Unsafe Chronic Suppurative Otitis Media
Reading File
Finding Sources
Finding Sources
Searching PubMed
cholesteatoma chronic suppurative otitis media
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Searching Images
cholesteatoma middle ear otoscopy
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
I now have all the information needed for a comprehensive answer. Here is the full clinical overview:
Unsafe Chronic Suppurative Otitis Media (CSOM)
Definition & Terminology
Unsafe CSOM — also called CSOM with cholesteatoma — refers to chronic middle ear disease complicated by a cholesteatoma: an epidermal inclusion of keratinizing squamous epithelium within the middle ear cleft or mastoid. It is "unsafe" because cholesteatoma has an inherent destructive capacity, eroding bone and causing life-threatening intracranial complications. This distinguishes it from "safe" CSOM (tubotympanic/mucosal type) which has a central perforation without bone erosion.
Classification of CSOM
| Feature | Safe CSOM (Mucosal/Tubotympanic) | Unsafe CSOM (Squamosal/Atticoantral) |
|---|---|---|
| Perforation site | Central (pars tensa) | Attic/posterosuperior (pars flaccida) |
| Cholesteatoma | Absent | Present |
| Discharge | Mucoid, odourless | Scanty, foul-smelling, purulent |
| Bone erosion | Absent | Present |
| Complications | Rare | Common and serious |
| Treatment | Medical ± myringoplasty | Surgery mandatory |
Cholesteatoma: Structure & Composition
A cholesteatoma consists of:
- An outer matrix of keratinizing squamous epithelium
- A perimatrix (fibrous subepithelial layer rich in inflammatory cells)
- Keratin debris (desquamated squames) at the centre
The term was coined by Johannes Müller (1838) because white-yellow keratin flakes grossly resemble cholesterol crystals — though cholesterol content is actually minimal. Cruveilhier earlier described it as a "pearly tumour" of the temporal bone.
Types
1. Congenital Cholesteatoma
- Behind an intact tympanic membrane with no prior otitis or surgery
- Appears as white mass anteromedial to malleus
2. Primary Acquired Cholesteatoma (most common unsafe CSOM)
- Arises from retraction pocket of pars flaccida (attic retraction) due to Eustachian tube dysfunction
- Also called attic/epitympanic cholesteatoma
3. Secondary Acquired Cholesteatoma
- Keratinizing epithelium migrates through a pre-existing perforation of pars tensa into the middle ear
Pathogenesis
Four major theories — these are not mutually exclusive:
A. Retraction Pocket / Invagination Theory (most accepted)
Chronic Eustachian tube dysfunction → negative middle ear pressure → inward retraction of pars flaccida → accumulates keratin in a closed space → enlarging cholesteatoma. Bacteria infect the keratin matrix forming biofilms, perpetuating epithelial proliferation and invasion.
B. Epithelial Invasion (Migration) Theory
Keratinizing squamous epithelium from the EAC surface migrates through a perforation margin into the middle ear. Supported by cytokeratin 10 expression studies and animal models.
C. Basal Cell Hyperplasia Theory
Basal cells of the pars flaccida actively invade the subepithelial tissue and middle ear through micro-breaks in the basement membrane, driven by inflammatory stimuli.
D. Mucosal Traction Theory
Opposing mucosal surfaces (medial tympanic membrane and lateral ossicles) mechanically propel pars flaccida tissue into the attic.
Mechanism of Bone Erosion
This is the key destructive feature distinguishing unsafe CSOM:
- Historically attributed to pressure necrosis — now refuted (cholesteatoma pressure ~1.3–11.9 mm Hg, well below capillary perfusion pressure of ~25 mm Hg)
- Current understanding: osteoclast-mediated resorption activated by inflammatory signalling
- The cholesteatoma perimatrix expresses RANKL (receptor activator of NF-κB ligand) on stromal cells and activated T-cells → triggers osteoclastogenesis
- Osteoclasts secrete acid phosphatase, collagenase, and acid proteases into the resorption lacunae
- The endochondral bone of the otic capsule is more resistant than the intramembranous bone of the middle ear and mastoid
- Enzymes involved include cathepsin-like proteases active at pH 4.0
Clinical Features
Symptoms
- Otorrhoea: scanty but offensive/foul-smelling (anaerobic bacteria in keratin matrix)
- Hearing loss: progressive conductive, occasionally mixed or sensorineural if labyrinth eroded
- Aural polyp: infected cholesteatoma may present as a bleeding polyp
- Otalgia: usually absent unless complicated
- Symptoms of complications (see below)
Signs (Otoscopy/Microscopy)
- Attic perforation or retraction pocket in the posterosuperior quadrant/pars flaccida
- Scutum erosion (lateral attic wall)
- Keratin/epithelial debris or "pearly white" mass within the retraction
- Granulation tissue or aural polyp obscuring view
- Crusting may mask the underlying pathology — thorough canal débridement is mandatory
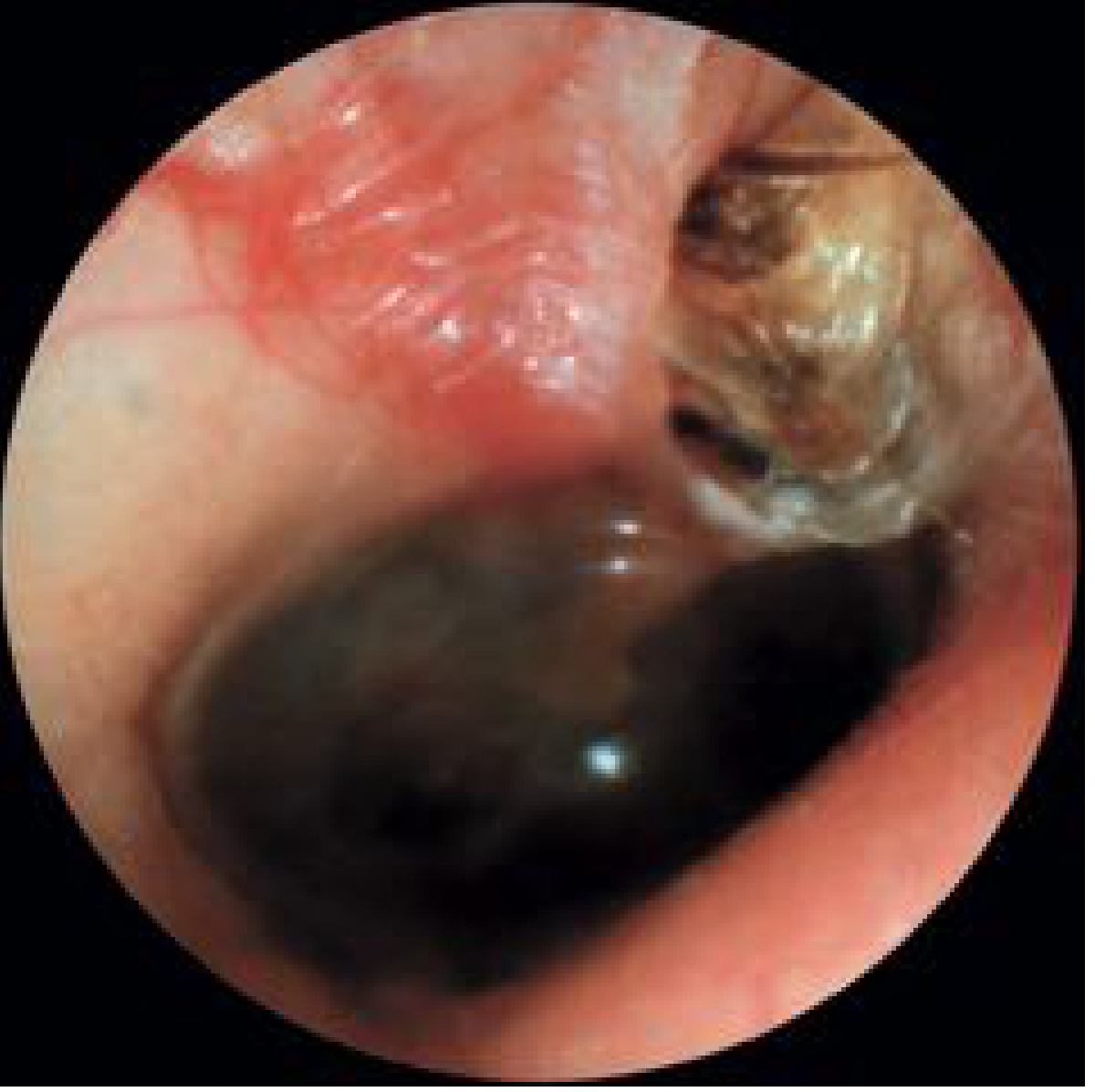
Otoscopic view: primary acquired cholesteatoma at pars flaccida with scutum erosion and keratin debris — Cummings Otolaryngology, Fig. 140.5
Investigations
| Investigation | Findings |
|---|---|
| Otoscopy/Microscopy | Attic retraction, keratin debris, scutum erosion, aural polyp |
| Pure Tone Audiometry | Conductive hearing loss (typically 40–60 dB); mixed/SNHL if labyrinth involved |
| HRCT Temporal Bone | Soft tissue opacity in attic/mastoid with bony erosion of scutum, ossicles, tegmen, sigmoid plate, labyrinth. CT does not confirm cholesteatoma but detects bone destruction |
| MRI (DWI) | Diffusion-weighted imaging (DWI-MRI, especially non-EPI DWI) now the gold standard for detecting residual/recurrent cholesteatoma post-surgery |
| Swab C&S | Anaerobes, Pseudomonas, Staph. aureus |
Complications
Complications are classified as extracranial (intratemporal) and intracranial:
Extracranial / Intratemporal
| Complication | Details |
|---|---|
| Conductive hearing loss | Ossicular chain erosion (long process of incus most common) |
| Sensorineural hearing loss | Labyrinthine erosion / toxic labyrinthitis |
| Labyrinthine fistula | Occurs in ~7–10% of cholesteatoma cases; horizontal (lateral) SCC most commonly affected (98%); presents with vertigo, positive fistula test |
| Facial nerve palsy | Erosion of bony fallopian canal — usually tympanic segment (up to 30% congenital dehiscence); progression from paresis → paralysis |
| Mastoiditis / Bezold's abscess | Extension through mastoid cortex |
| Petrositis (Gradenigo's syndrome) | Involvement of petrous apex; triad: otorrhoea, retroorbital pain, abducens palsy (VI nerve) |
| Ossicular chain erosion | Incus long process > stapes suprastructure > malleus handle |
Intracranial
| Complication | Details |
|---|---|
| Meningitis | Most common intracranial complication overall |
| Brain abscess | Temporal lobe (from tegmen erosion) or cerebellar (from sigmoid plate erosion) |
| Extradural abscess | Pus between dura and inner table |
| Subdural abscess / empyema | Less common |
| Lateral sinus thrombophlebitis | Thrombosis of sigmoid sinus |
| Otitic hydrocephalus | Raised ICP without ventricular dilation |
Management
Unsafe CSOM is a surgical disease. There is no definitive non-surgical treatment.
Pre-operative
- Aural toilet (canal débridement)
- Topical antibiotic drops (quinolone-based) to control active infection
- Systemic antibiotics if acutely infected or complication suspected
- CT temporal bone for surgical planning
- Audiometric assessment
Surgical Options
Canal Wall-Up (CWU) Mastoidectomy — Intact Canal Wall
- Posterior canal wall preserved
- Better cosmesis and hearing aid tolerance; faster healing
- Higher recidivism rate: 25–45% — often requires staged "second-look" at 6–12 months
- Preferred when disease is limited and follow-up is reliable
Canal Wall-Down (CWD) Mastoidectomy — Open Cavity
- Posterior canal wall removed → creates open mastoid bowl
- Better exposure of epitympanum and mesotympanum → easier complete disease removal
- Lower recidivism: 6–20%
- Requires long-term cavity debridement; bowl care issues (cerumen retention, infections)
- Preferred in: only-hearing ear, poor anesthetic risk, unreliable follow-up, extensive/recurrent disease, or when CWU leaves residual disease
| Feature | Canal Wall-Up | Canal Wall-Down |
|---|---|---|
| Recidivism | 25–45% | 6–20% |
| Second-look needed | Often yes | Usually not |
| Hearing aid use | Well tolerated | Problematic |
| Cavity care | Minimal | Long-term debridement |
| Anatomy | Preserved | Disrupted |
Radical Mastoidectomy
- Obliteration of the middle ear; combines mastoid and middle ear into one cavity
- Reserved for extensive, unresectable disease or poor surgical candidates
Modified Radical Mastoidectomy (Bondy)
- Canal wall-down with preservation of ossicular chain and intact tympanic membrane remnant
Intraoperative Considerations for Specific Complications
Labyrinthine fistula:
- Matrix over fistula carefully preserved during dissection
- Decision to use CWU vs CWD based on ability to completely eradicate cholesteatoma
Facial nerve palsy:
- Tympanomastoidectomy with removal of matrix from epineurium
- Decompression proximal and distal to the dehiscent segment
- Opening the epineurium only if cholesteatoma has invaded the nerve sheath
- Recovery to HB Grade I–II in 52–83% of patients
Post-operative Surveillance
- Regular follow-up with otoscopy/microscopy
- DWI-MRI at 12–18 months post-op to detect residual/recurrent disease (avoids second-look surgery in many centres)
- Audiological assessment and hearing rehabilitation as needed
- Hearing aids can be challenging in CWD cavities (↑ infection risk from mold occlusion and moisture trapping)
Key Points Summary
- Unsafe CSOM = CSOM with cholesteatoma — always associated with bone erosion and risk of serious complications
- Distinguishing feature: attic/posterosuperior perforation, foul-smelling scanty discharge, bony erosion on CT
- Bone destruction is osteoclast-mediated via RANKL/RANK pathway, not pressure necrosis
- Labyrinthine fistula occurs in ~7–10%; horizontal SCC is affected in 98% of cases
- Facial nerve palsy from cholesteatoma follows a slow progressive course
- Surgery is mandatory — CWU vs CWD choice balances recidivism risk against cavity morbidity
- Post-operative non-EPI DWI-MRI has replaced routine second-look surgery in many centres
Sources: Cummings Otolaryngology Head and Neck Surgery (Chapter 140, 143); K.J. Lee's Essential Otolaryngology
This is a shared conversation. Sign in to Orris to start your own chat.