Causes of acute paraparesis
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
I now have comprehensive material from multiple authoritative textbooks. Let me synthesize this into a well-organized answer.
Causes of Acute Paraparesis
Acute paraparesis (sudden-onset bilateral leg weakness) is a neurological emergency. The causes are best organized by anatomical level and mechanism.
Localizing the Level First
Before categorizing causes, the clinical pattern guides localization:
| Feature | Suggests |
|---|---|
| Flaccid legs, areflexia, sensory level | Spinal cord (acute "spinal shock" phase) |
| Flaccid legs, areflexia, no sensory level | Cauda equina or peripheral nerve/muscle |
| Spastic legs, sensory level | Spinal cord (UMN) |
| Sphincter involvement | Spinal cord or cauda equina |
| Dissociated sensory loss (pain/temp ↓, proprioception preserved) | Anterior cord syndrome |
I. Spinal Cord Causes (Most Common)
A. Compressive / Extrinsic
| Cause | Key Features |
|---|---|
| Spinal epidural abscess | Fever, back pain, progressive neurological deficit; percussion tenderness; MRI confirms; emergency decompression + IV antibiotics |
| Spinal epidural hematoma | Sudden severe radicular pain → weakness; associated with anticoagulation, trauma, coagulopathy, lumbar puncture; treat with surgical decompression |
| Acute disc herniation (midline, thoracic/cervical) | Acute cord compression; trauma or spontaneous |
| Metastatic spinal cord compression | Night pain worse lying flat; known or occult malignancy (breast, lung, prostate, myeloma); MRI urgent |
| Atlanto-axial subluxation | Rheumatoid arthritis, Down syndrome, trauma |
| Acute spondylotic myelopathy | Hyperextension injury on background of cervical spondylosis; pre-existing canal stenosis; ligamentum flavum buckling |
| Subdural hematoma / empyema | Rare; similar presentation to epidural |
B. Intrinsic / Noncompressive — Vascular
| Cause | Key Features |
|---|---|
| Spinal cord infarction (anterior spinal artery syndrome) | Hyperacute onset; paraplegia/paraparesis + spinothalamic loss (pain & temp ↓) with preserved dorsal columns (proprioception intact); causes include aortic atherosclerosis, aortic dissection, aortic surgery, vertebral artery dissection/occlusion, cardiogenic emboli, vasculitis, profound hypotension; MRI brain negative for bilateral ACA infarcts |
| Spinal AVM / arteriovenous fistula | Dural AVF most common in older men; stepwise or acute myelopathy |
| Hematomyelia (intramedullary hemorrhage) | Acute painful transverse myelopathy; causes: trauma, vascular malformation, vasculitis (polyarteritis nodosa, SLE), bleeding disorders, cord neoplasm |
| Spinal subarachnoid hemorrhage | Focal deficits at bleed level, headache possible; MRI ± LP |
C. Intrinsic / Noncompressive — Inflammatory / Demyelinating
| Cause | Key Features |
|---|---|
| Acute transverse myelitis (idiopathic) | Paraplegia + sensory level + sphincter dysfunction; follows viral illness in ~30%; thoracic cord in 60–70%; MRI T2 signal change ± enhancement |
| Multiple sclerosis | Partial or complete cord syndrome; relapsing history; MRI shows other lesions; CSF oligoclonal bands |
| Neuromyelitis optica spectrum disorder (NMOSD) | Anti-AQP4 or anti-MOG antibodies; longitudinally extensive lesion (≥3 segments); often severe |
| Sarcoidosis | Multisystem involvement; serum ACE, chest CT, biopsy |
| Systemic lupus erythematosus (SLE) | Myelopathy in known or new SLE; antiphospholipid syndrome overlap |
| Behçet's disease | Orogenital ulcers, uveitis; brainstem/cord involvement |
| Acute disseminated encephalomyelitis (ADEM) | Post-infectious/post-vaccination; multifocal CNS; commoner in children |
D. Intrinsic / Noncompressive — Infectious
| Cause | Key Features |
|---|---|
| Viral myelitis — HSV-2, VZV, EBV, CMV, enterovirus, HIV, HHV-6 | CSF PCR; HIV myelopathy in advanced disease |
| HTLV-1 myelopathy (tropical spastic paraparesis) | Subacute > acute; endemic regions; serology |
| West Nile virus | Acute flaccid paralysis (anterior horn cells); fever, encephalitis possible |
| Poliomyelitis | Acute flaccid paraparesis; anterior horn cell |
| Schistosomiasis | Endemic areas; lumbosacral cord; granulomatous myelitis |
| Neurosyphilis | Meningovascular; CSF VDRL |
| Lyme disease | Borrelia; radiculomyelopathy |
| Tuberculous myelitis / Pott's disease | Vertebral destruction, epidural spread; endemic setting |
II. Cauda Equina / Conus Medullaris
These produce lower motor neuron weakness (flaccid, areflexic) with saddle anaesthesia and prominent sphincter involvement:
- Massive midline lumbar disc prolapse — most common; acute back pain + bilateral leg weakness + urinary retention
- Cauda equina tumor (ependymoma, schwannoma, meningioma)
- Lumbosacral trauma
- HIV-related acute lumbosacral polyradiculopathy (CMV in immunocompromised)
- Spinal epidural abscess/hematoma at lumbar level
III. Cerebral / Supratentorial Causes (Rare)
Bilateral leg weakness with upper motor neuron signs but no sensory level:
| Cause | Key Features |
|---|---|
| Bilateral ACA territory infarction | Shoulder shrug (shoulder abduction) also affected |
| Superior sagittal sinus / cortical venous thrombosis | Headache, seizures, bilateral leg weakness from parasagittal involvement |
| Acute hydrocephalus | Bilateral leg weakness (stretching of periventricular fibers) |
| Parasagittal meningioma | More subacute; bilateral leg signs |
IV. Peripheral / Neuromuscular Causes
Can mimic paraparesis if bilateral and severe — absence of sensory level and UMN signs distinguishes:
| Cause | Features |
|---|---|
| Guillain-Barré syndrome | Ascending flaccid areflexic weakness; albuminocytologic dissociation in CSF |
| Acute anterior horn cell disease — Polio, West Nile | Fever + asymmetric flaccid paralysis |
| Myopathy / periodic paralysis | Metabolic (K⁺, Ca²⁺, phosphate, Mg²⁺ disturbances); no sensory loss; no reflexes lost early |
| Toxic myopathy / botulism | Descending paralysis (botulism); rhabdomyolysis |
Approach Summary
Acute paraparesis
├── Sensory level / UMN signs → Spinal cord MRI STAT
│ ├── Compressive? → Surgery/Antibiotics emergency
│ └── Noncompressive? → LP + autoimmune/infectious workup
├── No sensory level, flaccid/areflexic → Cauda equina vs. GBS vs. myopathy
└── Bilateral leg UMN + brain signs → Brain MRI (ACA infarct, SST)
MRI spine with contrast is the investigation of first choice in all cases where a spinal cord lesion is suspected. LP for CSF analysis follows once cord compression is excluded.
Sources: Harrison's Principles of Internal Medicine 22E (2025); Rosen's Emergency Medicine 10E; Bradley and Daroff's Neurology in Clinical Practice; Adams and Victor's Principles of Neurology 12E
55 Yr 2 episode of seizures and altered sensorium approach to pt .. history and examination
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Loading Image
I now have comprehensive, textbook-level material. Let me compile the full structured answer.
Approach to a 55-Year-Old with 2 Episodes of Seizures + Altered Sensorium
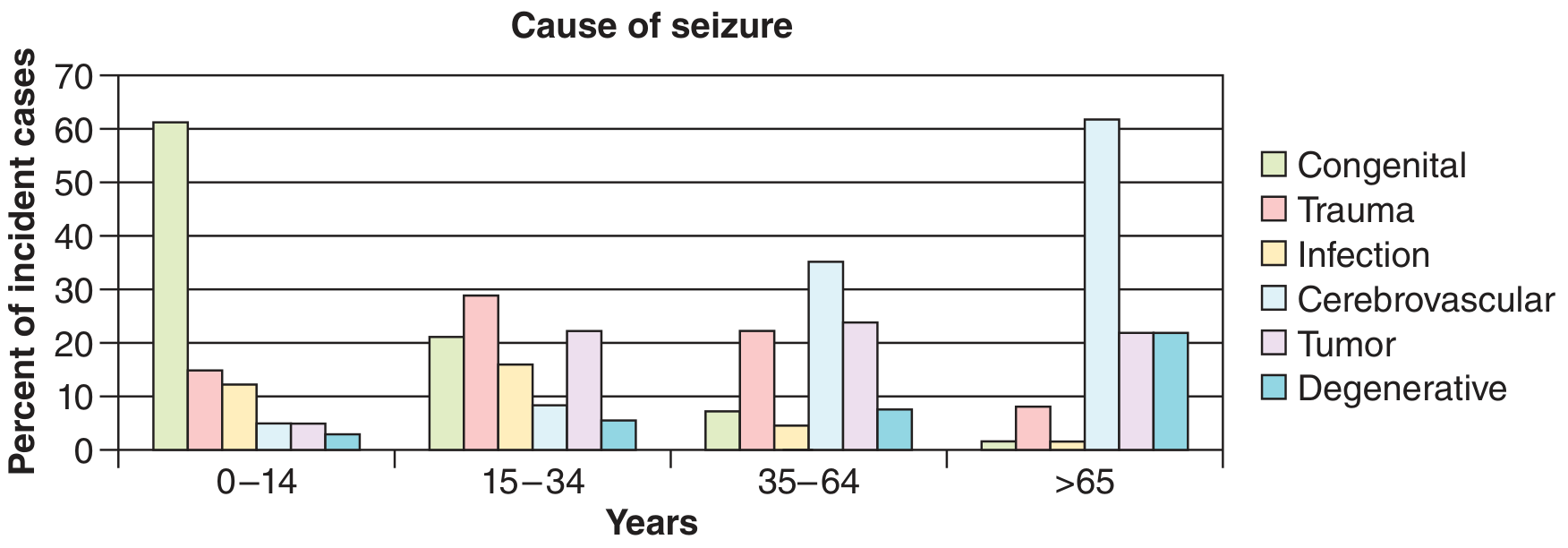
A 55-year-old presenting with new-onset seizures and altered sensorium is a neurological emergency until proven otherwise. In this age group (35–64 years), cerebrovascular disease is the single most common cause (~35%), followed by tumors (~24%), trauma (~22%), and metabolic/toxic causes. The altered sensorium adds urgency — it suggests either ongoing postictal state, status epilepticus, structural brain injury, meningoencephalitis, or a metabolic encephalopathy.
Step 1 — Initial Stabilization (Before History)
ABC + rapid bedside assessment:
- Airway, breathing, circulation
- Capillary blood glucose immediately (hypoglycemia is the most treatable cause)
- IV access, pulse oximetry, cardiac monitor
- GCS score
- Is the patient still seizing? → If so, manage as status epilepticus
Step 2 — HISTORY
A. Confirm It Was a Seizure (Not a Mimic)
From witness (paramount):
- Onset: sudden or gradual?
- Type of movements: tonic? clonic? tonic-clonic? focal limb jerking? head/eye deviation?
- Duration of episode (exact)
- Was there a cry/vocalization at onset?
- Did the patient fall? Sustain injury?
- Skin color: cyanosis, pallor?
- Loss of consciousness: complete or partial?
- Tongue biting (lateral tongue bite → seizure; tip bite → syncope)
- Incontinence (urinary/fecal)
- Post-episode behavior: confusion, drowsiness, Todd's paralysis, headache, amnesia?
Mimics to exclude:
- Syncope (preceded by prodrome — nausea, diaphoresis, pallor; recovery rapid)
- Cardiac arrhythmia (ventricular tachycardia; palpitations; ECG abnormality)
- TIA (loss of function, not spreading excitation; no post-ictal confusion)
- Hypoglycemic episode
- Psychogenic non-epileptic seizures (PNES)
- Migraine with aura
B. Characterize the Seizure Type
- Focal onset (aware or impaired awareness) vs. generalized → critical for workup
- Any aura beforehand? Olfactory, gustatory, déjà vu, rising epigastric feeling → temporal lobe
- Visual/sensory aura → occipital/parietal
- Second episode — same or different semiology? What interval?
- Did patient have eye/head deviation at onset? → indicates contralateral frontal focus
C. History of Present Illness
- Fever, headache, neck stiffness, photophobia → meningoencephalitis (urgent)
- Headache (progressive, worse lying flat) → raised ICP, SOL
- Focal weakness, speech disturbance, vision change → structural lesion
- Vomiting → raised ICP
- Recent head trauma (even trivial) → subdural/extradural hematoma
- Altered behavior/personality change over weeks → tumor, encephalitis (autoimmune/viral)
- Fever + rash → HSV encephalitis, bacterial meningitis, CNS vasculitis
- Polyuria, polydipsia → hypo-/hyperglycemia, hyponatremia
- Recent illness / vaccination → ADEM
- Recent travel → neurocysticercosis (endemic areas), cerebral malaria, TB
- Timeline of altered sensorium relative to seizures
D. Past Medical History
| Ask about | Relevance |
|---|---|
| Hypertension, diabetes, CAD, atrial fibrillation | Stroke, hypertensive encephalopathy |
| Prior stroke, TIA | Cortical scar → epileptic focus |
| Known malignancy (breast, lung, colon, kidney, melanoma) | Cerebral metastasis |
| HIV, immunocompromise | CNS lymphoma, CMV, toxoplasmosis, TB, cryptococcal |
| Liver disease (cirrhosis, hepatitis) | Hepatic encephalopathy |
| Renal disease | Uremic encephalopathy, hyponatremia |
| Thyroid disease | Hashimoto's encephalopathy; hypo/hyperthyroidism |
| Autoimmune disease (SLE, vasculitis) | CNS vasculitis |
| Prior seizures in childhood (febrile convulsions) | Lowers threshold |
| Prior brain surgery or CNS infection | Scar epilepsy |
E. Drug and Substance History
| Ask about | Relevance |
|---|---|
| Alcohol history — amount, last drink | Withdrawal seizures (48–72 h after cessation) |
| Benzodiazepines, barbiturates — recent reduction/stop | Withdrawal |
| Recreational drugs (cocaine, amphetamines) | Provoked seizures |
| Prescribed medications | Proconvulsant drugs (tramadol, tricyclics, fluoroquinolones, isoniazid, theophylline, lithium, clozapine) |
| Recent change in antiepileptic drug (AED) | Sub-therapeutic levels |
| Herbal / traditional medicines | CNS toxicity |
F. Family and Social History
- Family history of epilepsy
- Occupation (driver? — implications for treatment)
- Lives alone?
- Sleep deprivation, extreme physical or emotional stress
Step 3 — EXAMINATION
General Examination
| Sign | Suggests |
|---|---|
| Fever | Meningoencephalitis, brain abscess, septic encephalopathy |
| High BP (>180/120) | Hypertensive encephalopathy, posterior reversible encephalopathy syndrome (PRES) |
| Low BP | Sepsis, cardiogenic cause, Addisonian crisis |
| Tachycardia | Cardiac arrhythmia, sepsis, thyroid storm |
| Cyanosis / low SpO₂ | Hypoxic seizure |
| Pallor | Anaemia, blood loss (subdural hematoma) |
| Jaundice | Hepatic encephalopathy |
| Stigmata of chronic liver disease | Hepatic encephalopathy |
| Lymphadenopathy | Lymphoma, malignancy, TB |
| Skin rash | Meningococcal purpura, SLE, HSV, neurofibromatosis, tuberous sclerosis |
| Head trauma signs: Battle's sign, raccoon eyes, haemotympanum, CSF leak | Base of skull fracture → intracranial bleed |
| Tongue laceration (lateral) | Confirms seizure |
| Incontinence signs | |
| Needle marks / drug track marks | Substance abuse |
Neurological Examination (Systematic)
1. Level of Consciousness & Cognition
- GCS (E+V+M) — document precisely
- MMSE / orientation — time, place, person
- Is this true postictal drowsiness or new encephalopathy?
- Postictal state: usually improves steadily over 20–60 min; persisting confusion → structural/metabolic
2. Meningeal Signs
- Neck stiffness (Kernig's sign, Brudzinski's sign) → meningitis, SAH, encephalitis
- Note: may be absent in immunocompromised / elderly
3. Cranial Nerves
| CN | What to look for |
|---|---|
| CN II | Papilloedema (raised ICP), visual field defect |
| CN III, IV, VI | Pupil asymmetry (herniation!), gaze palsy, VI palsy (false localizing, raised ICP) |
| CN VII | Facial asymmetry → frontal lobe lesion (contralateral) |
| CN XII | Tongue deviation |
4. Motor System
- Focal weakness → contralateral structural lesion
- Todd's paralysis — transient hemiparesis after focal seizure (30 min – 36 h); clears spontaneously
- Tone: hypotonia (metabolic/toxic), spasticity (structural)
- Pronator drift
- Plantar response (upgoing Babinski → UMN lesion or postictal)
5. Sensory System
- Hemisensory loss → contralateral parietal lesion
6. Cerebellar
- Ataxia, nystagmus → posterior fossa lesion, drug toxicity (phenytoin, carbamazepine)
7. Deep Tendon Reflexes
- Hyperreflexia + Babinski → UMN lesion
- Hyporeflexia → metabolic, drug effect
8. Fundoscopy (MANDATORY)
- Papilloedema → raised ICP → LP contraindicated before imaging
Differential Diagnosis Framework (55-Year-Old)
Seizures + Altered Sensorium at 55 yrs
│
├── STRUCTURAL (most common in this age)
│ ├── Stroke (ischemic / hemorrhagic)
│ ├── Subdural / extradural hematoma
│ ├── Brain tumor (primary glioma or metastasis)
│ └── Brain abscess / granuloma (TB, NCC)
│
├── METABOLIC / SYSTEMIC
│ ├── Hypo/hyperglycemia
│ ├── Hyponatremia (Na⁺ <125 mEq/L)
│ ├── Uremic encephalopathy
│ ├── Hepatic encephalopathy
│ ├── Hypocalcemia / hypomagnesemia
│ ├── Hypoxia / hypercapnia
│ └── Hypertensive encephalopathy / PRES
│
├── INFECTIVE / INFLAMMATORY
│ ├── Bacterial meningitis
│ ├── Viral encephalitis (HSV most important)
│ ├── Autoimmune encephalitis (anti-NMDAR, LGI1, CASPR2)
│ ├── TB meningoencephalitis
│ └── Cerebral malaria / neurocysticercosis
│
├── TOXIC / DRUG
│ ├── Alcohol withdrawal (48–72 h after last drink)
│ ├── Benzodiazepine/barbiturate withdrawal
│ └── Drug toxicity (proconvulsants)
│
└── VASCULAR
└── Cerebral venous sinus thrombosis (CVST)
Red Flags Demanding Immediate Action
| Red Flag | Action |
|---|---|
| Fever + meningism | LP after imaging → treat empirically for bacterial meningitis + HSV encephalitis BEFORE LP |
| Focal deficits / papilloedema | CT brain before LP |
| Ongoing altered sensorium >30 min post-seizure | Rule out non-convulsive status epilepticus → urgent EEG |
| Severe hypertension + encephalopathy | Treat as PRES / hypertensive emergency |
| Known malignancy | MRI with contrast |
| Anticoagulated / alcoholic / elderly | Exclude intracranial bleed |
| Glucose < 3 mmol/L | IV dextrose immediately |
Sources: Adams and Victor's Principles of Neurology 12E; Harrison's Principles of Internal Medicine 22E; Bradley and Daroff's Neurology in Clinical Practice
25 Yr female with wasting of left hand wasting with motor weakness 3 month without sensory involved Differential
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Excellent — rich data from multiple authoritative sources. Let me now compose the full structured differential.
25-Year-Old Female: Left Hand Wasting + Pure Motor Weakness (3 months, No Sensory Loss)
Key Diagnostic Features to Lock In
The clinical signature here is:
- Young female, 25 years
- Unilateral (left hand)
- Wasting + weakness (lower motor neuron pattern — flaccid, ± fasciculations, ± hyporeflexia)
- 3 months — subacute/chronic, not acute
- No sensory loss — this is the critical differentiating feature
The absence of sensory involvement localizes the pathology to structures that carry only motor fibers — anterior horn cells, motor roots, motor division of peripheral nerves — or to intrinsic cord lesions that selectively damage anterior horn cells (with sensory fibers initially spared).
Localization First
| Level | Pattern | Sensory |
|---|---|---|
| Anterior horn cell | Pure LMN wasting, fasciculations, no sensory | Absent (pure motor cells) |
| Anterior root (motor root) | Pure LMN, segmental distribution | Absent |
| Intrinsic cord (syrinx, early) | Dissociated sensory loss + LMN at level | May be absent early |
| Lower trunk / medial cord brachial plexus | C8-T1 wasting | Usually some sensory in medial forearm/hand |
| Pure motor peripheral nerve | Deep palmar ulnar, posterior interosseous | No sensory |
| Muscle | Wasting without reflex change early | No sensory |
Differential Diagnosis (Most Likely to Least in This Patient)
1. Hirayama Disease (Benign Monomelic Amyotrophy / Juvenile Segmental Muscular Atrophy) ⭐ TOP DIAGNOSIS
"The most common presentation is one of an idiopathic, slowly progressive, painless weakness and atrophy in one hand or forearm... The condition remains limited to only a few myotomes." — Bradley and Daroff's Neurology
Why top diagnosis:
- Classic age of onset: teens to early 30s
- More common in women in Indian subcontinent (though 60% are male overall)
- Predominantly C7–T1 myotomes → intrinsic hand muscles + forearm
- "Oblique atrophy" pattern — thenar, hypothenar, interossei wasted; brachioradialis spared
- Pure LMN, no sensory loss, no UMN signs, no cranial nerve involvement
- Painless, slow progression over 2–3 years then stabilizes
- Proposed mechanism: dynamic cord compression during neck flexion (disproportionate dural sac growth)
- EMG: active and chronic denervation at C7–T1; NCS show reduced CMAPs, near-normal SNAPs
- MRI cervical spine (dynamic — flexion views): flattening of anterior cord, loss of attachment of posterior dura, asymmetric cord atrophy
2. Cervical Myelopathy — Syringomyelia
"Syringomyelia... damage to the fibers crossing in the anterior commissure destined for the spinothalamic tract... By the time descending tracts are affected, segmental motoneuron and corticospinal tract damage are almost always present." — Bradley and Daroff's
Features:
- Syrinx classically causes cape-like dissociated sensory loss (pain/temp lost, proprioception preserved)
- However, early syrinx can present with pure hand wasting (anterior horn cell damage at the level of the syrinx) before sensory fibres are involved
- Associated with Chiari malformation (Type I most common), trauma, tumors, arachnoiditis
- May later develop UMN signs in legs (descending tract involvement)
- 25-year-old female + no sensory involvement yet → must exclude with MRI
- MRI spine: T2 fluid-filled cavity within cord over multiple segments
3. True Neurogenic Thoracic Outlet Syndrome (TOS)
"True neurogenic TOS... compression of the lower trunk (C8/T1)... weakness and wasting of intrinsic muscles of the hand and diminished sensation on the palmar aspect of the fifth digit." — Harrison's Principles
Features:
- Caused by anomalous fibrocartilaginous band from elongated C7 transverse process (cervical rib) to first rib
- C8/T1 compression → hand intrinsic wasting
- Pain is mild or absent — can initially appear as pure motor
- Sensory loss on 5th digit/medial forearm — may be minimal or absent early
- Young women, often exacerbated by arm position
- CXR: elongated C7 transverse process / cervical rib
- EMG/NCS: reduced SNAP medial forearm, low CMAP ulnar > median
- Treatment: surgical band resection (wasting usually does not recover but progression halts)
4. Multifocal Motor Neuropathy (MMN) with Conduction Block
"Pure LMN involvement... enters the differential diagnosis of benign focal amyotrophy and the PMA variant of ALS. It is important to search for this since it is treatable." — Bradley and Daroff's
Features:
- Autoimmune; anti-GM1 antibodies in ~50%
- Asymmetric, pure motor — starts distally, upper > lower limbs
- No sensory loss (distinguishes from CIDP)
- Fasciculations may be present
- Can begin with focal hand wasting in a young adult
- EMG/NCS: focal conduction block outside carpal tunnel → pathognomonic
- Treatable — high-dose IVIG; this makes the diagnosis essential to establish
- CK mildly elevated; anti-GM1 IgM elevated
5. Compressive Focal Motor Neuropathy
Pure motor syndromes can arise from compression of purely motor nerve branches:
| Nerve Compressed | Pattern |
|---|---|
| Deep palmar branch of ulnar nerve (at Guyon's canal — motor division) | Hypothenar + interossei wasting; no sensory loss |
| Posterior interosseous nerve (radial nerve deep branch) | Wrist/finger extension weakness; no sensory loss |
| Anterior interosseous nerve | Loss of pinch (FPL + FDP index); no sensory loss |
- Relevant in occupational/repetitive trauma
- EMG/NCS confirms level and type
6. Cervical Radiculopathy — C8/T1 Root (Predominantly Motor)
Features:
- Disc prolapse or osteophyte compressing C8 or T1 motor root
- Can produce hand intrinsic weakness and wasting
- Sensory involvement is typical but can be minimal or overlooked
- C8: ring/little finger, medial forearm sensory if affected; intrinsic + FDP weakness
- T1: intrinsic hand muscles (abductor pollicis brevis, interossei)
- MRI cervical spine confirms
- 25-year-old: less common but possible (disc herniation)
7. Pancoast Tumor (Superior Sulcus Tumor)
Features:
- Lung apex tumor invading lower brachial plexus (C8/T1)
- Progressive hand intrinsic wasting
- Horner's syndrome (ptosis, miosis, anhidrosis) — highly specific association
- Usually painful (shoulder/arm pain)
- Sensory involvement of medial forearm/hand expected
- 25-year-old female: rare but must not miss — especially if smoker
- CXR, CT thorax, MRI brachial plexus
8. Lower Brachial Plexopathy — Other Causes
- Radiation plexopathy (prior radiotherapy)
- Neoplastic infiltration (lymphoma, metastasis — less likely at 25)
- Neuralgic amyotrophy (Parsonage-Turner syndrome) — usually starts with severe pain, then weakness; sensory loss usually follows
9. Spinal Muscular Atrophy (SMA) — Adult Onset
Features:
- SMA Type III (Kugelberg-Welander) / Type IV: onset in adulthood possible
- Pure LMN, symmetric or asymmetric, no sensory loss
- Proximal > distal typically, but distal hand involvement can occur
- Slowly progressive
- Genetic: SMN1 deletion/mutation
- Creatine kinase may be elevated
10. Monomelic Motor Neuropathy — Hereditary (CMT Variants)
- Charcot-Marie-Tooth (CMT) — usually bilateral, but rare asymmetric presentations
- Distal hereditary motor neuropathy (dHMN): pure motor, distal wasting, no sensory loss
- Family history may be absent in sporadic cases
- Nerve conduction/genetic testing
Less Likely in a 25-Year-Old Female (but to Exclude)
| Condition | Reason to list |
|---|---|
| ALS | Very unlikely at 25, usually absent UMN signs in early hand-onset PMA variant; rule out |
| Lead poisoning | Pure motor neuropathy; radial nerve ("wrist drop"); occupational exposure |
| Porphyria | Acute intermittent porphyria → pure motor neuropathy; episodic; urine porphyrins |
| Inclusion body myositis | Older patients; long finger flexors + quadriceps; CK elevated |
| Focal myositis | Rare; MRI muscle |
Summary Table
| Diagnosis | Pure Motor? | Age / Sex | Key Feature | Investigation |
|---|---|---|---|---|
| Hirayama disease | ✅ | Young adult, F | Oblique atrophy, stabilizes, neck flexion aggravates | Cervical MRI (flexion views), EMG |
| Syringomyelia | ✅ early | Young adult | Dissociated sensory loss (may be absent early), Chiari | MRI spine |
| Neurogenic TOS | ✅ | Young female | Cervical rib, C7 transverse process | CXR, NCS, EMG |
| MMN | ✅ | Any age | Conduction block, anti-GM1 | NCS (conduction block), anti-GM1 |
| Deep palmar ulnar neuropathy | ✅ | Any | Motor-only branch compression | NCS, EMG |
| C8/T1 radiculopathy | Mostly | Any | Disc prolapse | MRI cervical spine |
| Pancoast tumor | ± | Any | Horner's, shoulder pain | CXR, CT thorax |
| SMA adult | ✅ | Any | SMN1 mutation | Genetic testing, EMG |
| dHMN | ✅ | Young | Family history, bilateral eventually | NCS, genetics |
Priority Investigations
- MRI cervical spine (with dynamic flexion views if Hirayama suspected) — exclude syrinx, Chiari, cord atrophy, disc herniation
- EMG / Nerve conduction studies — localizes to anterior horn vs. root vs. plexus vs. peripheral nerve; identifies conduction block (MMN), characterizes denervation pattern
- Chest X-ray / CT thorax — cervical rib, Pancoast tumor
- Anti-GM1 IgM antibodies — MMN
- Genetic testing (SMN1, PMP22) — if SMA or CMT suspected
- Urine porphyrins — if episodic or occupational exposure
- Serum CK — elevated in myopathic or neurogenic conditions
- Blood lead levels — if occupational risk
Sources: Bradley and Daroff's Neurology in Clinical Practice; Harrison's Principles of Internal Medicine 22E; Goldman-Cecil Medicine
Unilateral with pure motor only
The previous answer already covered this — the case is unilateral (left hand only) and pure motor (no sensory). Let me sharpen the differential specifically around that constraint and rank it more tightly.
Unilateral Hand Wasting — Pure Motor Only (No Sensory)
The unilateral + pure motor combination is the key constraint. It eliminates most polyneuropathies and most metabolic/systemic causes. The lesion must be in a structure carrying motor signals exclusively or predominantly.
Anatomical Logic: Where Can a Purely Motor, Unilateral Lesion Sit?
Cortex / Corticospinal tract
↓ (UMN — wasting rare, no fasciculations)
Anterior Horn Cell ←── Pure motor, unilateral = MOST SPECIFIC LEVEL
↓
Anterior (Motor) Root ←── Pure motor if dorsal root spared
↓
Brachial Plexus lower trunk / medial cord
↓ (sensory usually co-involved, but minimal early)
Pure Motor Peripheral Nerve branch
↓ (deep palmar ulnar, AIN, PIN)
Muscle ←── No sensory, but wasting pattern differs
The Focused Differential (Unilateral, Pure Motor, Young Female, Subacute)
1. ⭐ Hirayama Disease (Benign Monomelic Amyotrophy)
The single most likely diagnosis in this exact clinical scenario.
- 25-year-old, unilateral hand/forearm wasting, pure LMN, no sensory loss, no UMN signs, no pain
- C7–T1 anterior horn cell degeneration, confined to one limb
- "Oblique atrophy" — all intrinsic hand muscles + forearm flexors wasted, brachioradialis spared (C5/6 — above the lesion)
- Mini-polymyoclonus (finger tremor on extension) — characteristic
- Progression over 2–3 years then arrests spontaneously
- ♀ in Indian subcontinent: less gender bias than in Japan
- No fasciculations at rest typically (distinguishes from ALS)
Investigations:
- EMG: chronic + active denervation C7–T1, normal SNAPs
- MRI cervical spine in neutral AND flexion: asymmetric cord atrophy, loss of posterior dural attachment, anterior displacement of posterior dura on flexion, intramedullary T2 signal at C5–C8
2. Syringomyelia (Early / Eccentric Syrinx)
- An eccentric syrinx can selectively destroy one side's anterior horn cells before crossing fibres or dorsal columns are involved — producing unilateral pure motor wasting
- Associated with Chiari I malformation (common in young women), post-traumatic, idiopathic
- Sensory loss (dissociated — pain/temp only, cape distribution) typically follows later
- Should always be excluded in any young person with unilateral hand wasting
- MRI spine: T2 hyperintense CSF-filled cavity; look for tonsillar descent (Chiari)
3. Multifocal Motor Neuropathy (MMN)
- Pure motor, asymmetric, distal upper limb onset — classic presentation
- Mimics anterior horn cell disease but is demyelinating and treatable
- No sensory loss (distinguishes it from CIDP)
- Unilateral hand onset is well-described
- Anti-GM1 IgM antibodies (~50%)
- NCS: focal conduction block outside standard entrapment sites — pathognomonic
- Responds to high-dose IVIG — critical not to miss
4. Pure Motor Focal Neuropathy (Compressive/Entrapment)
These are purely motor nerve branches — no sensory fibres:
| Nerve | Site | Muscles Wasted | Clue |
|---|---|---|---|
| Deep (motor) branch of ulnar nerve | Guyon's canal (motor division only) | Hypothenar, interossei, adductor pollicis | No sensory loss, occupational/cycle/crutch pressure |
| Anterior interosseous nerve (AIN) | Proximal forearm | FPL, FDP (index), pronator quadratus | "Pinch sign" — can't make O with thumb+index |
| Posterior interosseous nerve (PIN) | Radial tunnel | Finger/wrist extensors | Wrist drop without sensory loss |
- Deep ulnar motor branch = most relevant for intrinsic hand wasting without sensory loss
- Cause: ganglion, repetitive pressure (cyclists, typists), occupational
5. Neurogenic Thoracic Outlet Syndrome (True TOS)
- Anomalous fibrocartilaginous band, elongated C7 transverse process / cervical rib → compresses lower trunk (C8/T1)
- Wasting of intrinsic hand muscles, predominantly motor
- Sensory loss (medial forearm, 5th digit) is classically mild or absent — can appear pure motor
- Young women are disproportionately affected
- CXR: elongated C7 transverse process
- NCS/EMG: reduced CMAP ulnar > median; reduced SNAP medial forearm (if sensory affected at all)
6. C8 or T1 Motor Root Compression (Radiculopathy — Motor Predominant)
- Cervical disc herniation compressing C8 or T1 root
- Dorsal (sensory) root can be spared if compression is purely anterior (ventral root lesion)
- Hand intrinsic wasting (T1) or FDP/FPL weakness (C8)
- Young adults: soft disc prolapse possible
- MRI cervical spine confirms
7. Spinal Muscular Atrophy (SMA) — Focal / Asymmetric Variant
- Adult-onset SMA (Type III/IV) or distal hereditary motor neuropathy (dHMN) can start asymmetrically
- Pure LMN, no sensory loss — by definition
- Family history may be absent
- SMN1 gene testing; EMG shows widespread chronic denervation beyond clinically affected limb
8. Monomelic Amyotrophy Secondary to Intrinsic Cord Lesion
- Intramedullary tumor (ependymoma, astrocytoma) at cervical cord level
- Selective anterior horn involvement before sensory tracts affected
- Rare but must be excluded with MRI
What Makes This NOT ALS
ALS is in the differential for any pure motor wasting, but in this patient:
| Feature | This Patient | ALS |
|---|---|---|
| Age | 25 ♀ | Usually >40 |
| Distribution | Unilateral hand, confined | Spreads relentlessly |
| UMN signs | Absent | Usually present (brisk reflexes, Babinski) |
| Bulbar | Absent | Develops |
| Progression | 3 months, stable hand | Relentless progression |
| Fasciculations | Not mentioned | Widespread |
ALS remains a diagnosis of exclusion — all other treatable causes must be systematically ruled out before considering it.
Ranked Summary
| Rank | Diagnosis | Key Distinguishing Feature |
|---|---|---|
| 1 | Hirayama disease | Young, oblique atrophy, arrests, dynamic MRI |
| 2 | Syringomyelia (early eccentric) | MRI cord signal, Chiari |
| 3 | MMN | Conduction block on NCS, anti-GM1, treatable |
| 4 | Deep ulnar motor branch neuropathy | Hypothenar + interossei only, occupational |
| 5 | True neurogenic TOS | Cervical rib, C8/T1, young female |
| 6 | C8/T1 radiculopathy | MRI disc prolapse |
| 7 | SMA / dHMN | Genetic, EMG beyond affected limb |
| 8 | Intramedullary tumor | MRI cord |
Minimum Workup
- MRI cervical spine — neutral + flexion (Hirayama), T2 (syrinx, Chiari, tumor, disc)
- EMG + NCS — pattern of denervation, conduction block, SNAP intact?
- Anti-GM1 IgM — MMN
- CXR — cervical rib / elongated C7 transverse process
- SMN1 gene test — if EMG shows widespread denervation beyond one limb
Sources: Bradley and Daroff's Neurology in Clinical Practice; Harrison's Principles of Internal Medicine 22E; Goldman-Cecil Medicine
Procide in case of meningitis with raised ict and how to collect csf in large hydrocephalus case
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
I have enough textbook material combined with core clinical knowledge. Let me give a comprehensive answer on both questions.
Procedure in Meningitis with Raised ICP + CSF Collection in Hydrocephalus
These are two related but distinct clinical problems that require precise decision-making.
PART 1: Approach to LP in Suspected Meningitis with Raised ICP
The Core Dilemma
Lumbar puncture (LP) is essential to diagnose meningitis — but herniation is a real risk if ICP is significantly raised. However, antibiotics must not be delayed waiting for imaging or LP.
"Antibiotics should not be delayed awaiting successful lumbar puncture. Antibiotics will not affect PCR or bacterial antigen testing." — Rosen's Emergency Medicine
Step 1 — Start Empirical Treatment IMMEDIATELY
Before any imaging or LP:
| Drug | Dose | Target |
|---|---|---|
| Ceftriaxone | 2 g IV q12h | S. pneumoniae, N. meningitidis, H. influenzae |
| Vancomycin | 40–60 mg/kg/day ÷ q8–12h | Resistant pneumococcus |
| Ampicillin | 2 g IV q4h | Add if age >50, immunocompromised, or Listeria suspected |
| Dexamethasone | 0.15 mg/kg q6h × 4 days | Start with or just before first antibiotic dose → reduces mortality and neurological sequelae in pneumococcal meningitis |
| Aciclovir | 10 mg/kg IV q8h | If HSV encephalitis cannot be excluded |
If blood cultures can be drawn within 10 minutes — do so before antibiotics. If not feasible, give antibiotics first.
Step 2 — Identify Features of Raised ICP / Risk of Herniation
Perform CT brain BEFORE LP if ANY of the following are present:
| Feature | Why Dangerous |
|---|---|
| Papilloedema on fundoscopy | Confirms raised ICP |
| New focal neurological deficit | Suggests mass lesion |
| Depressed consciousness (GCS ≤12) | Impaired autoregulation |
| Seizure within last 1 week | May indicate focal lesion |
| Immunocompromised state (HIV, transplant, steroids) | Atypical mass lesion (toxoplasmosis, lymphoma) |
| Known CNS lesion / SOL | Obvious |
| Abnormal posturing / decerebrate | Impending herniation |
If NONE of the above → LP is safe without prior CT.
Step 3 — Performing the LP Safely When ICP is Raised
If CT shows:
- No midline shift, no mass lesion, no herniation → LP can proceed, but with precautions
- Signs of herniation / large mass / obliterated cisterns → LP is contraindicated (see alternative CSF collection below)
Safe LP Technique When ICP is Elevated:
-
Position: Lateral decubitus (left lateral) — never sitting position when ICP is raised (sitting raises CSF pressure further at lumbar level)
-
Needle: Use a pencil-point (atraumatic) spinal needle (Whitacre 22G) — lower risk of post-LP herniation than cutting needle; smaller bore
-
Level: L3–L4 or L4–L5 interspace (below the conus at L1–L2)
-
Stylet IN: Always remove stylet slowly, never rapidly decompress
-
Measure opening pressure: Normal <20 cm H₂O; in meningitis often 20–40+ cm H₂O
-
Volume removed: Collect minimum volume necessary (3–4 tubes, ~8–12 mL total) — do not open tap and let CSF drain freely
-
Do NOT drain large volumes — risk of downward herniation
-
Mannitol cover: If opening pressure >40 cm H₂O or patient deteriorates during LP:
- Stop immediately
- Give Mannitol 0.5–1 g/kg IV over 20 minutes
- Consider neurosurgical consult for EVD
-
After LP: Keep patient flat for 1 hour; monitor closely for signs of coning (Cushing's triad — hypertension, bradycardia, irregular respirations)
Step 4 — Managing ICP in Meningitis (Adjunctive Measures)
| Intervention | Indication / Details |
|---|---|
| Head elevation 30° | Reduces ICP while maintaining cerebral perfusion |
| Mannitol 20% (0.5–1 g/kg IV) | Acute ICP rise; osmotic diuresis; duration 3–4 hours |
| Hypertonic saline (3%) | Alternative to mannitol; preferred in hypovolemic patients |
| Avoid hypotonic fluids | Use isotonic NS for maintenance |
| Hyperventilation (PCO₂ 30–35 mmHg) | Temporary bridge only; vasoconstriction reduces CBF |
| Dexamethasone | Reduces cerebral oedema in bacterial meningitis |
| Avoid hyperthermia | Fever worsens ICP; paracetamol/cooling |
| Avoid hyponatraemia | SIADH common in meningitis; fluid restriction if Na falls |
| External ventricular drain (EVD) | If obstructive hydrocephalus develops; provides ICP monitoring + CSF drainage |
When LP is Absolutely Contraindicated in Meningitis
| Condition | Action |
|---|---|
| CT shows transtentorial herniation / uncal herniation | NO LP; treat ICP; consider EVD |
| Posterior fossa mass / cerebellar herniation risk | NO LP (most dangerous) |
| Overlying skin infection at LP site | Avoid direct LP; consider C1–C2 lateral cervical puncture by neurosurgeon |
| Thrombocytopenia < 20,000 or coagulopathy (INR >1.5) | Correct first; or obtain CSF from EVD if already in situ |
PART 2: CSF Collection in Large / Obstructive Hydrocephalus
When LP is contraindicated (obstructive hydrocephalus with high ICP), CSF must be obtained by alternative routes. This requires neurosurgical input.
Option 1 — External Ventricular Drain (EVD) / Ventriculostomy
Method of choice in acute obstructive hydrocephalus with meningitis.
- Kocher's point (most common site): 1 cm anterior to the coronal suture, 2–3 cm lateral to midline (mid-pupillary line) on the non-dominant side
- Catheter placed into the frontal horn of the lateral ventricle
- Provides:
- Direct CSF sampling (ventricular CSF in meningitis) — highest diagnostic yield
- Continuous ICP monitoring
- Therapeutic CSF drainage to control ICP
- Note: ventricular CSF in meningitis may show higher cell counts, lower glucose, higher protein than lumbar CSF — interpret accordingly
Option 2 — Sampling Via Existing VP Shunt / Reservoir
If the patient already has a ventriculoperitoneal (VP) shunt or Ommaya reservoir:
- Shunt tap (by neurosurgeon or trained neurologist):
- Insert 23–25G butterfly needle into the shunt reservoir under sterile conditions
- Aspirate 2–3 mL CSF gently (do not aspirate hard — risk of pulling choroid plexus)
- Send for cell count, protein, glucose, culture, sensitivity
- Also culture the shunt hardware itself if shunt infection suspected
- The shunt itself may be the source of infection (S. epidermidis, S. aureus — shunt meningitis)
Option 3 — C1–C2 Lateral Cervical Puncture (Cisternal Puncture)
- Performed under fluoroscopic guidance between C1 and C2 vertebrae
- Accesses the lateral cervical subarachnoid space
- Used when:
- LP site infected / impossible
- Communicating hydrocephalus but lumbar route blocked
- Requires experienced operator (neurosurgery/interventional radiology)
Option 4 — Cisternal Puncture (Suboccipital)
- Needle inserted into the cisterna magna (suboccipital approach)
- Access between the posterior arch of C1 and the foramen magnum
- Rarely used; significant risk of brainstem injury if not performed by an expert
- Reserved for special circumstances (lumbar block, spinal deformity)
Summary Flowchart
Suspected Meningitis
|
↓
IMMEDIATELY: Blood cultures + empirical antibiotics + dexamethasone + aciclovir
|
↓
Any risk features for raised ICP?
(focal signs, papilloedema, GCS≤12, immunocompromised, seizure)
|
YES | NO
| → LP immediately (lateral decubitus, thin needle, minimal volume)
↓
CT Brain STAT
|
No herniation → LP with ICP precautions
|
Herniation / obstructive hydrocephalus
|
↓
NEUROSURGERY: EVD / shunt tap / Ommaya reservoir
↓
Ventricular CSF sent for analysis + culture
ICP controlled with EVD drainage
Sources: Bradley and Daroff's Neurology in Clinical Practice; Rosen's Emergency Medicine 10E; Harrison's Principles of Internal Medicine 22E
This is a shared conversation. Sign in to Orris to start your own chat.