Latest guidelines for management of hypertension in pregnancy
Reading File
Finding Sources
Searching PubMed
"hypertension" AND "pregnancy" AND "management"
Searching the Web
hypertension in pregnancy management guidelines 2024 2025 ACOG ISSHP
Reading File
Reading File
Searching PubMed
"preeclampsia" AND "antihypertensive" AND "treatment"
Reading Web Page
hypertension pregnancy management antihypertensive guidelines 2025
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
I now have all the information needed to compile a comprehensive, well-structured answer.
Management of Hypertension in Pregnancy
Classification
Hypertension in pregnancy is defined as BP ≥ 140/90 mmHg. The table below summarizes the five recognized subtypes:
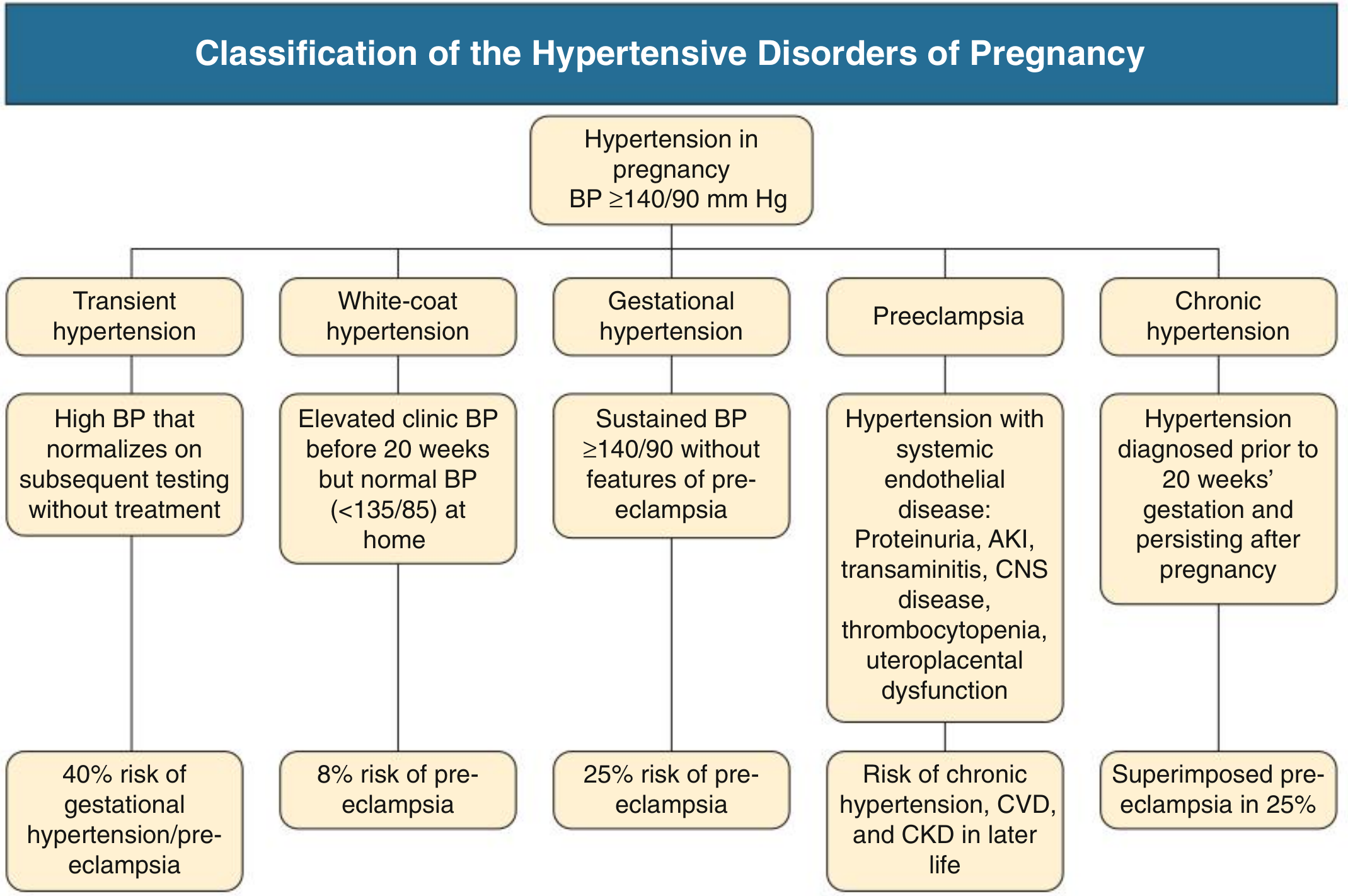
— Comprehensive Clinical Nephrology, 7th Ed.
| Disorder | Definition | Key Risk |
|---|---|---|
| Gestational hypertension | New HTN ≥20 weeks, no proteinuria/organ dysfunction | 25% progress to preeclampsia |
| Preeclampsia | New HTN ≥20 weeks + organ dysfunction or uteroplacental dysfunction | Eclampsia, HELLP, CVD later in life |
| Chronic hypertension | HTN before 20 weeks or persisting >12 weeks postpartum | Superimposed preeclampsia in 25% |
| White-coat hypertension | Office BP ≥140/90 but home BP <135/85 | 8% risk of preeclampsia |
| Eclampsia | Seizure in woman with preeclampsia | Maternal/fetal mortality |
Key diagnostic update: Proteinuria is no longer required to diagnose preeclampsia if other organ dysfunction criteria (AKI, transaminitis, thrombocytopenia, CNS or hepatic abnormalities, uteroplacental dysfunction) are present.
Blood Pressure Thresholds for Treatment
Non-Severe Hypertension (BP 140–159/90–109 mmHg)
- The CHIPS trial demonstrated that tight BP control (target DBP 85 mmHg) versus less-tight control (target DBP 100 mmHg) did not impair fetal outcomes but significantly reduced severe hypertension, thrombocytopenia, and transaminitis in the mother.
- The 2025 AHA/ACC Hypertension Guideline now recommends treating chronic hypertension in pregnancy to a goal of < 140/90 mmHg — a shift from prior guidance that deferred treatment for BP below 160/105.
- Previous guidance (ACOG 2013) advised against treatment if BP <160/105 in the absence of end-organ damage, citing concern for reduced uteroplacental perfusion; this has been updated based on the CHIPS and CHAP trial evidence.
Severe Hypertension (SBP ≥ 160 or DBP ≥ 105–110 mmHg)
- Treatment is mandatory to prevent stroke and maternal cardiovascular complications.
- Per 2025 AHA/ACC: antihypertensive therapy must be initiated within 30–60 minutes of confirmed severe readings (confirmed on repeat measurement within 15 min).
- Target: reduce BP promptly but avoid precipitous drops that could compromise uteroplacental flow.
Antihypertensive Drug Selection
First-Line Oral Agents
| Drug | Notes |
|---|---|
| Methyldopa | Centrally acting α2-agonist; most extensive safety data; safe for fetus; drawback: multiple daily dosing, sedation |
| Labetalol | Combined α/β-blocker; preferred over pure β-blockers due to α-blockade preserving uteroplacental flow; oral and IV forms |
| Long-acting nifedipine | Once-daily dosing; CCB; effective; can cause edema |
Acute/IV Management (Severe Hypertension)
| Drug | Dosing | Notes |
|---|---|---|
| Labetalol IV | 20 mg IV, escalate to 40 mg at 10 min if inadequate | First-line IV agent |
| Hydralazine IV/IM | 5–10 mg IV/IM q20 min | Second-line; increased risk of maternal hypotension and placental abruption vs. labetalol |
| Nicardipine IV | Extensive safety data as tocolytic | Effective alternative |
| Oral nifedipine | Can be used for urgent control | Effective in non-IV setting |
Second-Line Oral Agents
- Metoprolol (long-acting formulation available; less data than labetalol)
- Verapamil, diltiazem (limited data; no evidence of fetal harm)
Agents to Avoid / Contraindicated
| Agent | Reason |
|---|---|
| ACE inhibitors | Multiple fetal anomalies (fetopathy: renal tubular dysgenesis, oligohydramnios, skull ossification defects) |
| ARBs | Same risks as ACE inhibitors |
| Direct renin inhibitors | Contraindicated (2025 AHA/ACC expanded list) |
| Atenolol | Associated with fetal growth restriction |
| Sodium nitroprusside | Risk of fetal cyanide poisoning if used >4 hours; avoid; also on 2025 AHA/ACC contraindicated list |
| MRAs (spironolactone) | Theoretical inadequate virilization of male fetuses; eplerenone may be safer alternative |
| Diuretics | Avoided — may impair plasma volume expansion of pregnancy (used only when specifically indicated, e.g., pulmonary edema) |
— Brenner & Rector's The Kidney; Goodman & Gilman's Pharmacological Basis of Therapeutics; 2025 AHA/ACC Guideline
Preeclampsia: Prevention
Low-Dose Aspirin
- Recommended beginning at 10–12 weeks for patients with chronic hypertension or at elevated risk of preeclampsia.
- Mechanism: inhibits thromboxane-mediated platelet aggregation and vasoconstriction while sparing prostacyclin production.
- USPSTF recommends 81 mg/day for all high-risk women starting 12–28 weeks (optimally before 16 weeks).
Calcium Supplementation
- Recommended in populations with low dietary calcium intake (Cochrane evidence supports reduction in severe preeclampsia and maternal mortality).
Preeclampsia: Definitive Treatment — Delivery
- Delivery is the only cure for preeclampsia.
- At ≥37 weeks: delivery is indicated.
- Severe preeclampsia with fetal maturity: delivery after maternal stabilization.
- Severe preeclampsia at <34 weeks in a stable patient: expectant management may allow fetal maturation, with close in-hospital monitoring.
Seizure Prophylaxis: Magnesium Sulfate
- Gold standard for eclampsia prevention and treatment in women with severe preeclampsia.
- Given for CNS manifestations: headache, visual disturbance, altered mental status.
- Loading dose: 4–6 g IV over 15–20 min; maintenance: 1–2 g/hr IV.
- Also recommended postpartum: ~20% of eclampsia cases occur >48 hours after delivery.
- Monitor for toxicity: respiratory depression (Mg >7–8 mEq/L), loss of patellar reflexes. Antidote: calcium gluconate.
Chronic Hypertension in Pregnancy: Pre-conception Approach
- Evaluate for secondary causes before or early in pregnancy: renal artery stenosis, primary hyperaldosteronism, pheochromocytoma, obstructive sleep apnea (40% of pregnant women with HTN had OSA in one polysomnography study).
- Switch to pregnancy-safe agents before conception.
- Counsel on risks: preeclampsia, preterm birth, IUGR.
- Screen for superimposed preeclampsia throughout pregnancy.
Postpartum Management
- Most antihypertensives are compatible with breastfeeding; nifedipine and labetalol are preferred.
- BP often spikes in the first 3–5 days postpartum — monitor closely.
- Women with preeclampsia have long-term cardiovascular risk: chronic hypertension, CKD, CVD later in life — flag for ongoing surveillance.
- A 2024 systematic review (PMID 39806714) found nifedipine and labetalol are the most studied oral agents for postpartum hypertension management.
2025 AHA/ACC Key Updates (Pregnancy-Specific)
The 2025 AHA/ACC Hypertension Guideline introduced several pregnancy-specific changes:
- Treat chronic HTN to <140/90 mmHg (not the prior permissive 160/105 threshold for mild HTN)
- Severe HTN: medications must be given within 30–60 minutes of confirmed BP ≥160/110
- Expanded contraindicated list: now explicitly includes atenolol, ACE inhibitors, ARBs, direct renin inhibitors, nitroprusside, and MRAs
- Low-dose aspirin starting 10–12 weeks for preeclampsia prevention
Summary: Quick-Reference Algorithm
BP ≥ 140/90 in pregnancy?
│
├─ Before 20 weeks → Likely chronic HTN (rule out white-coat)
│ → Switch to safe agent (methyldopa / labetalol / nifedipine)
│ → Screen for secondary HTN
│ → Aspirin 81 mg from 10–12 weeks
│
├─ After 20 weeks, no features of preeclampsia → Gestational HTN
│ → Monitor weekly; treat BP to <140/90
│
├─ After 20 weeks + organ dysfunction → Preeclampsia
│ → Admit; antihypertensives + MgSO₄ prophylaxis
│ → Plan delivery based on gestation and severity
│
└─ BP ≥ 160/110 (severe) at any time
→ IV labetalol or oral nifedipine
→ Treat within 30–60 minutes
→ MgSO₄ if any CNS features
Recent evidence note: A 2024 network meta-analysis (PMID 38488570) comparing antihypertensives for preeclampsia found nifedipine and labetalol had comparable efficacy and safety profiles; hydralazine was associated with more maternal hypotension. A 2025 systematic review (PMID 40410291) of non-severe hypertension treatment confirmed that BP-lowering reduces maternal morbidity without compromising fetal growth when targets remain above placental perfusion thresholds.
This is a shared conversation. Sign in to Orris to start your own chat.