Describe morphological change in cell injury
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
reversible cell injury morphology swelling
Loading Image
Searching Images
necrosis apoptosis cell death morphology histology
I have all the core content needed. The read_file from Robbins Basic Pathology gave me comprehensive detail on reversible injury, irreversible injury, necrosis (and its patterns), and apoptosis. Let me compile this into a thorough answer.
Morphological Changes in Cell Injury
Cell injury produces a spectrum of structural changes depending on the type, duration, and severity of the injurious stimulus. The response ranges from fully reversible derangements to irreversible cell death.
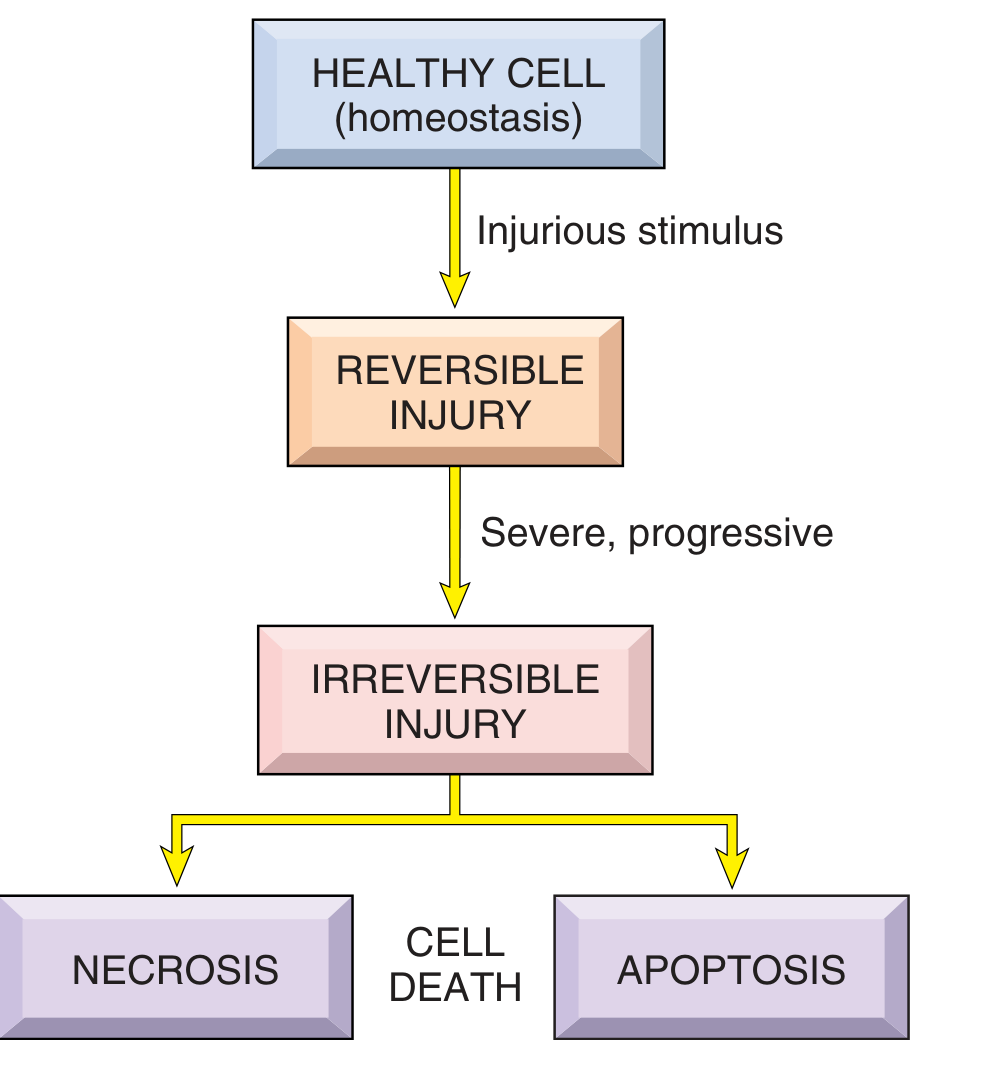
1. Reversible Cell Injury
Reversible injury is a derangement of function and morphology from which cells can recover if the damaging stimulus is removed. Two cardinal morphological features define it:
A. Cellular Swelling (Hydropic Change / Vacuolar Degeneration)
- Most common and earliest manifestation; results from failure of energy-dependent Na⁺/K⁺-ATPase pumps → influx of Na⁺ and water into the cell.
- Gross: Organ pallor, increased turgor, increased weight.
- Light microscopy: Small clear vacuoles in the cytoplasm (distended, pinched-off segments of the ER).
- Electron microscopy: Plasma membrane blebbing, blunting/loss of microvilli, loosening of intercellular attachments; mitochondrial swelling with amorphous phospholipid-rich densities; ER dilation with detachment of ribosomes; nuclear chromatin clumping.
- Cytoplasm may contain myelin figures — whorled phospholipid masses derived from damaged cellular membranes.
B. Fatty Change (Steatosis)
- Appearance of lipid vacuoles in the cytoplasm.
- Principally in organs involved in fat metabolism, especially the liver.
- Common with hypoxic, toxic (e.g., alcohol, CCl₄), or metabolic injury.
C. Increased Eosinophilia
- Injured cytoplasm becomes redder (more eosinophilic on H&E) due to loss of cytoplasmic RNA (which normally binds the basic dye hematoxylin) and denaturation of cytoplasmic proteins. This change becomes more pronounced as injury progresses toward necrosis.
2. Irreversible Cell Injury → Cell Death
If injury is severe or prolonged, cells pass the "point of no return" and die. Irreversible injury is consistently characterized by:
- Inability to restore mitochondrial function (oxidative phosphorylation/ATP)
- Membrane structural loss and dysfunction
- Loss of DNA/chromatin structural integrity
Death manifests as necrosis or apoptosis, with distinct morphologies.
3. Necrosis
Necrosis is characterized by cell swelling, membrane rupture, organelle breakdown, and release of cellular contents — triggering a local inflammatory response.
Nuclear Changes (most reliable morphological indicators):
| Change | Description |
|---|---|
| Pyknosis | Nuclear shrinkage and increased basophilia (chromatin condensation) |
| Karyorrhexis | Fragmentation of the pyknotic nucleus |
| Karyolysis | Fading/dissolution of the nucleus due to DNase digestion |
Cytoplasmic Changes:
- Increased eosinophilia (pink on H&E) — due to loss of normal basophilic RNA and binding of eosin to denatured proteins.
- Loss of normal cytoplasmic structure; cells appear "glassy" or granular.
- Myelin figures may be more prominent.
- Calcification may appear in long-standing necrotic foci.
4. Patterns of Tissue Necrosis
Different organs and injury types produce characteristic patterns:
| Pattern | Gross/Micro Appearance | Causes/Location |
|---|---|---|
| Coagulative necrosis | Preserved tissue architecture ("ghost cells"); firm, pale-yellow area; protein denaturation halts autolysis | Ischemia in most solid organs (heart, kidney, spleen) |
| Liquefactive necrosis | Complete digestion — creamy/pus-like liquid; no tissue architecture | Brain infarcts; bacterial abscesses (due to neutrophil enzymes) |
| Caseous necrosis | "Cheese-like" appearance; structureless, granular debris; surrounded by granulomatous inflammation | Tuberculosis and fungal infections |
| Fat necrosis | Chalky-white areas; enzymatic destruction of fat; saponification (calcium soaps) | Acute pancreatitis; traumatic fat necrosis of breast |
| Fibrinoid necrosis | Homogeneous, bright pink (eosinophilic) deposits in vessel walls on H&E; immune complex deposition | Malignant hypertension; autoimmune vasculitis |
| Gangrenous necrosis | Not a distinct pattern — usually coagulative necrosis with superimposed infection; "dry" vs "wet" gangrene | Ischemic limbs; diabetic foot |
5. Apoptosis
Apoptosis is programmed cell death — orderly, energy-dependent elimination of single cells without inflammation. It is both physiological (embryogenesis, lymphocyte deletion) and pathological (DNA-damaged cells, viral infections).
Morphological Features of Apoptosis:
- Cell shrinkage — cell becomes smaller; cytoplasm is dense and eosinophilic; organelles are more tightly packed (contrast with cell swelling in necrosis).
- Chromatin condensation — nuclear chromatin condenses into dense masses beneath the nuclear envelope (characteristic crescentic pattern).
- Nuclear fragmentation — nucleus breaks into two or more fragments (karyorrhexis).
- Plasma membrane blebbing — surface bulges form but membrane integrity is maintained (no leakage of contents).
- Apoptotic body formation — cell fragments into membrane-bound apoptotic bodies containing cytoplasm, organelles, and nuclear fragments.
- Phagocytosis — apoptotic bodies are rapidly engulfed by adjacent cells or macrophages; no inflammation results (contrast with necrosis).
- On H&E: apoptotic cells appear as round/oval eosinophilic masses, sometimes with condensed nuclear fragments.
6. Comparison: Necrosis vs. Apoptosis
| Feature | Necrosis | Apoptosis |
|---|---|---|
| Cell size | Enlarged (swelling) | Reduced (shrinkage) |
| Nucleus | Pyknosis → karyorrhexis → karyolysis | Fragmentation into nucleosome-sized pieces |
| Plasma membrane | Disrupted | Intact, with blebs |
| Cellular contents | Leaked out | Retained in apoptotic bodies |
| Inflammation | Yes (contents act as DAMPs) | No |
| Energy dependence | No (passive) | Yes (ATP-dependent) |
| Programmed? | No (accidental) | Yes |
| Physiological role | Pathological only | Normal + pathological |
7. Other Emerging Forms of Cell Death
Recent classifications recognize additional regulated necrotic pathways with distinct morphological/biochemical profiles:
- Necroptosis — morphologically resembles necrosis (cell swelling, membrane rupture) but is programmed via RIPK3/MLKL; pro-inflammatory.
- Pyroptosis — gasdermin-mediated pore formation → cell swelling and rupture; highly pro-inflammatory; associated with inflammasome activation (caspase-1/11).
- Ferroptosis — iron-dependent, lipid peroxidation-driven; mitochondria appear smaller with increased membrane density on EM; no nuclear fragmentation; distinct from classical necrosis or apoptosis.
Summary
The progression of cell injury follows a continuum:
Healthy cell → Reversible injury (cellular swelling, fatty change, ER changes) → Irreversible injury → Cell death (necrosis with nuclear pyknosis/karyorrhexis/karyolysis and tissue-specific patterns, or apoptosis with chromatin condensation and apoptotic body formation).
The morphological appearance depends on whether injury is sublethal or lethal, the cell type (parenchymal, neural, immune), the tissue microenvironment, and the mechanism (ischemia, toxins, immune attack, DNA damage).
Sources: Robbins & Kumar Basic Pathology, p. 18–26; Robbins, Cotran & Kumar Pathologic Basis of Disease
This is a shared conversation. Sign in to Orris to start your own chat.