Oral hypoglycemic drugs
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
oral hypoglycemic drugs mechanism of action diabetes
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Searching Images
sulfonylurea meglitinide thiazolidinedione SGLT2 inhibitor DPP4 inhibitor antidiabetic drug classes comparison table
I have comprehensive content from Katzung's pharmacology textbook covering all major oral hypoglycemic drug classes. Now I'll compile a thorough, well-structured answer.
Oral Hypoglycemic Drugs
Oral hypoglycemic (antidiabetic) agents are used primarily for Type 2 Diabetes Mellitus. They work through distinct mechanisms targeting different aspects of glucose homeostasis. The major classes are outlined below.
1. Sulfonylureas
Mechanism
Sulfonylureas bind to a 140-kDa sulfonylurea receptor on pancreatic β-cells, closing ATP-sensitive K⁺ channels → membrane depolarization → Ca²⁺ influx → insulin exocytosis. They are insulin secretagogues (require functioning β-cells).
Generations & Agents
| Generation | Drug | Dose | Half-life | Notes |
|---|---|---|---|---|
| 1st | Tolbutamide | 500 mg TID (max 3g/d) | 4–5 h | Safe in elderly/renal impairment |
| 1st | Chlorpropamide | 100–500 mg/d | 32 h | Avoid in elderly; SIADH risk |
| 1st | Tolazamide | 100–1000 mg/d | 7 h | Slower absorption |
| 2nd | Glipizide | 5–40 mg/d | 4 h | Lower hypoglycemia risk |
| 2nd | Glyburide (glibenclamide) | 1.25–20 mg/d | 10 h | Hepatic + biliary excretion |
| 2nd | Glimepiride | 1–4 mg/d | 5 h | Once daily; least hypoglycemia |
Key Points
- Risk of hypoglycemia is the primary adverse effect; especially with chlorpropamide and glyburide
- Metabolized by liver; metabolites renally excreted
- Second-generation agents have higher receptor affinity → lower doses → fewer drug interactions
- Contraindicated in Type 1 DM, pregnancy, severe hepatic/renal disease
- UKPDS confirmed no excess cardiovascular mortality with sulfonylureas
2. Meglitinides (Non-sulfonylurea Secretagogues)
Agents
- Repaglinide (0.5–4 mg before meals)
- Nateglinide (60–120 mg before meals)
Mechanism
Same receptor/channel as sulfonylureas but bind at a different site. Very rapid onset and short duration → reduce postprandial hyperglycemia. Must be taken with meals; skipping a meal = skip the dose.
Adverse Effects
Hypoglycemia (less than sulfonylureas); repaglinide metabolized entirely by liver (safe in renal impairment).
3. Biguanides — Metformin
Mechanism
Metformin's primary action: inhibits hepatic gluconeogenesis (reduces hepatic glucose output). Secondary: improves peripheral insulin sensitivity, reduces intestinal glucose absorption. Mechanism involves activation of AMP-activated protein kinase (AMPK) and inhibition of mitochondrial complex I.
Pharmacokinetics
- Not metabolized; excreted unchanged by kidney
- Half-life ~6 hours; 500–2550 mg/day in divided doses
- No hypoglycemia (does not stimulate insulin secretion)
- Weight-neutral or causes mild weight loss
Adverse Effects
- GI: nausea, diarrhea, abdominal discomfort (start low, titrate up)
- Lactic acidosis (rare but serious) — risk increases if renal/hepatic impairment, excessive alcohol, contrast media
- Reduces vitamin B12 absorption (long-term use)
Contraindications
- eGFR <30 mL/min/1.73 m² (avoid); reduce dose if eGFR 30–45
- Hold before IV contrast dye
- First-line agent for Type 2 DM per ADA/ACP guidelines
4. Thiazolidinediones (TZDs / Glitazones)
Agents
- Pioglitazone (15–45 mg/day)
- Rosiglitazone (4–8 mg/day; limited use due to CV concerns)
Mechanism
Bind PPARγ (peroxisome proliferator-activated receptor gamma) in adipose, muscle, and liver → increase insulin sensitivity, redistribute fat from visceral to subcutaneous, increase GLUT4 expression. They are insulin sensitizers — require insulin to work.
Key Points
- Slow onset of full effect (6–12 weeks)
- Cause fluid retention → edema, contraindicated in heart failure (NYHA Class III/IV)
- Rosiglitazone: increased risk of MI (FDA black box warning; restricted use)
- Pioglitazone: possible bladder cancer risk (long-term use); however, reduces cardiovascular events
- Cause weight gain; bone fractures in women
5. Alpha-Glucosidase Inhibitors
Agents
- Acarbose (25–100 mg TID with meals)
- Miglitol (25–100 mg TID with meals)
Mechanism
Competitively inhibit intestinal α-glucosidase and pancreatic α-amylase enzymes → delay carbohydrate digestion and glucose absorption → blunt postprandial glucose spike.
Key Points
- No hypoglycemia as monotherapy
- GI side effects common: flatulence, bloating, diarrhea (fermentation of undigested carbs in colon)
- If hypoglycemia occurs (e.g., combined with sulfonylurea), treat with glucose (not sucrose — sucrose absorption also blocked)
- Modest HbA1c reduction (~0.5–0.8%)
6. DPP-4 Inhibitors (Gliptins)
Agents
Sitagliptin, saxagliptin, linagliptin, alogliptin, vildagliptin
Mechanism
Inhibit dipeptidyl peptidase-4 (DPP-4) → prevent degradation of endogenous GLP-1 and GIP → increased incretin levels → glucose-dependent insulin secretion + suppression of glucagon.
Key Points
- Weight-neutral; low risk of hypoglycemia
- HbA1c reduction: ~0.5–1.0%
- Linagliptin: primarily biliary excretion — no dose adjustment in renal failure
- Others: renal dose adjustment required
- Adverse effects: nasopharyngitis, possible pancreatitis, joint pain (FDA warning)
- Saxagliptin/alogliptin: slightly increased heart failure risk (post-marketing data)
7. SGLT2 Inhibitors (Gliflozins)
Agents
- Empagliflozin, Dapagliflozin, Canagliflozin, Ertugliflozin
Mechanism
Block sodium-glucose co-transporter 2 (SGLT2) in the proximal renal tubule → prevent renal glucose reabsorption → glycosuria → lower plasma glucose. Also increase sodium excretion.
Key Points
- Weight loss (caloric loss via urine) + BP reduction
- Cardiovascular benefit: reduce mortality and hospitalization for heart failure — empagliflozin approved for HFrEF and HFpEF; dapagliflozin for HFrEF
- Renoprotective: slow progression of diabetic kidney disease
- Adverse effects: urinary tract infections, genital mycotic infections (glycosuria creates favorable environment), volume depletion, rare euglycemic diabetic ketoacidosis (in Type 1 or perioperative)
- Do not initiate if eGFR <20 mL/min/1.73 m²; some agents require eGFR ≥45 for glucose-lowering efficacy
8. GLP-1 Receptor Agonists (Incretin Mimetics)
Strictly injectable, but oral semaglutide is now available:
Agents
- Injectable: exenatide, liraglutide, dulaglutide, semaglutide (SC), albiglutide
- Oral: semaglutide (Rybelsus)
Mechanism
Mimic GLP-1 → stimulate glucose-dependent insulin secretion, inhibit glucagon, slow gastric emptying, suppress appetite.
Key Points
- Significant weight loss
- Cardiovascular benefit: liraglutide and semaglutide reduce MACE (MI, stroke, CV death); approved for HFrEF
- Adverse effects: nausea, vomiting (especially early); pancreatitis risk; contraindicated in personal/family history of MEN2 or medullary thyroid cancer
- HbA1c reduction: 1.0–1.5%
Summary Table
| Class | Mechanism | Hypoglycemia | Weight | Key Benefit | Key Risk |
|---|---|---|---|---|---|
| Sulfonylureas | ↑ insulin secretion (K⁺ channel) | Yes | ↑ | Potent, inexpensive | Hypoglycemia |
| Meglitinides | ↑ insulin secretion (postprandial) | Low | ↑ | Flexible dosing | Short duration |
| Metformin | ↓ hepatic gluconeogenesis | No | ↓/neutral | First-line, CVD neutral | Lactic acidosis (rare) |
| TZDs | PPARγ → ↑ insulin sensitivity | No | ↑ | Durable effect, pioglitazone CV+ | Edema, fractures |
| α-Glucosidase inhibitors | Delay carb digestion | No | Neutral | Postprandial glucose | GI side effects |
| DPP-4 inhibitors | ↑ incretin (GLP-1/GIP) | No | Neutral | Well tolerated | Nasopharyngitis |
| SGLT2 inhibitors | ↑ glucosuria | No | ↓ | CV + renal protection | Genital infections, DKA |
| GLP-1 agonists (oral) | Incretin mimetic | No | ↓↓ | Weight loss, CV benefit | Nausea, pancreatitis |
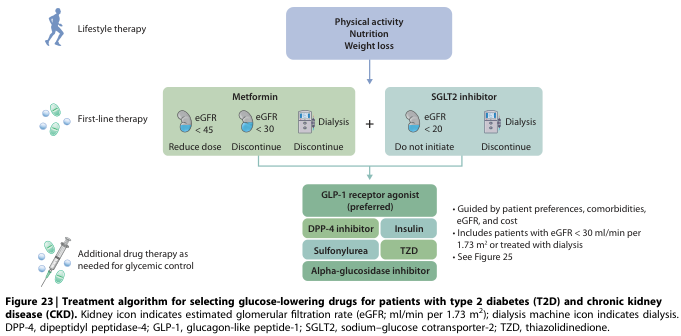
Clinical Approach to Drug Selection
- All patients: Metformin (first-line) + lifestyle modification
- With ASCVD or high CV risk: Add GLP-1 RA (liraglutide, semaglutide) or SGLT2 inhibitor
- With heart failure: Add SGLT2 inhibitor (empagliflozin/dapagliflozin)
- With CKD: Add SGLT2 inhibitor (if eGFR ≥20) for renoprotection
- Need to minimize hypoglycemia: DPP-4 inhibitors, GLP-1 RA, SGLT2i, TZDs
- Need weight loss: GLP-1 RA or SGLT2 inhibitor
- Cost-sensitive: Sulfonylureas or metformin (generic)
Sources: Katzung's Basic and Clinical Pharmacology, 16th Edition — Chapter 41; Goldman-Cecil Medicine, International Edition
This is a shared conversation. Sign in to Orris to start your own chat.