Histoplasmosis
Reading File
Finding Sources
Searching PubMed
histoplasmosis
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have comprehensive material. Here is a complete, well-organized overview of histoplasmosis.
Histoplasmosis
Organism & Morphology
Histoplasma capsulatum is a thermally dimorphic fungus:
- Mold phase (room temperature, environment): produces characteristic spiked macroconidia (8–15 µm) and small microconidia (2–5 µm). The microconidia are the infectious particles — small enough to reach the terminal bronchioles.
- Yeast phase (37°C, in host): small (2–5 µm), oval yeasts with narrow-based budding, found intracellularly within macrophages.
Fig. 1 — Mold phase: Spiked spherical macroconidia of H. capsulatum (lacto-phenol cotton blue stain)
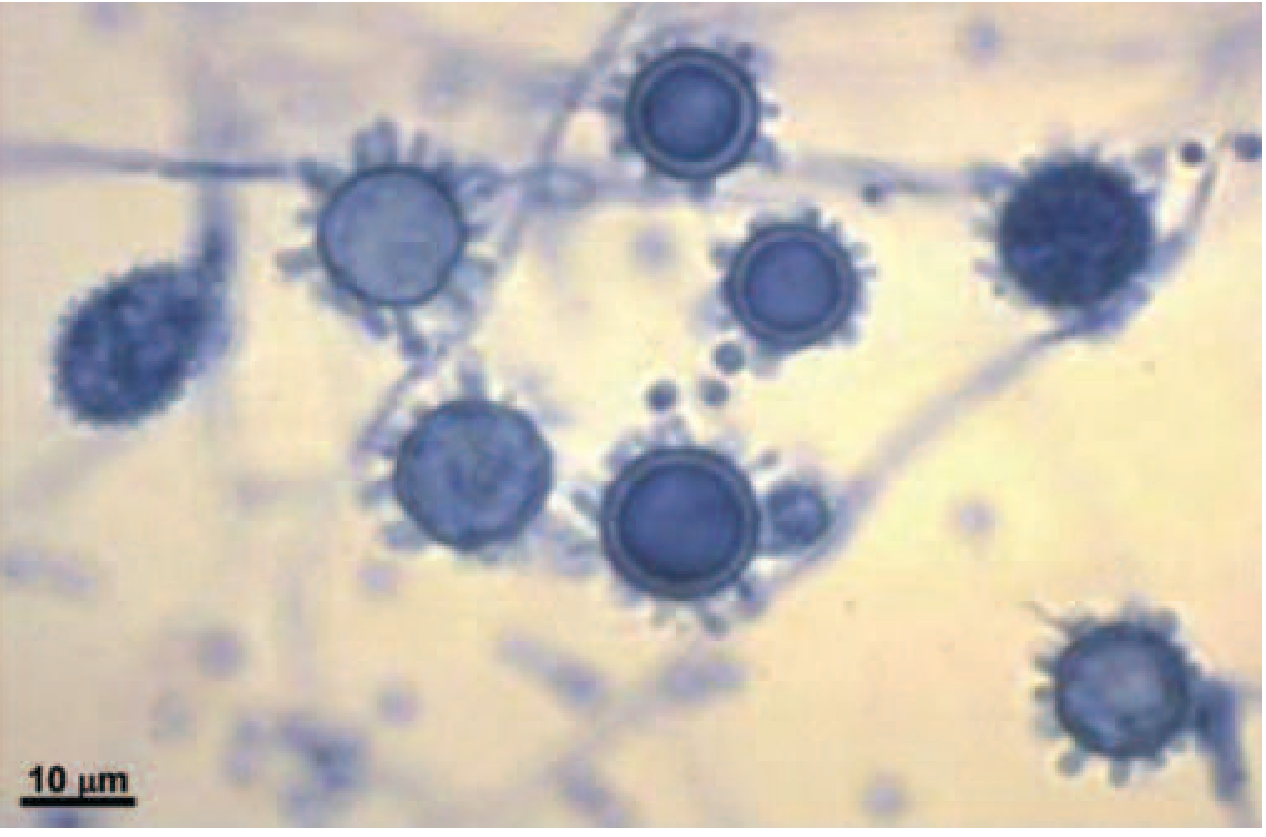
Fig. 2 — Yeast phase: Small narrow-budding yeasts from BAL fluid (Grocott's methenamine silver stain)
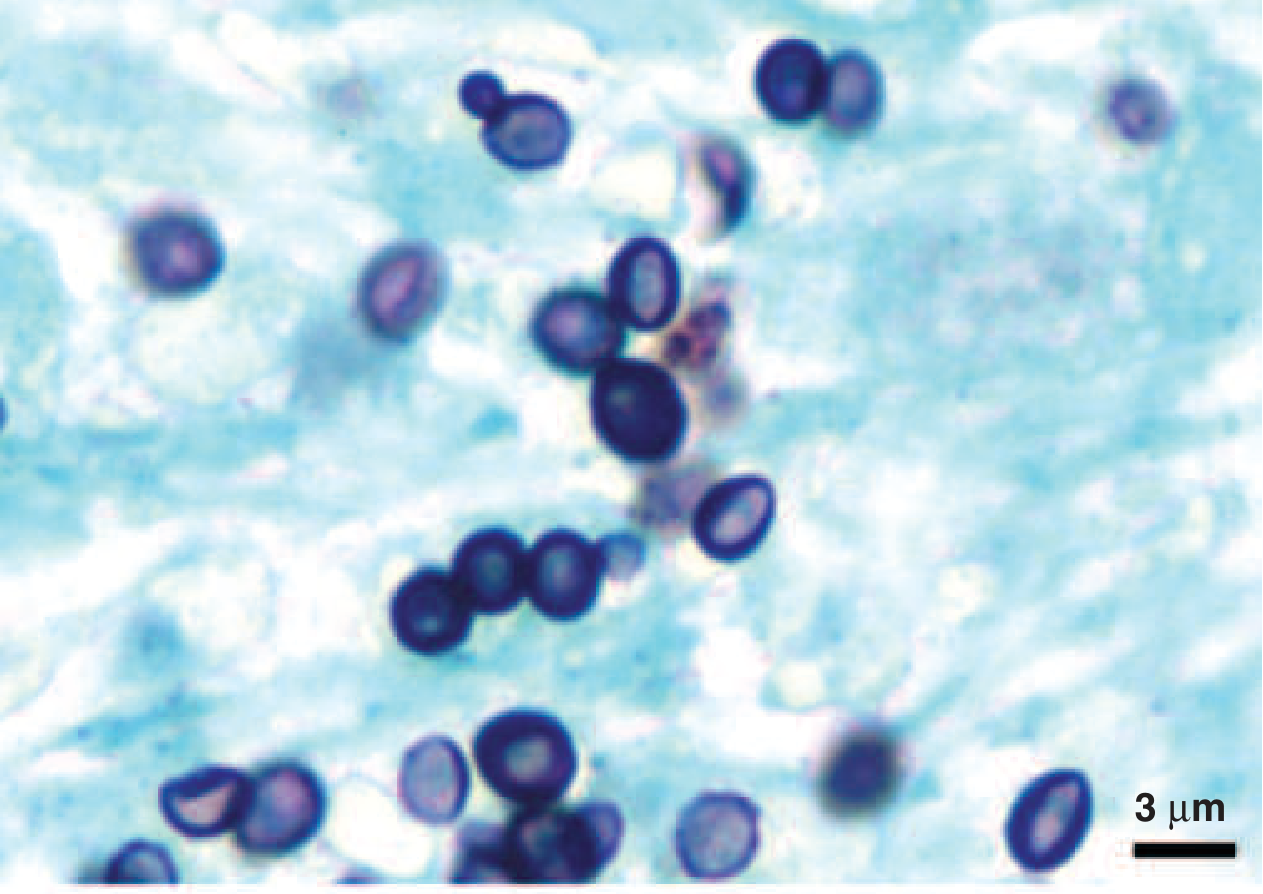
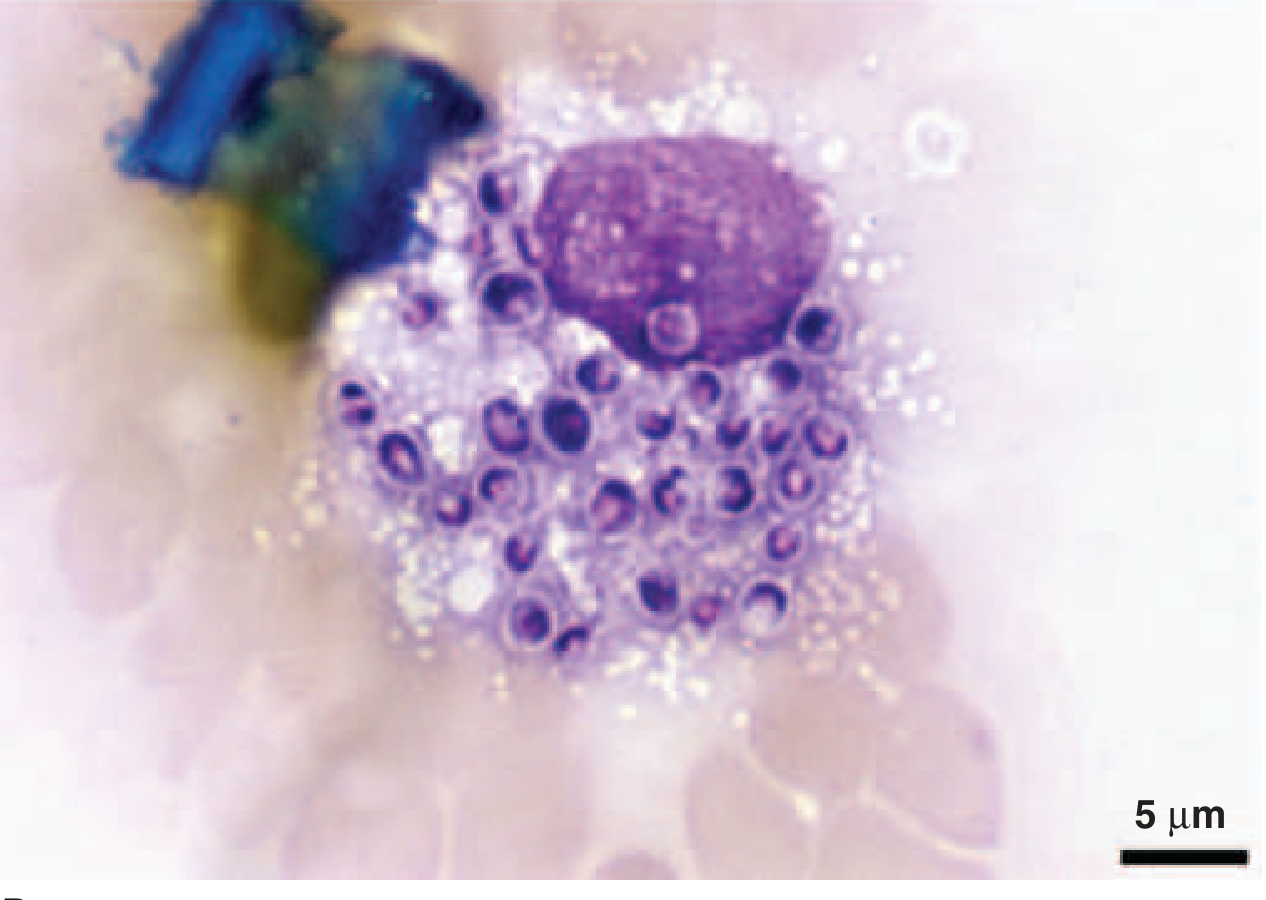
Fig. 3 — Intracellular yeasts of H. capsulatum within an alveolar macrophage (Giemsa stain — disseminated histoplasmosis in AIDS)
Epidemiology
- Most prevalent endemic mycosis in North America, particularly the Ohio and Mississippi River valleys; also Mexico, Central/South America (especially Brazil), Africa, and Asia.
- Up to 75% of adults in endemic areas show serologic/radiographic evidence of prior infection without clinical illness.
- The organism thrives in humid, acidic soil enriched with bird or bat droppings (roosting sites, caves, chicken coops, old buildings).
- High-risk exposures: spelunking, excavation, demolition/renovation of old buildings, cleaning chicken coops, cutting dead trees.
- Outside endemic areas, most cases represent imported disease (travelers, immigrants).
- At-risk populations are growing due to immunosuppressive therapies (anti-TNF agents, transplant immunosuppression, etc.).
— Harrison's Principles of Internal Medicine 22E, p. 218
Pathogenesis
- Inhalation of microconidia → reach alveolar spaces
- Engulfed by alveolar macrophages → transform to yeast (calcium- and iron-dependent process)
- Yeasts evade intracellular killing and can spread to regional lymph nodes and hematogenously
- In immunocompetent hosts, cell-mediated immunity (IFN-γ, TNF-α pathways) eventually controls infection → granuloma formation → calcification
- In immunocompromised hosts: uncontrolled intracellular proliferation → progressive disseminated histoplasmosis (PDH)
Clinical Syndromes
1. Asymptomatic / Subclinical Infection
The majority of exposures in immunocompetent individuals. Healed infection leaves calcified pulmonary nodules or "buckshot" calcifications in the lung and spleen — frequently found incidentally on CT.
2. Acute Pulmonary Histoplasmosis
| Feature | Light Exposure | Heavy Exposure |
|---|---|---|
| Inoculum | Small | Large |
| Onset | 1–4 weeks post-exposure | 1–4 weeks post-exposure |
| Symptoms | Flu-like: fever, chills, headache, myalgia, dry cough | As left + dyspnea, respiratory distress |
| CXR | Focal infiltrate ± mediastinal adenopathy | Diffuse interstitial/reticulonodular infiltrates, miliary pattern |
| Dissemination | Uncommon | ~40% of cases |
| Course | Self-limited weeks | May → respiratory failure |
Inflammatory complications (not true dissemination):
- Arthritis/arthralgia + erythema nodosum: 5–10% of cases; resembles sarcoidosis; responds to anti-inflammatories
- Pericarditis: <10%; chest pain, pericardial rub; may cause tamponade; managed with anti-inflammatories ± drainage
3. Subacute Pulmonary Histoplasmosis
- Subacute onset: fever, cough, chest pain
- CXR: mediastinal lymphadenopathy + patchy infiltrates
- Most recover in weeks; some have prolonged fatigue
4. Chronic Cavitary Histoplasmosis
- Occurs in smokers with underlying structural lung disease (bullous emphysema)
- Productive cough, dyspnea, low-grade fever, night sweats, weight loss
- CXR/CT: upper-lobe infiltrates, cavitation, pleural thickening — closely resembles pulmonary tuberculosis
- Progressive without treatment; ~15% relapse rate even with adequate therapy
5. Progressive Disseminated Histoplasmosis (PDH)
- ~1 in 2,000 infections; ~70% in immunocompromised hosts
- Risk factors: AIDS (CD4 <200/µL), solid organ transplant, anti-TNF-α agents (infliximab, adalimumab, etanercept), methotrexate, corticosteroids, extremes of age
- Manifestations:
- Fever, weight loss, cough, dyspnea (most common)
- Hepatosplenomegaly (~50%), lymphadenopathy (~33%)
- Sepsis-like syndrome in severe immunosuppression
- Meningitis or focal brain lesions (5–10%)
- Oral mucosal ulcerations, GI ulcerations/bleeding
- Adrenal insufficiency (adrenal gland involvement)
- Thrombocytopenia, pancytopenia
- CXR abnormal in 70%: diffuse interstitial or reticulonodular infiltrates, miliary pattern
6. Mediastinal Complications
- Mediastinal lymphadenopathy: hilar/mediastinal nodes undergo necrosis and coalesce → large masses compressing great vessels, airways, or esophagus; may create bronchoesophageal fistulae
- Fibrosing mediastinitis: uncommon but serious; progressive fibrosis encases mediastinal structures → superior vena cava syndrome, pulmonary vessel obstruction, airway obstruction; fatal in up to one-third of cases; no effective antifungal treatment at this stage (fibrosis is irreversible)
- Broncholithiasis: calcified lymph nodes erode into bronchi → hemoptysis, obstruction, tracheoesophageal fistulae
7. Pulmonary Nodules
- Histoplasma nodules must be differentiated from malignancy
- Calcification favors histoplasmosis but does not exclude cancer
- PET scan can be positive in histoplasmosis (mimics malignancy)
- Slow enlargement (up to 2 mm/year) is not progressive infection and does not require treatment
Diagnosis
| Test | Best Use | Notes |
|---|---|---|
| Urine antigen (MVista®) | PDH, severe acute pulmonary disease | Most sensitive for PDH (>90%); lower sensitivity in focal/subacute disease; cross-reacts with Blastomyces, Coccidioides, Paracoccidioides |
| Serum antigen | PDH | Less sensitive than urine antigen |
| Histopathology/cytopathology | Rapid diagnosis of moderate-severe or disseminated disease | GMS or PAS staining; BAL, bone marrow, liver biopsy |
| Culture | Chronic/subacute disease | Gold standard, but slow — results take up to 4 weeks; BSL-3 pathogen |
| Serology (complement fixation, immunodiffusion) | Subacute/chronic disease | Less useful in acute or disseminated disease; may be negative in immunosuppressed |
| Antigen monitoring | Response to therapy, relapse detection | Serial urine antigen testing |
Before initiating immunosuppressive therapy for presumed sarcoidosis, active histoplasmosis must be excluded — the two conditions share many clinical and radiographic features (mediastinal adenopathy, granulomas, elevated ACE, elevated ESR/CRP). — Fishman's Pulmonary Diseases and Disorders
Treatment (IDSA 2007 / ATS 2011 Guidelines)
Mild to Moderate Acute Pulmonary Histoplasmosis
- If symptomatic >3 weeks or moderate disease: Itraconazole 200 mg TID × 3 days (load), then 200 mg BID × 6–12 weeks
- Fluconazole is less effective; ketoconazole should not be used
- Echinocandins are intrinsically ineffective against Histoplasma
Moderately Severe to Severe Acute Pulmonary Histoplasmosis
- Liposomal amphotericin B 3–5 mg/kg/day × 1–2 weeks → then itraconazole 200 mg TID × 3 days, then 200 mg BID × 12 weeks
- Deoxycholate amphotericin 0.7–1 mg/kg/day if liposomal unavailable or low nephrotoxicity risk
- Corticosteroids (methylprednisolone 0.5–1 mg/kg IV, or prednisone 40–60 mg/day) for hypoxemia or respiratory distress, especially in HIV patients at risk for IRIS
Chronic Cavitary Histoplasmosis
- Itraconazole 200 mg TID × 3 days load, then 200 mg BID × minimum 1 year (extending to 18–24 months reduces ~15% relapse rate)
Progressive Disseminated Histoplasmosis (PDH)
- Liposomal amphotericin B × 1–2 weeks → itraconazole for minimum 1 year total
- Mild-moderate PDH in immunocompetent hosts: itraconazole alone may suffice
Immunocompromised / HIV
- Itraconazole 200 mg once or twice daily indefinitely if immunosuppression cannot be reversed
- In HIV: continue until CD4 >200/µL (with ART)
- Annual urine Histoplasma antigen monitoring on maintenance therapy
Pleural Effusion from Histoplasmosis
- Lymphocytic exudate; pleural biopsy may show noncaseating granulomas
- Usually self-limited — no antifungal treatment needed unless:
- Effusion persists >3–4 weeks, OR
- Patient is immunocompromised
Key Associations to Remember
| Feature | Detail |
|---|---|
| Endemic region | Ohio and Mississippi River valleys |
| Infection source | Bird/bat droppings in soil |
| Dimorphism | Mold at RT → yeast at 37°C (inside macrophages) |
| Intracellular pathogen | Evades killing inside macrophages |
| Mimics TB | Chronic cavitary form: upper lobe, cavitation |
| Mimics sarcoidosis | Mediastinal adenopathy, granulomas, elevated ACE |
| Mimics malignancy | Nodules + positive PET scan |
| Most sensitive test for PDH | Urine Histoplasma antigen |
| Drug class NOT to use | Echinocandins (intrinsic resistance) |
| Serious irreversible complication | Fibrosing mediastinitis |
| Presumed ocular histoplasmosis syndrome | Punched-out chorioretinal scars, peripapillary atrophy, CNV — no vitreous cells |
Recent Evidence (2024)
Two recent systematic reviews are relevant:
- Histoplasmosis in HIV patients in Europe (2024) — Systematic review highlighting histoplasmosis as an underrecognized imported opportunistic infection in HIV patients in Europe (PMID 38993488)
- WHO Fungal Priority Pathogens List systematic review (2024) — Histoplasma included; review documents global burden and diagnostic gaps, particularly in low-income countries (PMID 38935903)
Sources: Harrison's Principles of Internal Medicine 22E; Fishman's Pulmonary Diseases and Disorders; Murray & Nadel's Textbook of Respiratory Medicine
This is a shared conversation. Sign in to Orris to start your own chat.