indication of triple therapy in Rheumatoid arthritis
Reading File
Finding Sources
Finding Sources
Searching PubMed
"triple therapy" AND "rheumatoid arthritis" AND methotrexate
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of Triple Therapy in Rheumatoid Arthritis:
What Is Triple Therapy?
Triple therapy in RA refers to the combination of three conventional (synthetic) DMARDs:
| Drug | Class |
|---|---|
| Methotrexate (MTX) | Folate antagonist / anchor DMARD |
| Sulfasalazine (SSZ) | Aminosalicylate |
| Hydroxychloroquine (HCQ) | Antimalarial |
Indications
1. Inadequate Response to Methotrexate Monotherapy
The primary indication. When a patient with RA has moderate to high disease activity despite methotrexate monotherapy, triple therapy is indicated before escalating to biologics — particularly when poor prognostic factors are absent.
"If disease activity remains high, then methotrexate can be combined with hydroxychloroquine and/or sulfasalazine or combined with a biologic DMARD." — Goldman-Cecil Medicine
2. Alternative to MTX + Biologic (TNF Inhibitor)
Triple therapy is considered as effective as methotrexate + a TNF inhibitor (e.g., etanercept) in controlling clinical disease activity, making it the preferred cost-effective option when biologics are not accessible, affordable, or appropriate.
"Triple therapy with methotrexate, sulfasalazine, and hydroxychloroquine appears to be as effective, clinically, as etanercept and methotrexate." — Katzung's Basic and Clinical Pharmacology, 16th Ed.
"Sulfasalazine is a component of 'triple therapy' (along with hydroxychloroquine and methotrexate), which has been shown to be as effective as methotrexate plus a TNF inhibitor." — Goldman-Cecil Medicine
3. Phase II Treatment in ACR/EULAR Treat-to-Target Strategy
Per the treatment algorithm, if Phase I (methotrexate ± glucocorticoids) fails to achieve target at 6 months and poor prognostic factors are absent, the algorithm calls for adding/switching to a second csDMARD — with the most frequently used combination being MTX + SSZ + HCQ (triple therapy).
4. Cost-Conscious or Biologic-Contraindicated Patients
Triple therapy is significantly more economical than biologic therapy and is preferred when:
- Biologics are cost-prohibitive
- Patient has contraindications to biologic agents (e.g., active/latent infections, certain malignancies)
- The patient/physician prefers to exhaust csDMARD options before initiating biologics
Where Triple Therapy Fits in the Treatment Algorithm
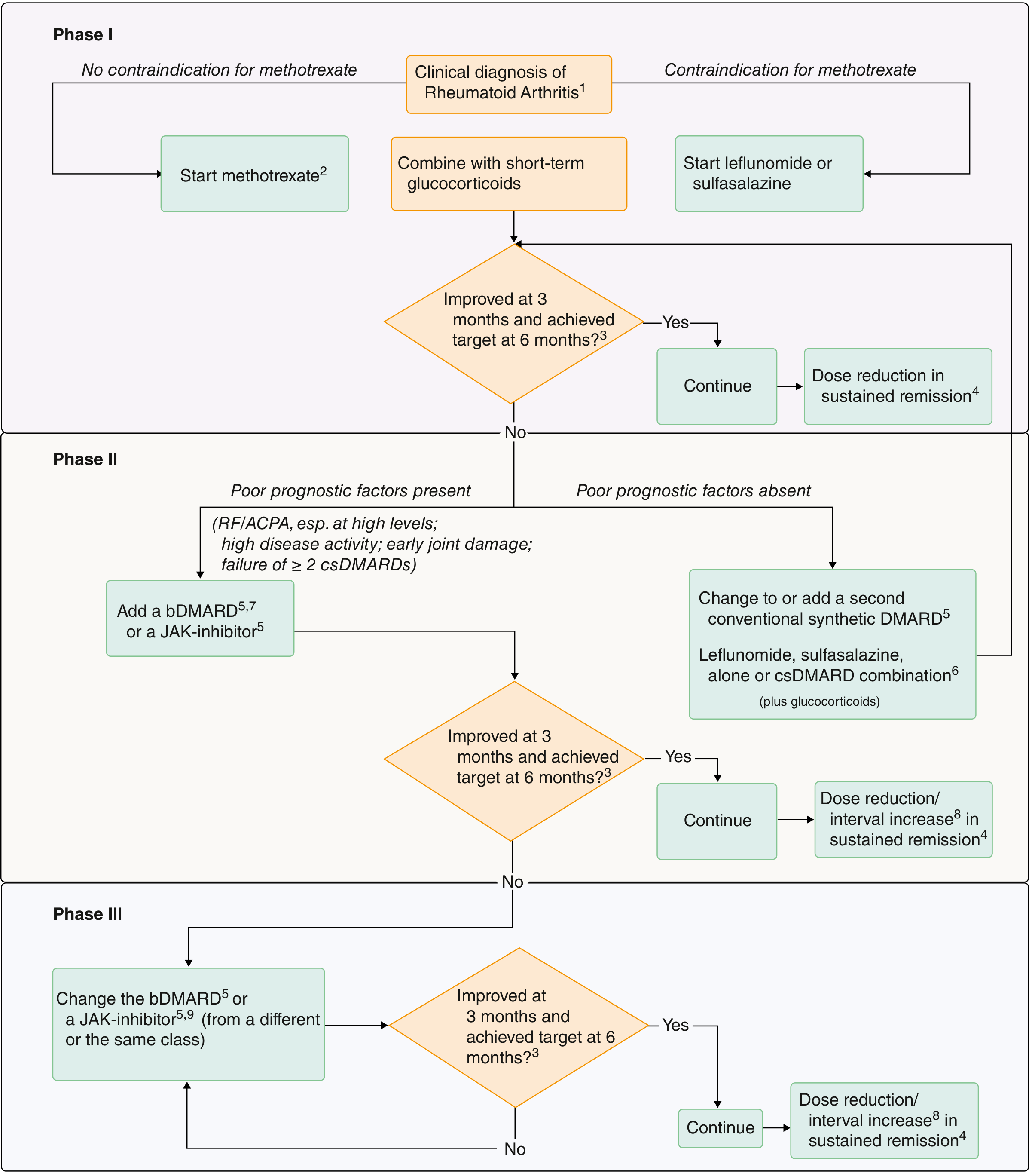
- Phase I: Start methotrexate (± short-term glucocorticoids)
- Phase II (no poor prognostic factors): Change to or add a second csDMARD → triple therapy (MTX + SSZ + HCQ) is the most frequently used combination (footnote 6 in the figure)
- Phase II (poor prognostic factors present): Add a bDMARD or JAK inhibitor instead
Poor prognostic factors that favor skipping directly to biologics include: high RF/ACPA titers, high disease activity, early joint damage, or failure of ≥ 2 csDMARDs.
Key Evidence
- The TEAR trial (Treatment of Early Aggressive RA) demonstrated that oral triple therapy was comparable to MTX + etanercept as initial therapy in early aggressive RA — cited in Harrison's Principles of Internal Medicine 22E (2025).
- The 2015 ACR guidelines strongly recommend combination traditional DMARDs for patients with moderate or high disease activity refractory to DMARD monotherapy (Katzung's, 16th Ed.).
- However, radiologic outcomes with triple therapy may not be as favorable as with MTX + biologic, which is a consideration in patients with high erosive burden.
Important Caveats
| Point | Detail |
|---|---|
| Radiologic outcomes | Clinical efficacy is similar to MTX + TNF-inhibitor, but radiographic progression may not be as well controlled |
| SSZ + MTX alone | Sulfasalazine or azathioprine plus methotrexate (without HCQ) does not add therapeutic benefit — the three-drug combination is what confers efficacy |
| Monitoring | MTX: CBC + LFTs every 8–12 weeks; SSZ: CBC monthly for 6 months; HCQ: ophthalmology screening after 5 years |
| Pregnancy | MTX is absolutely contraindicated in pregnancy — triple therapy must be modified |
Sources: Goldman-Cecil Medicine (International Ed.), Katzung's Basic and Clinical Pharmacology 16th Ed., Harrison's Principles of Internal Medicine 22E (2025)
This is a shared conversation. Sign in to Orris to start your own chat.