Cushing’s syndrome
Reading File
Reading File
Finding Sources
Searching PubMed
"Cushing syndrome" diagnosis treatment
Running Command
Reading File
Searching Images
Cushing syndrome clinical features moon face buffalo hump
Running Command
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Loading Image
Loading Image
Reading File
Running Command
Running Command
Running Command
Running Command
I now have comprehensive content from Nelson 2024 (Chapter 619), Harrison's 22E (2025), Sabiston Surgery, and Campbell-Walsh Urology. I have all the information needed for a thorough response. Let me now compose the comprehensive answer.
Cushing's Syndrome
Cushing's syndrome is the clinical state resulting from chronic exposure to excess glucocorticoids, whether from exogenous administration or endogenous overproduction.
Epidemiology & Etiology
Endogenous Cushing's syndrome is rare, affecting 5–10 individuals per million per year. Causes are classified as:
| Category | Cause | Frequency |
|---|---|---|
| Exogenous | Iatrogenic glucocorticoid use | Most common overall |
| ACTH-dependent | Cushing disease (pituitary corticotroph microadenoma) | ~70–75% of endogenous |
| ACTH-dependent | Ectopic ACTH syndrome (neuroendocrine tumors, bronchogenic carcinoma) | ~10% of endogenous |
| ACTH-dependent | Ectopic CRH syndrome | <1% |
| ACTH-independent | Adrenal adenoma | ~90% of primary adrenal |
| ACTH-independent | Adrenocortical carcinoma (ACC) | <10% of primary adrenal |
| ACTH-independent | Bilateral macro/micronodular hyperplasia (PPNAD, BMAH) | Rare |
Cushing disease (the pituitary subtype) accounts for ~80% of ACTH-dependent disease and affects females up to two-thirds of the time. - Campbell-Walsh Urology
Pathophysiology
The zona fasciculata of the adrenal cortex normally secretes ~20 mg cortisol/day under tight HPA axis regulation. Cortisol follows a circadian rhythm — peak ~1 hour after waking, nadir ~11 PM. This rhythm is invariably lost in Cushing's.
Excess glucocorticoids cause:
- Upregulation of gluconeogenesis, lipolysis, and protein catabolism
- Excess cortisol overwhelms renal 11β-HSD2, allowing mineralocorticoid effects → hypertension, hypokalemia, edema
- Suppression of gonadotropins → hypogonadism/amenorrhea
- Suppression of the hypothalamic-pituitary-thyroid axis → ↓ TSH
ACTH excess (in pituitary/ectopic disease) also drives POMC-derived peptides → hyperpigmentation at skin folds and scars. - Harrison's 22E, 2025
Clinical Features
Most features are non-specific individually; the diagnosis should be considered when multiple features co-exist, especially in a young patient.
Classic Signs & Symptoms
Highly discriminatory (more specific):
- Broad (>1 cm), violaceous/purplish striae (see image below)
- Proximal myopathy — difficulty rising from a chair without using hands
- Skin thinning with easy bruising
- Pathologic fractures (osteoporosis) at young age
Common but less specific:
- Central/truncal obesity with thin limbs
- Moon facies (rounded, plethoric face)
- Dorsocervical fat pad ("buffalo hump")
- Supraclavicular fat pads
- Hypertension (present in >70% of cases)
- Hyperglycemia / type 2 diabetes
- Hirsutism, acne
- Hypogonadism, menstrual irregularities
In ectopic ACTH syndrome specifically:
- Hyperpigmentation of knuckles, scars, pressure areas (from excess ACTH/POMC products)
- More rapid onset: edema, severe hypokalemia, hypertension
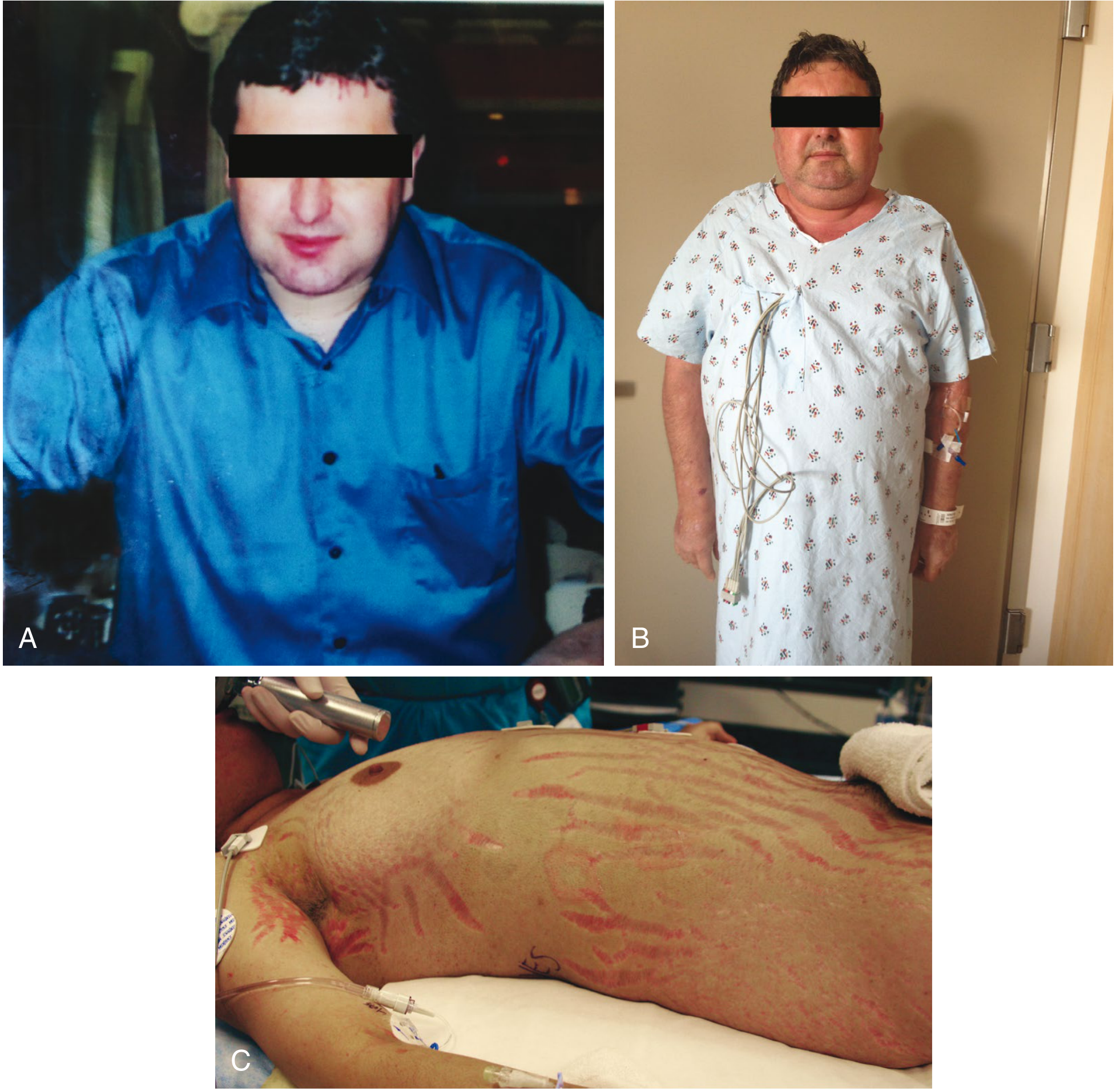
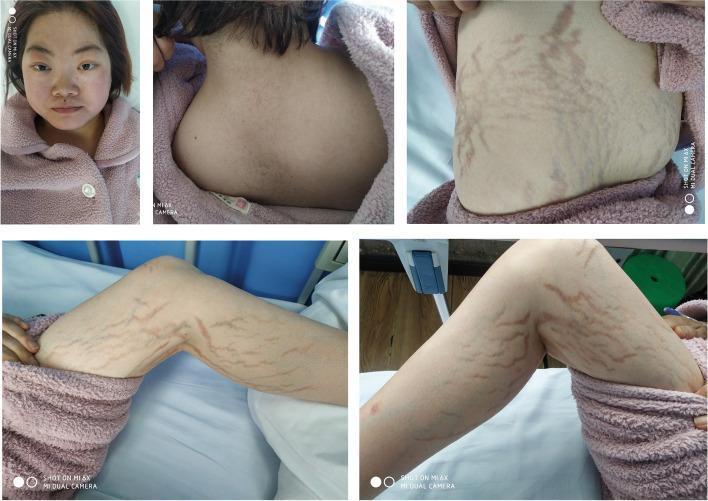
Serious complications:
- Deep vein thrombosis and pulmonary embolism (hypercoagulable state)
- Psychiatric symptoms: anxiety, depression, psychosis
- Increased cardiovascular mortality (5× excess) if untreated
- Opportunistic/fungal infections
Pediatric Features (Nelson 2024 — Chapter 619)
| System | Manifestations |
|---|---|
| Dermatologic | Facial plethora, acne, easy bruising, moon facies, hirsutism, violaceous striae (unusual <7 yr) |
| Growth | Growth deceleration + weight gain; short stature (key differentiator from simple obesity) |
| Cardiovascular | Hypertension, coagulopathy |
| Gonadal | Amenorrhea, virilization, gynecomastia |
| Neurologic | Headaches |
| Metabolic | Impaired glucose tolerance, diabetes, nephrolithiasis, fractures |
| Psychologic | Depression, anxiety, irritability, fatigue |
Key pediatric differentiator: Children with simple obesity are tall, while those with Cushing's are short with a decelerating growth rate. - Nelson 2024, Ch. 619
Diagnosis
The most critical first step is establishing the diagnosis before any differential testing to avoid erroneous imaging or surgery.
Step 1 — Confirm Hypercortisolism (Biochemical Diagnosis)
Three first-line tests (perform ≥2):
| Test | Detail |
|---|---|
| 24-hour urinary free cortisol (UFC) | Collect at least twice; unequivocally elevated (>3× ULN) establishes diagnosis |
| Late-night salivary cortisol | Cortisol nadir disruption; >550 ng/dL has sensitivity 93%, specificity 100%. If UFC is 1–3× normal, perform two salivary measurements |
| 1-mg overnight dexamethasone suppression test (DST) | Administer 1 mg dexamethasone at 11 PM; morning cortisol <1.8 μg/dL = normal suppression |
| Midnight plasma/serum cortisol | >4.4 μg/dL strongly suggests Cushing's (Nelson 2024) |
Always exclude exogenous glucocorticoid use first.
Pseudo-Cushing states (elevated cortisol without true Cushing's — must exclude):
- Pregnancy, severe depression, alcohol dependence, morbid obesity, poorly controlled diabetes, glucocorticoid resistance
Step 2 — Differential Diagnosis: ACTH-Dependent vs. Independent
Measure plasma ACTH (two measurements):
| ACTH Level | Interpretation |
|---|---|
| ≤5 pg/mL (undetectable) | ACTH-independent → adrenal imaging (CT) |
| Normal/elevated | ACTH-dependent → pituitary MRI + further testing |
| Very high | Suggests ectopic ACTH syndrome |
Step 3 — Localize the Source
If ACTH-independent: CT of adrenals (unenhanced — Hounsfield units distinguish benign from malignant)
If ACTH-dependent:
- Pituitary MRI (gadolinium T1): up to 40–50% of corticotroph microadenomas are MRI-negative
- High-dose dexamethasone suppression test (HDDST): 2 mg q6h × 48h — pituitary adenomas often suppress; ectopic tumors typically do not (but ~10–15% of ectopic tumors may also suppress → test has limitations)
- Bilateral inferior petrosal sinus sampling (BIPSS) with CRH: the gold standard for Cushing disease vs. ectopic ACTH
- Central/peripheral ACTH ratio >2 at baseline or >3 after CRH = Cushing disease
- No gradient → ectopic ACTH source; proceed to chest/abdomen CT, octreotide scan, or ⁶⁸Ga-DOTATATE PET/CT
Pediatric ACTH cutoffs (Nelson 2024):
- <10 pg/mL → ACTH-independent
- 10–20 pg/mL → CRH test to differentiate
-
20 pg/mL → ACTH-dependent
Diagnostic Algorithm
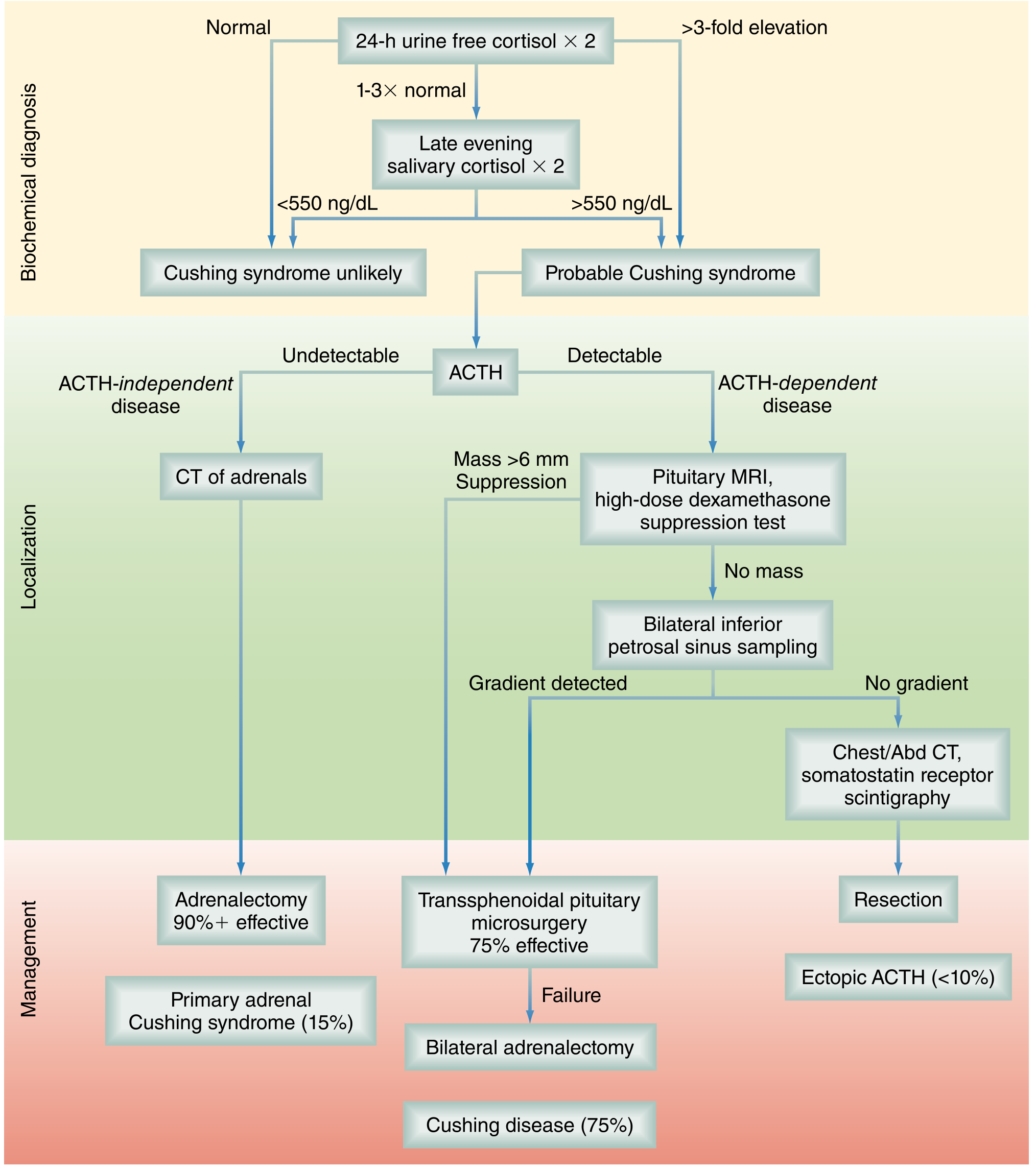
Treatment
Treatment is cause-specific and multidisciplinary (endocrinology, neurosurgery, adrenal surgery).
Cushing Disease (Pituitary Source)
- First-line: Transsphenoidal pituitary microsurgery (endoscopic approach)
- Initial cure rate 60–80% with experienced surgeon; lower for invasive/macroadenomas
- Postoperative subnormal cortisol on day 1–2 = indicator of cure
- Long-term follow-up essential — relapse occurs in a significant proportion
- Recurrence options: Re-operation, fractionated radiotherapy, stereotactic radiosurgery, pharmacologic therapy, or bilateral adrenalectomy
ACTH-Independent (Adrenal Source)
- Adrenalectomy (laparoscopic for benign; open for suspected malignancy)
- Cure rate >90% for solitary adenoma
- Perioperative stress-dose steroids: hydrocortisone 100 mg IV q8h × 24h
- HPA axis recovery may take >1 year — glucocorticoid replacement needed until recovery
Ectopic ACTH Syndrome
- Resect the primary tumor when localized
- If tumor cannot be located: bilateral adrenalectomy or medical therapy
Medical/Pharmacologic Therapy
Used preoperatively to reduce cortisol rapidly, for unresectable/metastatic disease, or as bridging therapy:
| Drug | Mechanism |
|---|---|
| Metyrapone | Inhibits 11β-hydroxylase (CYP11B1) — reduces cortisol synthesis |
| Ketoconazole / Levoketoconazole | Inhibits multiple steroidogenic enzymes |
| Osilodrostat | Cortisol synthesis inhibitor (FDA-approved) |
| Mitotane | Adrenolytic; cytotoxic to adrenal cortex (FDA-approved) |
| Mifepristone | Glucocorticoid receptor antagonist |
| Pasireotide | Somatostatin analog; inhibits ACTH secretion (approved for persistent Cushing disease after surgery) |
| Aminoglutethimide / Etomidate | Reserved for acute/severe cases |
Special Considerations
- Nelson syndrome: After bilateral adrenalectomy for Cushing disease, the unresected pituitary adenoma may enlarge dramatically → marked hyperpigmentation; occurs in ~25% of adults. - Nelson 2024
- Postoperative replacement: Hydrocortisone 10 mg/m²/24h in three divided doses until HPA axis recovery
- Pediatric catch-up: After treatment, substantial catch-up growth and pubertal progress occur, but adult height is often compromised and bone density remains abnormal
Genetics (Nelson 2024 — Pediatric Perspective)
| Subtype | Key Genes |
|---|---|
| Cushing disease | USP8, MEN1, CDKIs, CDKN1B/p27Kip1, AIP, DICER1 |
| PPNAD / Carney complex | PRKAR1A (chr 17q22-24), PDE8B, PDE11A, PRKACA |
| BMAH | ARMC5 (~40%), GNAS1, MEN1, FH |
| Adrenal adenoma | PRKACA (somatic), CTNNB1, MC2R |
| Ectopic ACTH | RET, MEN1 |
| McCune-Albright | GNAS (somatic, activating) |
Recent Evidence (PubMed, 2024–2026)
- A 2024 systematic review (PMID 39730909) confirmed that prolactin-adjusted BIPSS improves discrimination of Cushing disease from ectopic ACTH syndrome.
- A 2024 meta-analysis (PMID 39086899) evaluated surgical vs. conservative management of mild autonomous cortisol secretion, finding surgical benefit particularly for metabolic outcomes.
- A 2024 systematic review on PPNAD (PMID 39006359) characterized pathogenic variants in primary pigmented nodular adrenocortical disease across 210 patients.
Sources: Nelson Textbook of Pediatrics 2024 (Ch. 619) | Harrison's Principles of Internal Medicine 22E, 2025 | Sabiston Textbook of Surgery | Campbell-Walsh Urology
This is a shared conversation. Sign in to Orris to start your own chat.