Bronchogenic carcinoma
Reading File
Finding Sources
Searching PubMed
"bronchogenic carcinoma" OR "lung cancer"[MeSH Terms] AND "diagnosis AND treatment"
Reading File
Reading File
Searching PubMed
"lung cancer"[MeSH Terms] AND "treatment"[MeSH Terms]
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Searching Images
bronchogenic carcinoma lung cancer pathology types
I now have comprehensive information to provide a thorough overview of bronchogenic carcinoma. Here is the complete reference:
Bronchogenic Carcinoma (Lung Cancer)
Definition & Epidemiology
Bronchogenic carcinoma refers to primary malignant epithelial tumors arising from the bronchial or bronchiolar epithelium (and alveolar cells). It is the leading cause of cancer-related death in high-resource countries, accounting for approximately one-third of cancer deaths in men and, since 1987, the leading cancer killer in women as well.
- ~237,000 new cases and ~130,000 deaths per year in the United States (2022 estimates)
- Peak incidence: 5th–6th decade
- At diagnosis: >50% already have distant metastases; ~25% have regional lymph node involvement
- Overall 5-year survival: ~20% (all stages); ~50% when localized at diagnosis
— Robbins & Kumar Basic Pathology, p. 485
Classification (2021 WHO)
Lung carcinomas are divided into two major clinical groups with further histologic subtypes:
Non-Small Cell Lung Cancer (NSCLC) — ~85%
| Subtype | Frequency | Location | Key Features |
|---|---|---|---|
| Adenocarcinoma | ~38% (most common) | Peripheral | TTF-1+; EGFR/KRAS/ALK mutations; common in women, non-smokers, <45 yr |
| Squamous Cell Carcinoma | ~20% | Central (major bronchi) | Keratin pearls; strong smoking association; cavitation; hypercalcemia (PTHrP) |
| Large Cell Carcinoma | ~3% | Peripheral/central | Diagnosis of exclusion; poorly differentiated |
Small Cell Lung Cancer (SCLC) — ~14%
- Neuroendocrine origin; strong smoking association
- Marked by TP53 and RB loss-of-function mutations
- Rapid growth, early widespread metastases
- Highly sensitive to chemotherapy initially, but almost universally recurs
- LD/ED staging (or TNM system per IASLC)
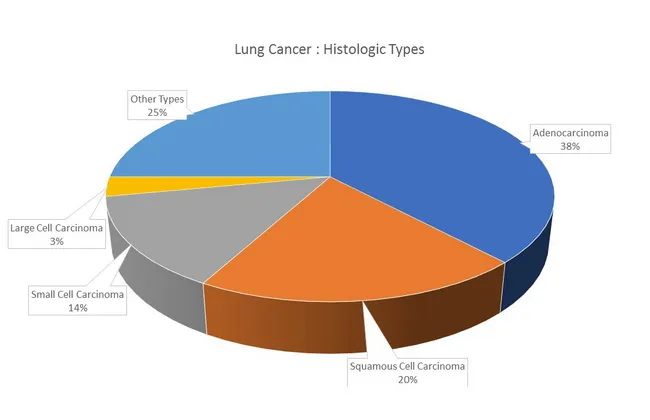
— Robbins & Kumar Basic Pathology, Table 11.5; Fishman's Pulmonary Diseases
Etiology & Pathogenesis
Smoking
- ~90% of lung cancers occur in current or recent smokers
- Risk is ~60× higher in heavy smokers (2 packs/day × 20 years) vs. non-smokers
- Even passive smoking increases risk
- Cessation reduces risk over time but never to baseline; genetic changes persist in bronchial epithelium
Other Carcinogens
- Asbestos: 5× risk in nonsmokers; 55× in heavy smokers exposed to asbestos (synergistic)
- Uranium, arsenic, chromium, nickel, vinyl chloride
- Radon gas (second leading cause of lung cancer)
- Silica (IARC Group 1 carcinogen)
Molecular Pathogenesis
Stepwise accumulation of driver mutations mirroring histologic progression:
| Mutation | Association |
|---|---|
| EGFR (exon 19 del / L858R) | Adenocarcinoma, women, non-smokers |
| KRAS (30% of adenocarcinomas) | Smokers; mutually exclusive with EGFR |
| ALK fusion | ~5% adenocarcinomas; younger, non-smokers |
| ROS1, HER2, MET, RET | Low frequency, targetable |
| TP53 + RB loss | SCLC (virtually always) |
Adenocarcinoma progression: Atypical adenomatous hyperplasia → AIS (lepidic pattern, formerly BAC) → Minimally invasive adenocarcinoma → Invasive adenocarcinoma
Squamous cell carcinoma progression: Squamous metaplasia → Dysplasia → CIS → Invasive SCC
— Robbins & Kumar Basic Pathology, pp. 485–488; Fishman's Pulmonary Diseases
Morphology
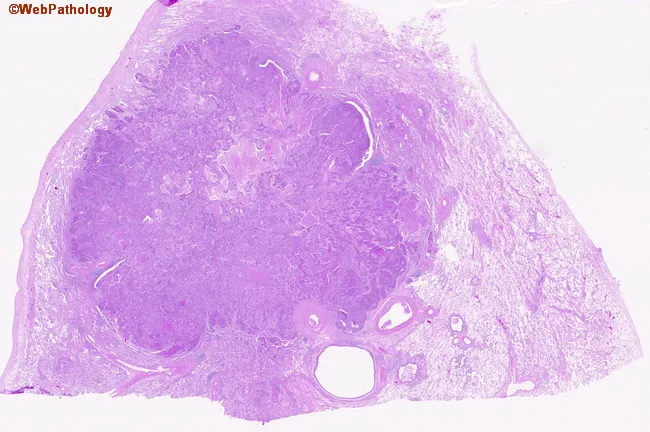
- Adenocarcinoma: Peripheral, gray-white, acinar/papillary/lepidic/solid/mucinous growth; TTF-1 positive
- Squamous cell carcinoma: Central (major bronchi), gray-white mass; keratin pearls in well-differentiated tumors; cavitation common; may shed cells detectable in sputum cytology
- SCLC: Central, soft, gray-white, extensive necrosis; small cells with scant cytoplasm, "salt and pepper" chromatin, no nucleoli; diffuse infiltration of mediastinum
- Large cell carcinoma: Large, poorly differentiated cells with no squamous/glandular/neuroendocrine differentiation
Clinical Features
Local/Intrathoracic Symptoms
- Cough — most common presenting symptom
- Hemoptysis — ~5% with hemoptysis and normal CXR have lung cancer
- Dyspnea, wheezing — airway obstruction (especially squamous cell/central tumors)
- Hoarseness — recurrent laryngeal nerve palsy (more common with left-sided tumors looping under aortic arch)
- Chest pain — pleural or chest wall invasion
- Dysphagia — extrinsic esophageal compression (subcranial lymph nodes)
Regional Syndromes
- SVC Syndrome — swelling of face, neck, upper torso/arms, dyspnea; lung cancer = 75% of SVC cases (4% NSCLC, 10% SCLC)
- Pancoast Syndrome (superior sulcus tumor): Shoulder/arm pain (brachial plexus C8–T2 invasion), upper extremity weakness/paresthesias, Horner syndrome (ptosis, miosis, anhidrosis) from sympathetic chain compression
- Phrenic nerve palsy — diaphragm elevation
- Postobstructive pneumonia / atelectasis — recurrent pneumonia in same lobe is a red flag
Metastatic Symptoms
- Bone pain (25%), brain symptoms (10%; lung cancer = ~2/3 of cancers presenting as brain metastases), adrenal, liver
- Constitutional: weight loss, weakness, anorexia, fever
— Fishman's Pulmonary Diseases, pp. 1331–1333
Paraneoplastic Syndromes
Present in up to 10% of lung cancer patients. Key syndromes:
| Syndrome | Tumor Type | Mechanism |
|---|---|---|
| SIADH (hyponatremia) | SCLC (10–45%) | Ectopic ADH |
| Ectopic ACTH (Cushing's) | SCLC (up to 50%) | Ectopic ACTH |
| Hypercalcemia | Squamous cell | PTHrP secretion |
| Lambert-Eaton myasthenic syndrome | SCLC | Anti-VGCC antibodies |
| Clubbing / Hypertrophic osteoarthropathy | Adenocarcinoma, SCC | Unknown |
| Trousseau syndrome (migratory thrombophlebitis) | Adenocarcinoma | Hypercoagulability |
Staging
NSCLC — TNM System (8th Edition, IASLC)
| Stage | 5-Year Survival |
|---|---|
| IA (~T1N0M0) | ~80–90% |
| IB (T2N0M0) | ~70% |
| IIA/IIB | ~30–55% |
| IIIA/IIIB (locally advanced) | ~10–30% |
| IV (metastatic) | ~5% |
SCLC — Traditional Staging
- Limited Disease (LD): Confined to one hemithorax, regional nodes (can encompass radiation field)
- Extensive Disease (ED): Beyond one hemithorax (most patients at diagnosis)
The IASLC now recommends TNM staging for SCLC as well.
Diagnosis & Workup
Imaging
- Chest CT — first-line for all suspected cases; defines size, location, nodal involvement
- PET-CT — functional staging; detects occult metastases
- Brain MRI (or CT with contrast) — recommended for stage II+ NSCLC and all SCLC; brain = common metastatic site
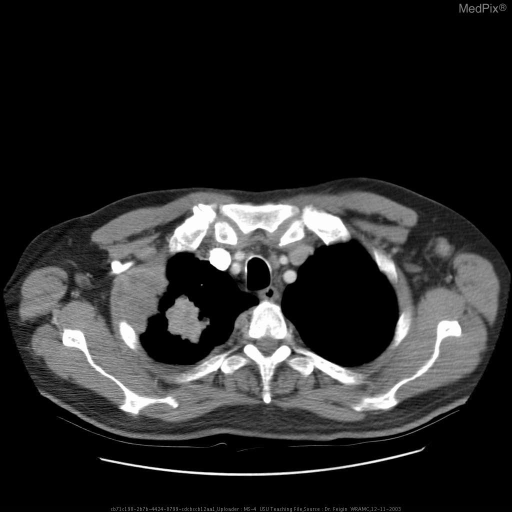
Tissue Diagnosis
- Flexible bronchoscopy — central lesions; diagnostic yield ~90% for endobronchial tumors
- CT-guided transthoracic needle biopsy — peripheral lesions; pneumothorax risk ~15%
- EBUS-TBNA — mediastinal lymph node staging; now first-line for mediastinal evaluation
- Thoracentesis — malignant pleural effusion (diagnosis + staging in one procedure)
- Strategy: whenever possible, biopsy a metastatic site to simultaneously achieve diagnosis + staging; ensure sufficient tissue for molecular profiling
Molecular Testing (for all advanced NSCLC)
Essential to guide targeted therapy:
- EGFR mutation (exon 19/21 especially)
- ALK rearrangement
- ROS1, KRAS G12C, BRAF V600E, MET exon 14, RET, NTRK
- PD-L1 expression (TPS score for immunotherapy eligibility)
Treatment Overview
NSCLC
| Stage | Treatment |
|---|---|
| I–II | Surgical resection (lobectomy/pneumonectomy) ± adjuvant chemotherapy |
| IIIA | Concurrent chemoradiotherapy ± surgery; durvalumab consolidation |
| IIIB/C | Concurrent chemoradiotherapy |
| IV (EGFR/ALK/ROS1+) | Targeted tyrosine kinase inhibitors (osimertinib, alectinib, etc.) |
| IV (PD-L1 ≥50%) | Pembrolizumab monotherapy or combination |
| IV (no driver, PD-L1 <50%) | Platinum-based doublet ± immunotherapy |
SCLC
- LD: Concurrent cisplatin/etoposide + thoracic radiotherapy; prophylactic cranial irradiation (PCI) in responders
- ED: Carboplatin/etoposide + atezolizumab or durvalumab; PCI
- Despite initial high response rates (60–80% in LD), relapse is nearly universal
- Overall 5-year survival: LD ~10–25%; ED ~2–5%
Key Comparisons: SCLC vs. NSCLC
| Feature | SCLC | NSCLC |
|---|---|---|
| Location | Central | Central (SCC) / Peripheral (adeno) |
| Doubling time | Rapid | Slower |
| Metastases | Early, widespread | Later |
| Chemo sensitivity | High (initially) | Moderate |
| Surgery | Rarely curative | Curative if early stage |
| Driver mutations | TP53, RB | EGFR, KRAS, ALK, ROS1, etc. |
| Paraneoplastic | SIADH, ACTH, Lambert-Eaton | Hypercalcemia, clubbing |
Sources: Robbins & Kumar Basic Pathology; Fishman's Pulmonary Diseases and Disorders; Murray & Nadel's Textbook of Respiratory Medicine; Robbins, Cotran & Kumar Pathologic Basis of Disease
This is a shared conversation. Sign in to Orris to start your own chat.