Classify sterilization methods and describe their principles with advantages and limitations
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
I now have all the material needed for a comprehensive, well-structured answer. Here it is:
Classification of Sterilization Methods
Sterilization is the total destruction of all microbial forms, including bacterial spores, mycobacteria, non-enveloped viruses, and fungi — distinguishing it from disinfection, which may leave resilient organisms viable. Methods are classified into three broad categories:
I. Physical Sterilants
1. Heat
a) Moist Heat — Steam Under Pressure (Autoclave)
Principle: Saturated steam under pressure achieves temperatures above 100°C. Reactive water molecules irreversibly denature microbial proteins by disrupting hydrogen bonds between peptide groups. The autoclave (essentially a sophisticated pressure cooker) replaces air in the chamber with pure saturated steam; pressure itself plays no direct sterilizing role — it serves only to elevate steam temperature.
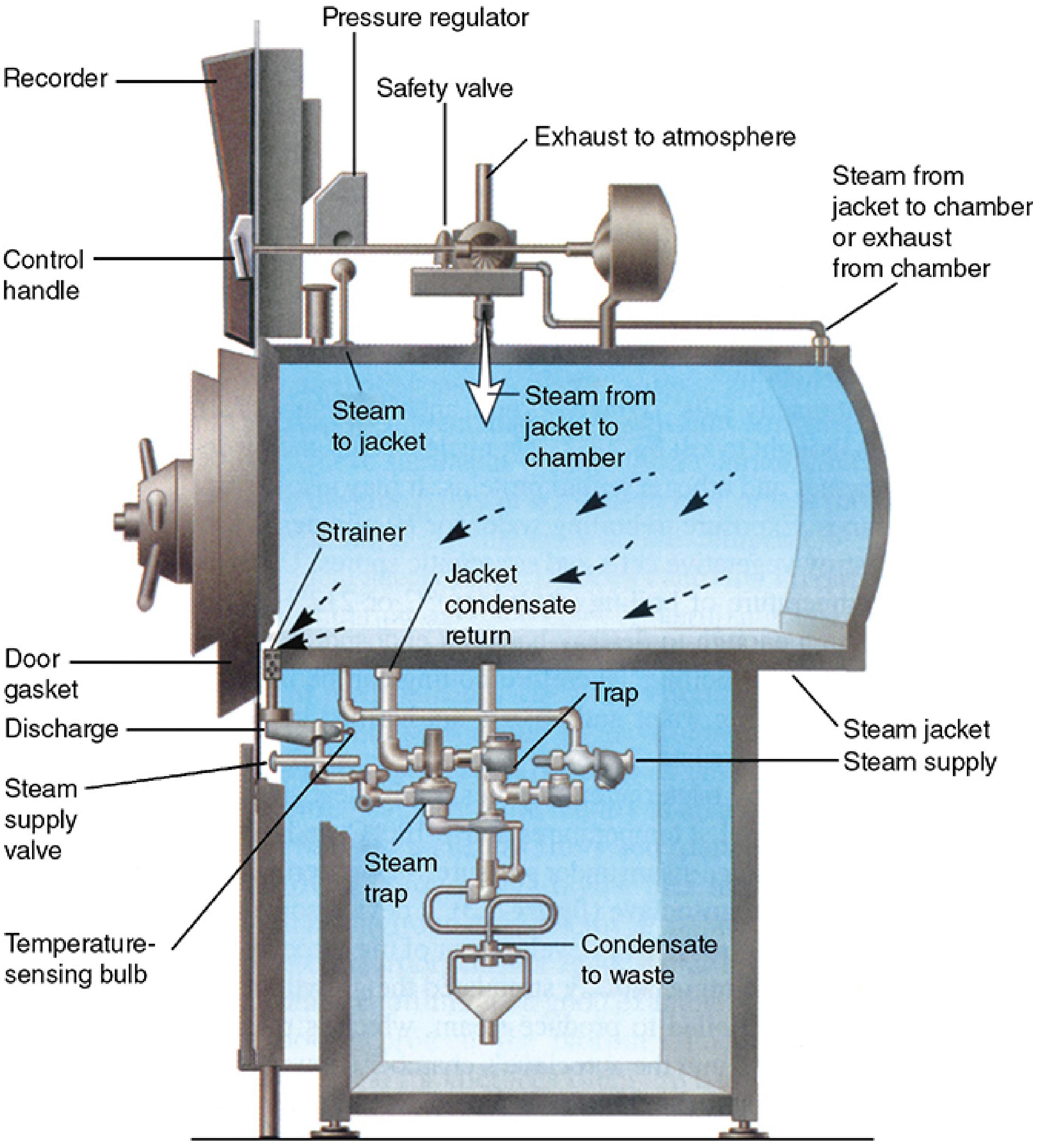
Standard cycle: 121°C for 15–20 minutes (spores directly exposed are killed in < 5 minutes at 121°C; the longer time accounts for variable steam penetration). A "flash" autoclave in operating rooms uses 134°C for 3 minutes.
Critical parameters: Time of exposure, temperature, and moisture content. A drop of 1.7°C increases required exposure time by ~48%.
| Advantages | Most popular and widely used; inexpensive; non-toxic; reliable; effective against all organisms including spores |
| Limitations | Corrosive to sharp instruments; cannot be used for heat- or pressure-sensitive items (certain plastics, lensed instruments, electronic components); may dull cutting edges |
b) Dry Heat (Hot-Air Oven)
Principle: Carbonization and oxidation of organic material without moisture. Requires higher temperatures because dry heat is far less efficient at protein denaturation than moist heat.
Standard cycle: 160°C for 2 hours; or 171°C for 1 hour; or 121°C for 6 hours.
| Advantages | No corrosion or dulling of instruments; inexpensive; suitable for metals, glassware, heat-resistant oils and waxes immiscible in water |
| Limitations | High temperatures required; prolonged exposure times; damages cloth, paper, rubber, and plastics; not suitable for most clinical instruments |
c) Incineration
Principle: Direct exposure to naked flame causes instantaneous carbonization and destruction of all microorganisms including spores. Used for wire loops in microbiology labs or for disposable contaminated waste.
| Advantages | Rapid and complete; eliminates waste |
| Limitations | Destroys the item; not applicable for reusable instruments or equipment |
2. Filtration
Principle: Physical removal (not killing) of microorganisms by passage through membrane filters with defined pore sizes.
- Bacterial removal: 0.22–0.45 μm pore size filters
- HEPA filters: Used for air sterilization (remove particles ≥ 0.3 μm with ≥ 99.97% efficiency)
| Advantages | Ideal for heat-labile fluids (serum, certain pharmaceuticals, enzyme solutions); removes both live and dead organisms |
| Limitations | Not effective for viruses (too small); only applicable to liquids and air (not solids); membranes can clog; does not kill — organisms remain viable if filter is breached |
3. Radiation
a) Ultraviolet (UV) Radiation
Principle: UV light (optimally at 254 nm wavelength) is absorbed by nucleic acids, causing thymine dimer formation and lethal DNA damage.
| Advantages | Effective for surface decontamination; useful for decontaminating air in critical hospital areas, biosafety cabinets, and facilities handling hazardous organisms |
| Limitations | Very poor penetration — cannot sterilize through glass, plastic, or opaque materials; only surface-level effect; potential harm to skin and eyes; items must be directly exposed |
b) Ionizing Radiation (Gamma rays, Cathode rays)
Principle: Carries far greater energy than UV. Causes direct DNA strand breaks and generates toxic free radicals and hydrogen peroxide from intracellular water, leading to cell death.
| Advantages | Excellent penetration — items can be packaged before irradiation and remain sterile; widely used industrially for disposable surgical supplies (gloves, plastic syringes, specimen containers), foodstuffs, and pharmaceuticals |
| Limitations | Expensive equipment; requires specialized facilities with radiation shielding; may degrade certain polymers or plastics; not practical for routine clinical use |
II. Gas/Vapor Sterilants
4. Ethylene Oxide (EO) Gas
Principle: Alkylating agent — replaces labile hydrogen atoms in DNA, RNA, and proteins, inactivating microorganisms by irreversibly alkylating nucleic acids and enzymes. Operates at 29°C–65°C.
Cycle: Generally 4 hours of exposure + mandatory 12-hour aeration period to allow toxic gas to diffuse out.
| Advantages | Highly effective against all microorganisms including spores; essential for heat- and pressure-sensitive items (artificial heart valves, certain endoscopes, electronic components, plastic tubing) |
| Limitations | Inflammable, potentially explosive; carcinogenic in animals; strict regulatory controls; prolonged aeration required; toxic residues; expensive; slow process (up to 7 days for polyvinyl chloride items) |
5. Hydrogen Peroxide Vapor
Principle: Oxidizing agent — generates reactive oxygen free radicals that attack cell membranes, enzymes, and DNA.
| Advantages | Effective sterilant; no toxic byproducts (breaks down to water and oxygen); does not require aeration; faster than EO |
| Limitations | Cannot be used with materials that absorb or react with hydrogen peroxide (cellulose-containing materials such as paper); limited penetration compared to EO |
6. Plasma Gas Sterilization
Principle: Hydrogen peroxide is vaporized, then exposed to microwave-frequency or radio-frequency energy, generating a plasma state with highly reactive free radicals (hydroxyl and hydroperoxyl radicals) that destroy microorganisms.
| Advantages | Efficient; no toxic byproducts; low temperature (< 50°C); has largely replaced EO for many applications; short cycle times |
| Limitations | Cannot be used with hydrogen-peroxide–absorbing or reactive materials; higher equipment cost; not suitable for long narrow lumens or cellulosic materials |
III. Chemical Sterilants (Liquid)
7. Peracetic Acid (0.2%)
Principle: Potent oxidizing agent. End products (acetic acid + oxygen) are non-toxic.
| Advantages | Excellent microbicidal activity including spores; non-toxic breakdown products; rapid action |
| Limitations | Corrosive to metals; must be used in specialized automated systems; limited shelf life once activated |
8. Glutaraldehyde (2%)
Principle: Bifunctional alkylating agent that cross-links amino groups in proteins and nucleic acids of microorganisms.
| Advantages | Broad-spectrum activity; widely used for heat-sensitive equipment (flexible endoscopes); available in office/clinical settings |
| Limitations | Significant safety concerns — irritating to skin, eyes, and respiratory tract; requires careful handling; slower sporicidal activity at room temperature; items must be thoroughly rinsed before use |
Summary Classification Table
| Method | Category | Spectrum | Key Use |
|---|---|---|---|
| Steam autoclave (121°C/15 min) | Physical – Moist heat | All (sterilizing) | General surgical/lab instruments |
| Dry heat oven (160°C/2 hr) | Physical – Dry heat | All (sterilizing) | Glassware, metals, oils |
| Incineration | Physical – Dry heat | All | Disposable waste, lab loops |
| Filtration (0.22 µm) | Physical – Mechanical | Bacteria, fungi (not viruses) | Heat-labile fluids |
| UV radiation (254 nm) | Physical – Radiation | All (surface only) | Air/surface decontamination |
| Ionizing radiation | Physical – Radiation | All (sterilizing) | Pre-packaged disposable supplies |
| Ethylene oxide gas | Gas/Vapor | All (sterilizing) | Heat-labile devices, plastics |
| H₂O₂ vapor | Gas/Vapor | All (sterilizing) | Instruments, closed systems |
| Plasma gas (H₂O₂) | Gas/Vapor | All (sterilizing) | Heat-sensitive instruments |
| Peracetic acid 0.2% | Chemical – Liquid | All (sterilizing) | Surgical instruments |
| Glutaraldehyde 2% | Chemical – Liquid | All (sterilizing) | Endoscopes, heat-sensitive equipment |
Sources: Medical Microbiology 9e (Murray); Sherris & Ryan's Medical Microbiology, 8th ed.; Dermatology 5e (Bolognia); Pfenninger & Fowler's Procedures for Primary Care, 3rd ed.
Find recent clinical guidelines on sterilization protocols
Reading File
Searching PubMed
"sterilization"[MeSH Terms] AND "guideline"[pt]
Searching PubMed
"disinfection sterilization"[MeSH Terms] AND "endoscope" AND "reprocessing"
Searching the Web
sterilization protocols clinical guidelines 2023 2024 CDC WHO AORN
Reading Article
Reading Article
Reading Article
Reading Article
Reading Web Page
sterilization protocols clinical guidelines recommendations
Searching PubMed
"Spaulding classification" AND "reprocessing" AND "medical devices"
Searching the Web
AAMI ST58 2024 chemical sterilization low temperature AAMI ST108 2023 water quality sterile processing
Reading Article
Reading Article
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.