10% for MVR)
- AF + superimposed age-related LA changes → higher embolic risk
8. Complications
| Complication | Mechanism |
|---|---|
| Atrial fibrillation | LA dilation → remodeling |
| Systemic thromboembolism | Stasis in LA/LAA, especially in AF |
| Pulmonary hypertension | Chronic elevation of pulmonary venous pressure |
| Right heart failure + TR | Pulmonary HTN → RV pressure overload |
| Pulmonary edema | Acute increase in LA pressure (e.g., tachycardia, pregnancy, AF with rapid ventricular rate) |
| Hemoptysis | Pulmonary venous HTN → bronchopulmonary vein rupture |
| Infective endocarditis | Less common than with regurgitant lesions |
9. High-Yield Summary
- Cause: Rheumatic fever → commissural fusion → "fish-mouth" valve
- Symptoms begin at MVA ≤1.5 cm²; severe at <1.0 cm²
- Key auscultation: Loud S1 → A2 → OS → rumbling diastolic murmur at apex; short S2–OS = severe; pre-systolic accentuation lost in AF
- Echo: Hockey-stick AMVL, fish-mouth orifice, MVA by planimetry or PHT (220/T½)
- Wilkins score ≤8 = favorable for PMBV; >8 = worse outcomes
- Rate control in AF is critical — tachycardia reduces diastolic filling time, precipitates pulmonary edema
- Anticoagulation = warfarin (not DOACs) for AF or prior embolism
- PMBV is first-line intervention for favorable-anatomy symptomatic severe MS
- Avoid vasodilators — can cause catastrophic CO drop
- Pregnancy + MS = high-risk combination requiring close monitoring
Sources: Braunwald's Heart Disease; Textbook of Clinical Echocardiography; Grainger & Allison's Diagnostic Radiology; Braunwald's Heart Disease (Geriatric Cardiology chapter); Creasy & Resnik's Maternal-Fetal Medicine
This is a shared conversation. Sign in to Orris to start your own chat.