Interferon alpha beta gamma their function and where they are used as treatment
Reading File
Finding Sources
Searching the Web
interferon alpha beta gamma functions clinical uses treatment
Searching PubMed
interferon alpha beta gamma therapy clinical
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Loading Image
I now have all the information needed for a comprehensive answer. Here is the full breakdown:
Interferons (IFN-α, IFN-β, IFN-γ): Functions and Clinical Uses
Overview
Interferons were first described by Isaacs and Lindemann as potent factors that "interfere with" viral replication. They are the body's first active defense against viral infection — an early warning system. Beyond antiviral activity, they activate the immune response, enhance T-cell recognition of infected cells, and block cell proliferation. They are classified into three main types:
| Property | IFN-α | IFN-β | IFN-γ |
|---|---|---|---|
| Classification | Type I | Type I | Type II |
| Previous name | Leukocyte IFN | Fibroblast IFN | Immune IFN |
| Number of genes | >20 | 1 | 1 |
| Principal source | Epithelium, leukocytes, pDCs | Fibroblasts | NK cells, T cells (ILC1) |
| Primary activator | Viruses (dsRNA) | Viruses (dsRNA) | Immune response |
| Acid stability | Stable | Stable | Labile |
| Homology with IFN-α | 100% | 30–50% | <10% |
— Medical Microbiology 9e, Table 10.2
Interferon Alpha (IFN-α) — Type I
Functions
- Produced rapidly (within hours) in response to double-stranded RNA (dsRNA), the replication intermediate of RNA viruses, and by interaction of enveloped viruses (HSV, HIV) with plasmacytoid dendritic cells (pDCs)
- Binds the IFNAR1/IFNAR2 heterodimeric receptor → activates JAK-STAT signaling → induces the antiviral state in neighboring cells
- The antiviral state involves two key enzymes:
- 2',5'-oligoadenylate synthetase → activates RNase L → degrades viral mRNA
- Protein kinase R (PKR) → phosphorylates eIF-2α → inhibits ribosome assembly and protein synthesis
- Upregulates MHC class I expression → enhances antigen presentation → makes infected cells better targets for cytotoxic T lymphocytes (CTLs)
- Activates NK cells to kill virally infected cells
- Triggers systemic flu-like symptoms (malaise, myalgia, chills, fever) — especially during viremia
Clinical Uses (Therapeutic)
| Condition | Agent |
|---|---|
| Chronic Hepatitis B | IFN-α (subcutaneous) |
| Chronic Hepatitis C | Pegylated IFN-α-2a or 2b + ribavirin (now largely superseded by DAAs) |
| Hairy cell leukemia | IFN-α-2b |
| Melanoma (adjuvant) | High-dose IFN-α-2b |
| Follicular lymphoma / Cutaneous T-cell lymphoma (CTCL) | IFN-α |
| Kaposi's sarcoma | IFN-α |
| Renal cell carcinoma | IFN-α (adjuvant/combination) |
| Condyloma acuminata (genital warts / HPV) | Intralesional IFN-α |
| Essential thrombocythemia / Polycythemia vera | IFN-α (pegylated) |
IFN-α was one of the first cytokines introduced into clinical oncology and remains relevant in adjuvant regimens and pegylated long-acting forms. — Sabiston Textbook of Surgery
Mechanism of Action Diagram (IFN-α / IFN-β)
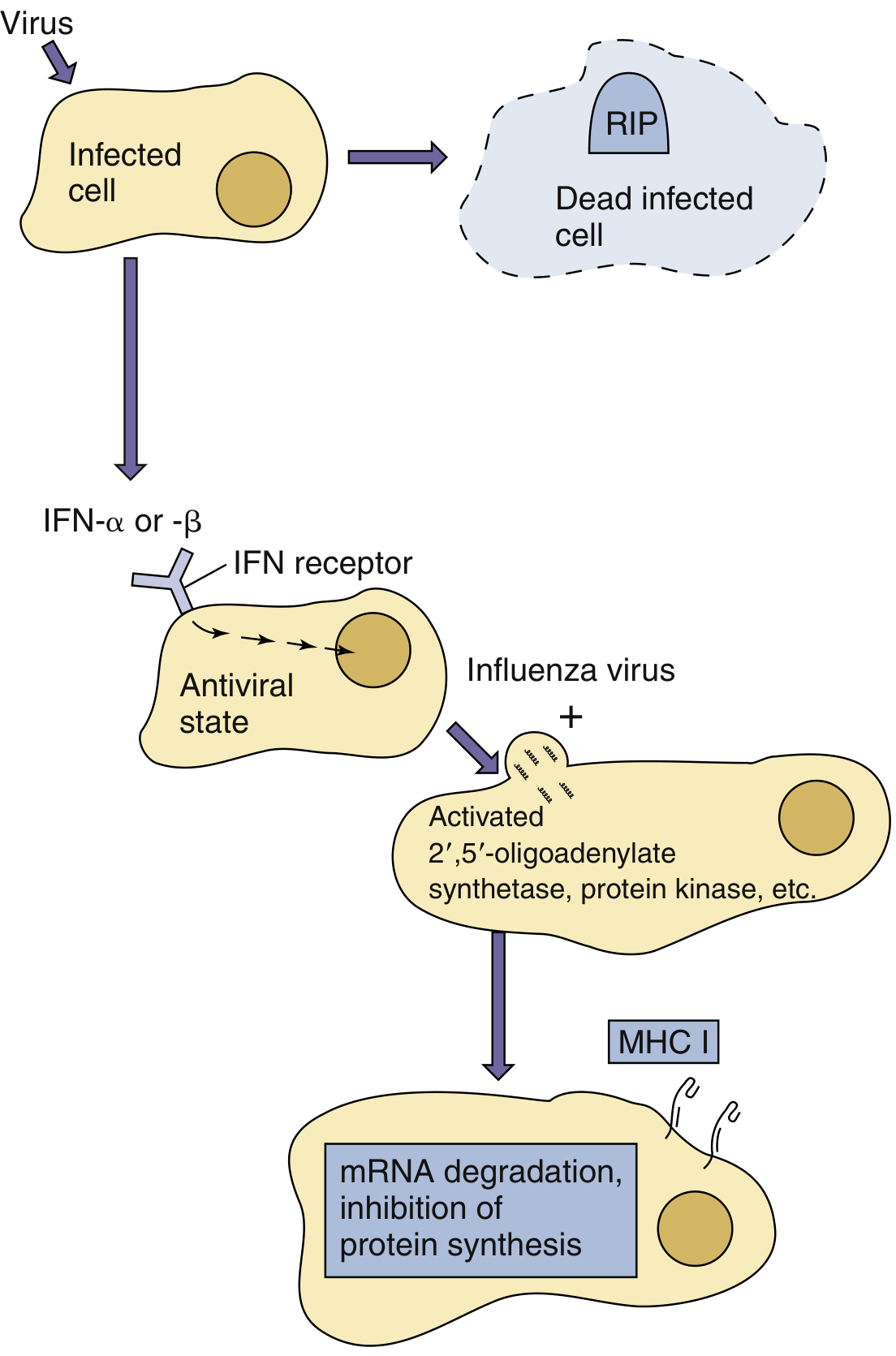
Viral infection triggers release of IFN-α/β, which binds IFN receptors on neighboring cells, induces antiviral enzymes (2',5'-oligoadenylate synthetase, PKR), and upon viral infection of those cells, leads to mRNA degradation and inhibition of protein synthesis. MHC I expression is also upregulated.
— Medical Microbiology 9e, Fig. 10.4
Interferon Beta (IFN-β) — Type I
Functions
- Produced mainly by fibroblasts (and other cells) in response to viral infection and dsRNA
- Shares the same IFNAR1/IFNAR2 receptor as IFN-α and the same antiviral signaling pathway (JAK-STAT)
- Upregulates MHC class I; activates NK cells; induces antiviral state
- In the CNS context: reduces neuroinflammation by:
- Reducing T-cell migration across the blood-brain barrier
- Shifting cytokine balance from pro-inflammatory (TH1) to anti-inflammatory (TH2)
- Reducing MMP (matrix metalloproteinase) activity
- Reducing MHC class II expression on CNS-resident cells
Clinical Uses (Therapeutic)
| Condition | Agent | Route |
|---|---|---|
| Relapsing-remitting multiple sclerosis (RRMS) | IFN-β-1a (Avonex, Rebif) | IM once weekly / SC 3×/week |
| Secondary progressive MS | IFN-β-1b (Betaseron, Extavia) | SC every other day |
| Clinically isolated syndrome (CIS) — high risk for MS | IFN-β-1a | SC/IM |
IFN-β reduces relapse rate and MRI lesion burden in MS, and delays conversion from CIS to clinically definite MS. — Goldman-Cecil Medicine; Bradley and Daroff's Neurology
Interferon Gamma (IFN-γ) — Type II
Functions
- Structurally distinct from type I IFNs (<10% homology); uses a different receptor (IFNGR1/IFNGR2) but also signals via JAK-STAT (JAK1/JAK2 → STAT1)
- Produced by activated T cells (TH1) and NK cells/ILC1 — appears later in infection as part of the adaptive response
- The defining cytokine of the TH1 response and also known as macrophage-activating factor (MAF):
- Activates macrophages → promotes production of reactive oxygen and nitrogen species → enhances intracellular killing (critical against mycobacteria, fungi, intracellular parasites)
- Promotes phagocytosis, recruitment, and inflammatory responses
- Upregulates MHC class II on macrophages → enhances antigen presentation to CD4 T cells
- Directs monocyte differentiation toward a macrophage phenotype
- Inhibits viral replication (though weaker antiviral effect than type I IFNs)
- Blocks cell proliferation
- Activates macrophages to produce more IFN-α and IFN-β (amplification loop)
Clinical Uses (Therapeutic)
| Condition | Rationale |
|---|---|
| Chronic granulomatous disease (CGD) | Enhances macrophage oxidative killing; reduces serious infections |
| Severe malignant osteopetrosis | Activates osteoclast function |
| Cutaneous T-cell lymphoma (CTCL) | Immune activation; used in patients who fail IFN-α; may be better tolerated in elderly |
| Lepromatous leprosy (investigational) | Macrophage activation to improve mycobacterial killing |
IFN-γ in CGD: given subcutaneously 3 times per week. It is FDA-approved as IFN-γ-1b (Actimmune) for CGD and osteopetrosis.
Common Side Effects of All Interferons
- Flu-like syndrome: fever, chills, myalgia, fatigue, headache (most common — caused by the systemic effects of the interferon response)
- Bone marrow suppression (neutropenia, thrombocytopenia)
- Elevated liver enzymes
- Depression and neuropsychiatric effects (especially IFN-α)
- Autoimmune phenomena (thyroiditis, SLE exacerbation — type I IFNs are a factor in SLE pathogenesis)
- Injection-site reactions
Summary Table
| IFN-α | IFN-β | IFN-γ | |
|---|---|---|---|
| Type | I | I | II |
| Source | Leukocytes, pDCs | Fibroblasts | T cells, NK cells |
| Receptor | IFNAR1/2 | IFNAR1/2 | IFNGR1/2 |
| Key mechanism | Antiviral state, NK activation, ↑MHC I | Antiviral state, ↓neuroinflammation, ↑MHC I | Macrophage activation, ↑MHC II, TH1 response |
| Main clinical uses | Hepatitis B/C, hairy cell leukemia, melanoma, CTCL, HPV, polycythemia vera | Multiple sclerosis (all forms) | Chronic granulomatous disease, osteopetrosis, CTCL |
Sources: Medical Microbiology 9e; Goldman-Cecil Medicine; Bradley and Daroff's Neurology in Clinical Practice; Sabiston Textbook of Surgery; Cellular and Molecular Immunology
Anakinra mechanism of action
Reading File
Finding Sources
Searching the Web
anakinra mechanism of action IL-1 receptor antagonist pharmacology
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Loading Image
I have everything needed. Here is the comprehensive answer:
Anakinra — Mechanism of Action
What Is Anakinra?
Anakinra (brand name Kineret) is a recombinant form of the human interleukin-1 receptor antagonist (IL-1Ra) — a naturally occurring endogenous protein. It consists of 153 amino acids (MW ~17.3 kDa) and is identical to the non-glycosylated form of human IL-1Ra, except for the addition of one N-terminal methionine. It is the oldest IL-1 inhibitor in clinical use.
Understanding IL-1 First (The Target)
To understand anakinra, you need to understand the IL-1 system:
- IL-1α and IL-1β are potent pro-inflammatory cytokines belonging to the IL-1 family
- They are primarily involved in innate immunity and drive systemic inflammation
- IL-1β is the dominant secreted form; it is produced as an inactive precursor (pro-IL-1β) that is cleaved by caspase-1 (activated by the NLRP3 inflammasome) into its active 17 kDa form
- When IL-1α or IL-1β binds the IL-1 receptor type I (IL-1RI) on cell surfaces, it recruits the IL-1 receptor accessory protein (IL-1RAcP), forming a signaling complex that activates NF-κB and MAPK pathways → triggering transcription of inflammatory genes (TNF, IL-6, IL-8, COX-2, adhesion molecules, MMPs)
- This cascade leads to: fever, synovitis, cartilage degradation, bone erosion, CRP production (via IL-6 → liver), and leukocyte recruitment
- IL-1β also stimulates further IL-6 production → hepatic acute-phase response (CRP ↑)
The body has a natural counter-regulator: endogenous IL-1Ra, secreted by macrophages and hepatocytes, which competitively occupies IL-1RI without activating it — a pure competitive antagonist. In inflammatory diseases, the balance tips toward excess IL-1 over IL-1Ra.
Mechanism of Action of Anakinra
Anakinra is a competitive receptor antagonist at the IL-1 receptor.
Step-by-step:
- Binding: Anakinra binds with high affinity to IL-1 receptor type I (IL-1RI) — with the same affinity as IL-1β itself
- Competitive blockade: By occupying IL-1RI, it prevents both IL-1α and IL-1β from binding to their receptor
- No signaling: Unlike IL-1α/β, anakinra binding does not recruit IL-1RAcP and does not trigger intracellular signaling — it is a pure antagonist (no agonist activity)
- Downstream effects blocked:
- No NF-κB or MAPK activation
- No transcription of pro-inflammatory cytokines (IL-6, TNF, IL-8)
- No COX-2 induction → reduced prostaglandin synthesis
- No metalloproteinase (MMP) upregulation → reduced cartilage/bone destruction
- Reduced CRP and acute-phase reactants (via IL-6 suppression)
- Reduced neutrophil recruitment and activation
"Anakinra is a recombinant form of human interleukin 1 (IL-1) receptor antagonist (IL-1Ra). It blocks the effect of IL-1α and IL-1β on IL-1 receptors, hence decreasing the immune response in inflammatory diseases." — Katzung's Basic and Clinical Pharmacology, 16th Ed.
Contextual Pathway Diagram
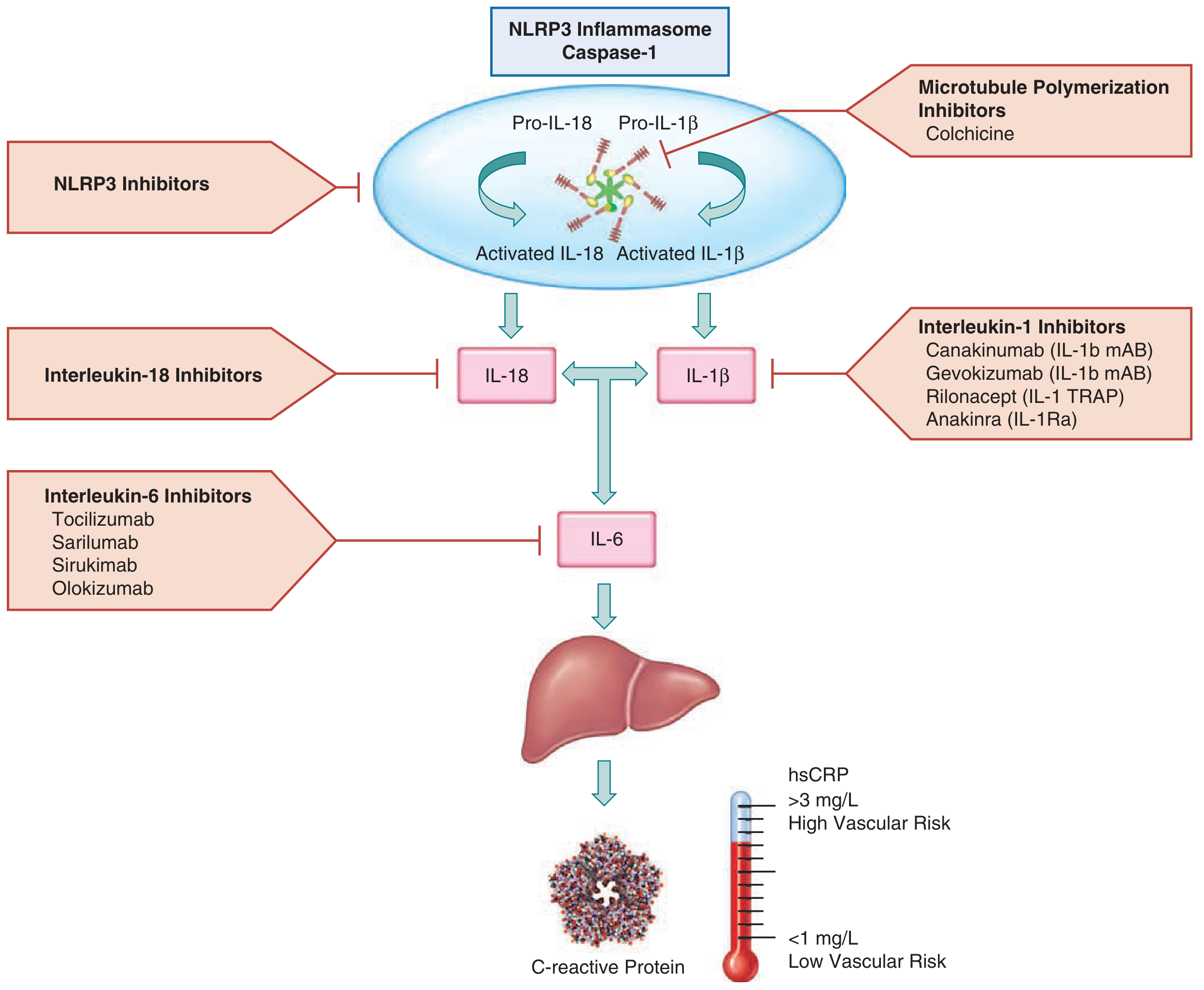
Anakinra (IL-1Ra) blocks IL-1β at the receptor level, downstream of the NLRP3 inflammasome and caspase-1 activation. This prevents the IL-1β → IL-6 → CRP cascade responsible for vascular and systemic inflammation.
— Fuster and Hurst's The Heart, 15th Ed.
Pharmacokinetics
| Parameter | Value |
|---|---|
| Route | Subcutaneous injection |
| Bioavailability | ~95% |
| Half-life | ~5.7 hours (range 3.1–28.2 h) |
| Dosing frequency | Once daily (RA); weight-based for CAPS (max 8 mg/kg/day) |
| Renal adjustment | Every-other-day dosing if CrCl <30 mL/min |
The short half-life (compared to monoclonal antibodies like canakinumab, which has a 26-day half-life) means daily dosing is required — a key practical disadvantage vs. newer IL-1 inhibitors.
Approved and Off-Label Uses
FDA-Approved
| Indication | Notes |
|---|---|
| Rheumatoid arthritis (moderate-severe, DMARD-refractory) | Blocks IL-1-driven synovitis and joint destruction; rarely used today (superseded by TNF blockers and IL-6 inhibitors) |
| Deficiency of IL-1 Receptor Antagonist (DIRA) | Replaces the missing endogenous IL-1Ra protein; lifelong treatment required |
| Cryopyrin-Associated Periodic Syndromes (CAPS) | Includes FCAS, Muckle-Wells, NOMID |
Key Off-Label Uses
- Adult-onset Still's disease (AOSD)
- Systemic juvenile idiopathic arthritis (sJIA)
- Recurrent pericarditis
- Gout (acute flares refractory to colchicine/NSAIDs)
- Hidradenitis suppurativa
- Pustular psoriasis
- Schnitzler syndrome
- COVID-19 (hyperinflammation/cytokine storm in non-intubated hospitalized patients — multiple trials showed mortality reduction)
- Kawasaki disease (refractory cases)
- STEMI — cardiac remodeling prevention (VCUART trials)
Comparison with Other IL-1 Inhibitors
| Drug | Target | Mechanism | Half-life |
|---|---|---|---|
| Anakinra | IL-1RI (receptor) | Blocks both IL-1α and IL-1β competitively | ~6 hours |
| Canakinumab | IL-1β (ligand) | Monoclonal antibody against IL-1β only | ~26 days |
| Rilonacept | IL-1β and IL-1α | IL-1 TRAP (fusion decoy receptor) | ~7 days |
Adverse Effects
- Injection-site reactions — most common (erythema, pain, swelling)
- Infections — increased risk (especially with concurrent TNF blockers — this combination is contraindicated)
- Neutropenia (monitor ANC)
- Headache
- Flu-like symptoms
Key point: Anakinra's mechanism is uniquely elegant — it mimics and amplifies the body's own endogenous braking system on IL-1 signaling. Rather than neutralizing the cytokine itself (like canakinumab), it occupies the receptor and silences the entire IL-1 signaling axis competitively.
Sources: Katzung's Basic and Clinical Pharmacology 16e; Fuster and Hurst's The Heart 15e; Fitzpatrick's Dermatology
Abatacept
Finding Sources
Searching the Web
abatacept mechanism of action CTLA-4 T cell costimulation pharmacology clinical uses
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Finding Sources
Reading File
Reading File
I have everything needed for a comprehensive, well-sourced answer.
Abatacept (Orencia)
What Is Abatacept?
Abatacept is a soluble recombinant fusion protein combining two components:
- The extracellular domain of human CTLA-4 (cytotoxic T-lymphocyte-associated antigen 4, also called CD152)
- The Fc region of human IgG1 (hinge, CH2, and CH3 domains — genetically modified to reduce complement activation)
Apparent molecular weight: ~92 kDa. It is produced by recombinant DNA technology in a mammalian cell expression system.
Understanding the Target: T-Cell Costimulation
Full T-cell activation requires two distinct signals:
| Signal | Interaction | Result |
|---|---|---|
| Signal 1 | Antigen-MHC complex on APC → binds TCR on T cell | Antigen recognition — necessary but insufficient alone |
| Signal 2 (costimulation) | CD80/CD86 (B7-1/B7-2) on APC → binds CD28 on T cell | Full activation: IL-2 production, T-cell proliferation, differentiation |
If Signal 1 occurs without Signal 2, the T cell becomes anergic (non-responsive) — a state of clonal unresponsiveness due to failure to produce IL-2.
CD28 is constitutively expressed on naïve T cells and provides the critical "go" signal.
CTLA-4 is an endogenous immune checkpoint — upregulated after T-cell activation — that also binds CD80/CD86 but with much higher affinity than CD28, delivering an inhibitory signal that dampens ongoing T-cell responses.
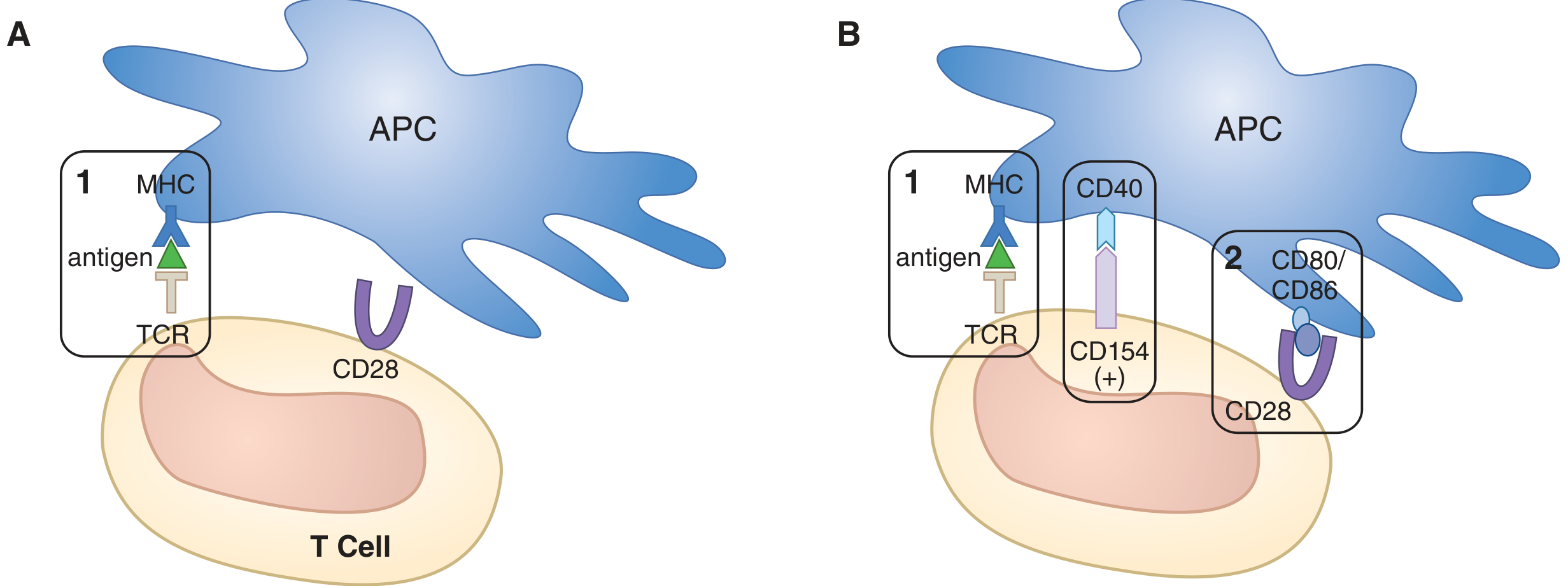
Signal 1 alone is insufficient. Signal 2 (CD28 binding CD80/CD86) completes T-cell activation. Additional costimulatory interactions (CD154–CD40) amplify the response. Without Signal 2, anergy ensues.
— Goodman & Gilman's Pharmacological Basis of Therapeutics, Fig. 38-3
Mechanism of Action
Abatacept mimics and amplifies the endogenous CTLA-4 inhibitory checkpoint:
- The CTLA-4 domain of abatacept binds CD80 (B7-1) and CD86 (B7-2) on APCs with high affinity — higher than CD28
- This competitively blocks CD28 from binding its co-stimulatory ligands on the APC
- Signal 2 is denied → T cells cannot become fully activated
- Without Signal 2, autoreactive T cells either:
- Enter clonal anergy (functional unresponsiveness)
- Fail to produce IL-2 → no proliferation or survival
- Are more susceptible to apoptosis
- Downstream consequences:
- Reduced T-cell proliferation and differentiation
- Reduced T-helper cell cytokine production (IL-17, IFN-γ, TNF)
- Reduced B-cell activation (T-dependent antibody responses impaired → less RF and anti-CCP production)
- Reduced synovial inflammation, osteoclast activation, and joint destruction
- An additional cell-extrinsic mechanism: CTLA-4 binding can physically remove CD80/CD86 from the APC surface (transendocytosis), further limiting availability of costimulatory ligands
"Abatacept is designed to inhibit T-cell activation by binding to ligands CD80 and CD86 on APCs, ultimately blocking CD80 and CD86 interaction with CD28 on T cells." — Katzung's Basic and Clinical Pharmacology, 16th Ed.
"CTLA-4-Ig competitively inhibits CD28 binding to CD80 and CD86 and thus inhibits the activation of T cells." — Goodman & Gilman's, 13th Ed.
Pharmacokinetics
| Parameter | Value |
|---|---|
| Routes | IV infusion or SC injection |
| Bioavailability (SC vs IV) | ~79% |
| Volume of distribution | ~0.11 L/kg |
| Systemic clearance | ~0.28 mL/h/kg |
| Terminal half-life | ~14.3 days |
| Dosing (IV) | Weight-based: 500 mg (<60 kg), 750 mg (60–100 kg), 1000 mg (>100 kg) — monthly infusions |
| Dosing (SC) | 125 mg weekly (adults ≥50 kg); weight-adjusted in children |
| Anti-drug antibody formation | <5% — does not affect clinical outcomes |
FDA-Approved Indications
| Indication | Details |
|---|---|
| Rheumatoid arthritis (RA) | Moderate-to-severe; monotherapy or with MTX/other csDMARDs; for DMARD-inadequate responders |
| Psoriatic arthritis (PsA) | Monotherapy or combination |
| Polyarticular juvenile idiopathic arthritis (pJIA) | Age ≥2 years; SC or IV |
| Acute GVHD prophylaxis | In combination with calcineurin inhibitor + methotrexate in adults and children ≥2 years undergoing allogeneic HSCT |
Key Off-Label Uses
- Systemic lupus erythematosus (SLE)
- Primary Sjögren syndrome
- Type 1 diabetes mellitus (prevention — trials ongoing)
- Inflammatory bowel disease
- Takayasu arteritis
- Psoriasis vulgaris
- Dermatomyositis
Clinical Highlights
- In early rapidly progressive RA, methotrexate + abatacept was superior to methotrexate alone — achieving minimal disease activity as early as 2 months and significantly inhibiting radiographic progression at 1 year
- May prevent progression from undifferentiated inflammatory arthritis to established RA
- Associated with decreased cardiovascular risk and increased HDL compared to TNF inhibitors in RA patients
- Lower rate of latent TB reactivation compared to TNF inhibitors — useful when TB risk is elevated
- Not combined with TNF inhibitors or other biologics — unacceptable increase in serious infections
Adverse Effects
| Effect | Notes |
|---|---|
| Infections | Most important — upper respiratory, urinary tract; screen for latent TB and viral hepatitis before starting |
| Headache, nasopharyngitis, nausea | Common |
| Infusion reactions / hypersensitivity / anaphylaxis | Rare |
| Lymphoma | Possible slight increase (debated); weight of evidence against general cancer increase |
| Live vaccines | Avoid — hold for ≥3 months after discontinuation |
| Contraindication | Concurrent use with TNF inhibitors or other biologics → ↑↑ serious infections |
Abatacept vs. CTLA-4 Checkpoint Inhibitors
This is conceptually important:
| Drug | CTLA-4 relationship | Effect |
|---|---|---|
| Abatacept | CTLA-4 agonist (mimics it) | Blocks T-cell activation → immunosuppressive |
| Ipilimumab | CTLA-4 antagonist (blocks it) | Releases T-cell brake → immunostimulatory (used in cancer) |
They operate on the same molecular target but in opposite directions — abatacept suppresses autoimmunity, ipilimumab unleashes anti-tumor immunity.
Sources: Katzung's Basic and Clinical Pharmacology 16e; Goodman & Gilman's 13e; Goldman-Cecil Medicine; Lippincott Pharmacology; Janeway's Immunobiology 10e; Sabiston Textbook of Surgery
This is a shared conversation. Sign in to Orris to start your own chat.