Anatomy of lung
Reading File
Finding Sources
Finding Sources
Searching Images
lung anatomy lobes bronchi
Reading File
Reading File
Finding Sources
Searching Images
lung lobes fissures hilum anatomy diagram
Reading File
Loading Image
Loading Image
Reading File
I now have comprehensive content. Let me compile the full anatomy of the lung answer.
Anatomy of the Lung
— Gray's Anatomy for Students; Murray & Nadel's Textbook of Respiratory Medicine
Overview
The two lungs are organs of respiration occupying the right and left pleural cavities on either side of the mediastinum. Air enters and exits via the main bronchi (branches of the trachea). The right lung is slightly larger than the left because the heart bulges more to the left side.
Each lung has a half-cone shape with:
- Base — rests on the diaphragm
- Apex — projects above rib I into the root of the neck
- Two surfaces — costal (adjacent to ribs/intercostal spaces) and mediastinal (against the mediastinum and vertebral column)
- Three borders — inferior (sharp, separates base from costal surface), anterior (sharp), and posterior (smooth and rounded)
Lobes and Fissures
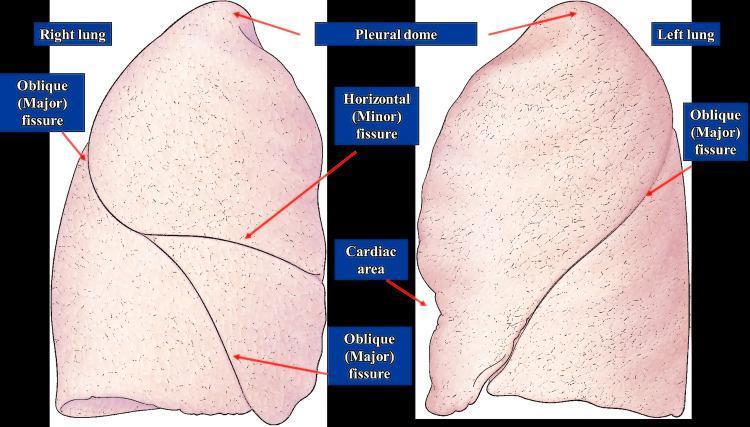
Right Lung — 3 lobes, 2 fissures
| Fissure | Course | Lobes Separated |
|---|---|---|
| Oblique (major) fissure | Starts between T II–IV spinous processes, crosses 5th interspace laterally, follows rib VI anteriorly | Upper from lower lobes |
| Horizontal (minor) fissure | Follows the 4th rib anteriorly to the midaxillary line | Upper from middle lobe |
- Superior (upper) lobe — anterolateral thoracic wall, apex into neck
- Middle lobe — anteroinferior (unique to right lung)
- Inferior (lower) lobe — posterior and inferior thoracic wall
Medial surface relations: superior vena cava, azygos vein, esophagus, right subclavian vessels.
Left Lung — 2 lobes, 1 fissure
| Fissure | Course |
|---|---|
| Oblique (major) fissure only | Slightly more oblique than right; begins T III–IV, crosses 5th interspace, follows rib VI |
- Superior lobe — anterolateral wall, with the lingula projecting as a tongue-like extension over the cardiac notch
- Inferior lobe — posterior and inferior wall
- Cardiac notch — concavity on medial surface due to heart projection into left pleural cavity
Medial surface relations: heart, aortic arch, thoracic aorta, esophagus, left subclavian vessels.
Hilum and Root
The root of each lung is a short tubular collection of structures attaching the lung to the mediastinum, covered by a pleural sleeve that reflects as visceral pleura. The hilum is the region where structures enter/leave, outlined by this pleural reflection.
Structures within the root/hilum:
- 1 pulmonary artery (superior at hilum)
- 2 pulmonary veins (inferior at hilum)
- Main bronchus (somewhat posterior)
- Bronchial vessels, nerves, lymphatics
A thin pulmonary ligament extends inferiorly from the hilum to the mediastinum, stabilizing the lower lobe and accommodating movement during breathing.
Key relations: vagus nerves pass posterior to the roots; phrenic nerves pass anterior to the roots.
On the right, the lobar bronchus to the superior lobe branches from the main bronchus within the root and lies superior to the pulmonary artery. On the left, it branches within the lung.
Bronchial Tree
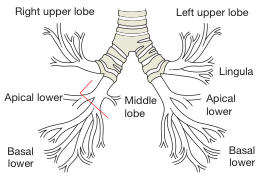
The trachea extends from C6 (lower neck) to T4/5 (mediastinum) where it bifurcates at the carina into:
| Feature | Right main bronchus | Left main bronchus |
|---|---|---|
| Width | Wider | Narrower |
| Angle | More vertical | More oblique |
| Clinical relevance | Foreign bodies lodge here more often | — |
Hierarchy of division:
- Main (primary) bronchi → enter hilum
- Lobar (secondary) bronchi → one per lobe
- Segmental (tertiary) bronchi → each supplies a bronchopulmonary segment
Bronchopulmonary Segments
Each bronchopulmonary segment is an independent unit with its own segmental bronchus, artery, and separated (in part) by intersegmental veins. They are the smallest resectable lung units.
| Right Lung | Left Lung |
|---|---|
| Upper lobe: apical, posterior, anterior | Upper lobe: apicoposterior, anterior, superior lingular, inferior lingular |
| Middle lobe: lateral, medial | (no middle lobe) |
| Lower lobe: superior, medial basal, anterior basal, lateral basal, posterior basal | Lower lobe: superior, anteromedial basal, lateral basal, posterior basal |
(Right lung: 10 segments; Left lung: 8–10 segments depending on classification)
Blood Supply
Pulmonary (Functional) Circulation
- Pulmonary arteries — carry deoxygenated blood from the right ventricle to the lungs for gas exchange
- Pulmonary veins (2 per side) — return oxygenated blood to the left atrium
Bronchial (Nutritive) Circulation
- Right bronchial artery — usually from the 3rd posterior intercostal artery
- Left bronchial arteries (×2) — arise directly from the thoracic aorta at T5 level and below the left bronchus
- Bronchial arteries run on posterior surfaces of bronchi supplying pulmonary tissue
Bronchial venous drainage:
- Partly into pulmonary veins / left atrium
- Right side → azygos vein; Left side → superior intercostal vein or hemiazygos vein
Innervation
The lungs and visceral pleura are supplied through the anterior and posterior pulmonary plexuses lying anteriorly and posteriorly to the tracheal bifurcation. Branches originate from:
| Source | Effect |
|---|---|
| Vagus nerve (parasympathetic) | Bronchoconstriction, increased secretion |
| Sympathetic trunks | Bronchodilation, vasoconstriction |
Lymphatic Drainage
Lymph drains centripetally:
- Superficial (subpleural) plexus → around the margins of the lung → bronchopulmonary nodes at the hilum
- Deep (peribronchial) plexus → along the bronchi → pulmonary nodes within the lung → bronchopulmonary (hilar) nodes → tracheobronchial nodes → right and left bronchomediastinal trunks
From the right side, lymph enters the right lymphatic duct. From the left side, most lymph drains into the thoracic duct.
Cellular Architecture of the Airway (Microscopic)
(Murray & Nadel's Respiratory Medicine)
The airway epithelium is pseudostratified columnar and contains:
| Cell Type | Function |
|---|---|
| Ciliated cells | ~50% of epithelial cells; ~200 cilia per cell beating at 8–15 Hz for mucociliary clearance |
| Goblet cells | Mucus secretion |
| Basal cells | Progenitor cells; self-renew and differentiate into secretory and ciliated cells |
| Club cells | Secretory; increase distally toward bronchioles |
| Neuroendocrine cells | Chemoreceptors; sparse |
| Ionocytes | Ion transport |
Submucosal glands (serous + goblet cells) are present in larger airways and decrease distally.
Clinical Correlates
- Foreign body aspiration → tends to lodge in the right lower lobe (wider, more vertical right main bronchus)
- Cardiac notch on left lung → used in pericardiocentesis approach
- Lingula of left lung → left-sided equivalent of right middle lobe; commonly affected in aspiration
- Bronchopulmonary segments → basis for surgical segmentectomy and bronchoscopic localization of lesions
- Phrenic nerve (anterior to root) vs vagus nerve (posterior to root) — important in thoracic surgery
Sources: Gray's Anatomy for Students (pp. 211–215); Murray & Nadel's Textbook of Respiratory Medicine, 2-Volume; Bailey and Love's Surgery (bronchial tree diagram)
Embryology of lung
Finding Sources
Finding Sources
Searching Images
lung embryology development stages pseudoglandular canalicular saccular alveolar
Finding Sources
Finding Sources
Loading Image
Reading File
Reading File
Reading File
Reading File
Loading Image
Searching Images
surfactant type I type II pneumocyte alveolar development
Reading File
Loading Image
I now have comprehensive, detailed content from three authoritative sources. Let me compile the full answer.
Embryology of the Lung
— Murray & Nadel's Textbook of Respiratory Medicine; Fishman's Pulmonary Diseases and Disorders; Mulholland & Greenfield's Surgery
Origin
The lung originates from the ventral foregut endoderm — the same anterior gut tube that gives rise to the thyroid, stomach, liver, and pancreas. As an organ of exceptional structural complexity, the lung is formed from contributions of all three embryonic germ layers:
| Germ Layer | Contribution |
|---|---|
| Endoderm | Respiratory epithelium (airways, alveoli) |
| Mesoderm | Mesenchyme → smooth muscle, vessels, cartilage, fibroblasts, pleura |
| Ectoderm | Innervation |
Initial Specification (~Week 4)
- At ~week 4 (Carnegie Stage 10), a cluster of Nkx2-1 (TTF-1 / thyroid transcription factor-1)-expressing cells in the ventral foregut endoderm become specified as respiratory progenitors — the earliest known marker of the respiratory lineage
- WNT signaling (WNT2 and WNT2B from foregut mesoderm; β-catenin in endoderm) is critical for specification — loss of canonical WNT signaling prevents Nkx2-1 expression and lung formation
- The respiratory diverticulum appears as a ventral outpouching of the foregut
- The tracheoesophageal septum forms, dividing the trachea (ventral) from the esophagus (dorsal)
- Failure → tracheoesophageal fistula (TEF) ± esophageal atresia (~1 in 3,500 live births)
Five Stages of Lung Development
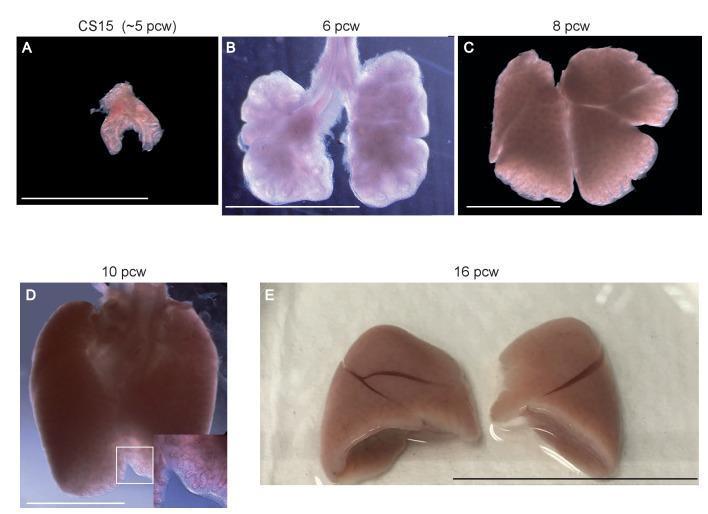
| Stage | Timing (Human) | Key Events |
|---|---|---|
| Embryonic | Weeks 4–7 | Lung bud formation, tracheal separation |
| Pseudoglandular | Weeks 5–17 | Branching morphogenesis, conducting airways |
| Canalicular | Weeks 16–28 | Acinar formation, vascularization |
| Saccular | Weeks 26–36 | Primitive saccules, pneumocyte differentiation, surfactant |
| Alveolar | Week 36 → early childhood | Secondary septation, mature alveoli |
Stage 1 — Embryonic (Weeks 4–7)
- Left and right lung buds bud from the posterior respiratory primordium into the surrounding mesenchyme
- Left-right asymmetry is already specified at this stage (as part of visceral situs determination)
- Pleural cavities begin separating from pericardial and peritoneal spaces by week 3
- All lung lobes become enclosed in pleura (mesothelium — a thin squamous epithelial layer)
- Lung buds rapidly elongate and initiate branching as they transition to the next stage
Stage 2 — Pseudoglandular (Weeks 5–17)
- Named for the gland-like histologic appearance: epithelial tubules separated by thick mesenchyme
- The lung bud undergoes branching morphogenesis → main bronchi → lobar bronchi → segmental bronchi → terminal bronchioles (all 23 generations of conducting airways established by week 16)
- Key molecular drivers of branching:
- FGF10 (fibroblast growth factor 10) from mesenchyme → signals through FGFR2b on epithelium → stimulates bud outgrowth
- Sprouty genes (Spry1, Spry2) act as negative feedback inhibitors of FGF signaling to shape branching pattern
- BMP4 at bud tips restricts branching
- Wnt7b in epithelium → maintains airway smooth muscle differentiation
- Proximal epithelial cells are tall columnar; distal cells are cuboidal
- Vasculature branches in parallel with the epithelium; smooth muscle cells surround developing airways and vessels
- No gas exchange possible at this stage — acini not yet formed
- Clinical note: Arrest here → congenital pulmonary airway malformation (CPAM), bronchogenic cysts, pulmonary sequestration
Stage 3 — Canalicular (Weeks 16–28)
- Airway branching is nearly complete
- Epithelial acini appear at the distal ends of terminal bronchioles
- Terminal bronchioles divide into respiratory bronchioles → alveolar ducts
- Vasculature becomes abundant and closely apposed to the epithelium (primitive air-blood barrier begins to form)
- Mesenchyme becomes progressively thinner between epithelial tubules
- Type I (AT1) and Type II (AT2) alveolar epithelial cells begin to differentiate:
- AT1 cells: flat, squamous; cover ~95% of alveolar surface; responsible for gas exchange
- AT2 cells: cuboidal; produce surfactant (begin appearing in late canalicular stage)
- Gas exchange becomes theoretically possible from the late canalicular phase (~24–26 weeks)
- Viability threshold: Premature infants ≥22–25 weeks may be viable; AAP defines lower limit at 22 weeks
Stage 4 — Saccular (Weeks 26–36 / Terminal Sac Phase)
- Primitive saccules develop at the distal ends of the bronchial tree — wide-lumen, thin-walled structures lined by AT1 and AT2 cells
- AT1 cell differentiation increases air space size; fusion of epithelial and endothelial basal laminae brings AT1 cells and capillaries into close contact → primitive alveolo-capillary barrier
- AT2 cells (Type II pneumocytes) produce and secrete surfactant — a lipoprotein complex that lowers alveolar surface tension and prevents collapse during expiration
- Surfactant = phospholipids (predominantly DPPC) + surfactant proteins (SP-A, B, C, D)
- SP-B and SP-C are critical for surface tension reduction
- Vasculature has fully invested the lung parenchyma
- Gas exchange sufficient for immediate postnatal survival is now possible
Respiratory Distress Syndrome (RDS): Premature birth before adequate surfactant production causes alveolar collapse, hypoxia, and respiratory failure. Management: antenatal betamethasone (<34 weeks) to accelerate surfactant production; postnatal exogenous surfactant replacement; CPAP/ventilatory support.
Stage 5 — Alveolar (Week 36 → Early Childhood, ~8 years)
- Secondary septation: primitive saccule walls develop secondary septa (crests) that subdivide them into true alveoli, dramatically increasing gas-exchange surface area
- At birth: ~50 million alveoli
- Adult: ~300–500 million alveoli
- Total surface area at maturity: ~70 m²
- Septa form, lengthen, and thin as the interstitial fibroblasts lay down elastin and collagen scaffolding
- The alveolar surface area continues to grow postnatally through multiplication of alveoli (predominantly 0–2 years) and then through alveolar enlargement with lung volume growth
Alveolar Structure at Maturity
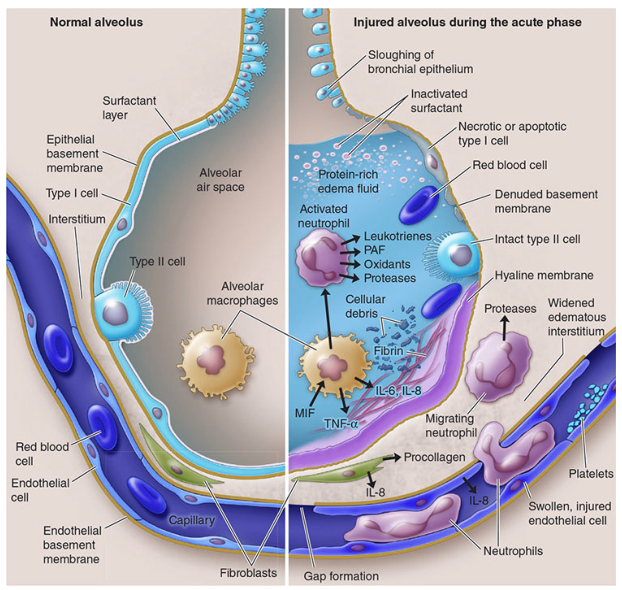
| Cell Type | Features |
|---|---|
| Type I pneumocyte (AT1) | Flat, covers ~95% of alveolar surface; gas exchange; vulnerable to injury |
| Type II pneumocyte (AT2) | Cuboidal, ~5% surface; surfactant production; stem cell (regenerates AT1 after injury) |
| Alveolar macrophage | First-line immune defense; phagocytes |
| Endothelial cells | Line capillaries; part of alveolo-capillary membrane |
Key Molecular Signals Summary
| Signal | Source | Role |
|---|---|---|
| Nkx2-1 (TTF-1) | Epithelium | Master regulator of respiratory fate specification |
| WNT2/WNT2B | Mesoderm | Foregut respiratory progenitor specification |
| FGF10 | Mesenchyme | Stimulates bud outgrowth and branching |
| FGFR2b | Epithelium | Receptor for FGF10 |
| Sprouty 1/2 | Epithelium | Negative feedback on FGF → shapes branching pattern |
| BMP4 | Bud tips | Restricts branching, promotes differentiation |
| Wnt7b | Epithelium | Airway smooth muscle differentiation |
| Glucocorticoids | Systemic | Accelerate AT2 maturation and surfactant synthesis |
Congenital Abnormalities by Stage
| Stage | Defect | Example |
|---|---|---|
| Embryonic | Tracheoesophageal separation failure | TEF ± esophageal atresia (VACTERL) |
| Pseudoglandular | Abnormal branching | CPAM, bronchogenic cyst, sequestration |
| Canalicular/Saccular | Premature birth, surfactant deficiency | RDS (hyaline membrane disease) |
| Alveolar | Arrested secondary septation | Bronchopulmonary dysplasia (BPD) in preterm infants |
Sources: Murray & Nadel's Textbook of Respiratory Medicine (pp. 64–70); Fishman's Pulmonary Diseases and Disorders (Chapter 4); Mulholland & Greenfield's Surgery, 7e (pp. 5287–5288)
Embryology of lung
The answer I just provided covers lung embryology in full detail. Here's a concise summary for quick reference:
Embryology of the Lung
Origin
The lung arises from the ventral foregut endoderm (~week 4). All three germ layers contribute:
- Endoderm → respiratory epithelium
- Mesoderm → smooth muscle, vessels, cartilage, pleura
- Ectoderm → innervation
The respiratory diverticulum appears as a ventral outpouching of the foregut. The tracheoesophageal septum divides trachea from esophagus — failure causes TEF ± esophageal atresia.
Nkx2-1 (TTF-1) is the earliest marker of respiratory progenitor cells; WNT signaling (WNT2/WNT2B) is critical for their specification.
Five Developmental Stages

| Stage | Weeks | Key Events | Clinical Relevance |
|---|---|---|---|
| Embryonic | 4–7 | Lung buds form; trachea separates from esophagus; left-right asymmetry specified | TEF, tracheal atresia |
| Pseudoglandular | 5–17 | Branching morphogenesis; all 23 generations of conducting airways form by wk 16; gland-like histology (thick mesenchyme) | CPAM, bronchogenic cyst, sequestration |
| Canalicular | 16–28 | Acini appear; vasculature closely apposed to epithelium; AT1 and AT2 cells differentiate; late canalicular = first viable gas exchange | Threshold of viability ~24–26 wks |
| Saccular | 26–36 | Primitive saccules form; AT2 cells produce surfactant; alveolo-capillary barrier established | RDS (surfactant deficiency in preterm) |
| Alveolar | 36 wks → ~8 yrs | Secondary septation → 50 million alveoli at birth → ~300–500 million at maturity; surface area ~70 m² | BPD in preterm infants |
Alveolar Cell Types (at maturity)

| Cell | Coverage | Function |
|---|---|---|
| Type I pneumocyte (AT1) | ~95% of surface | Gas exchange; thin for diffusion |
| Type II pneumocyte (AT2) | ~5% of surface | Surfactant production; stem cell (regenerates AT1 after injury) |
| Alveolar macrophage | Air space | Innate immune defense |
Key Molecular Drivers
| Molecule | Role |
|---|---|
| Nkx2-1 / TTF-1 | Master transcription factor for respiratory fate |
| WNT2/WNT2B + β-catenin | Progenitor specification in foregut |
| FGF10 → FGFR2b | Mesenchyme-to-epithelium signal driving bud outgrowth and branching |
| Sprouty 1/2 | Negative feedback on FGF; shapes branching geometry |
| BMP4 | Restricts branching at bud tips |
| Glucocorticoids | Accelerate AT2 maturation and surfactant synthesis (basis for antenatal betamethasone) |
Surfactant & RDS
- Surfactant = phospholipids (mainly DPPC) + proteins SP-A, SP-B, SP-C, SP-D
- Produced by AT2 cells starting in the saccular phase
- SP-B and SP-C are essential for surface tension reduction
- Deficiency in premature neonates → Respiratory Distress Syndrome (RDS)
- Prevention: antenatal betamethasone (<34 weeks gestation)
- Treatment: postnatal exogenous surfactant + CPAP/ventilation
Sources: Murray & Nadel's Textbook of Respiratory Medicine; Fishman's Pulmonary Diseases and Disorders; Mulholland & Greenfield's Surgery, 7e
This is a shared conversation. Sign in to Orris to start your own chat.