Precocioua puberty types features investigation treatment in detail
Reading File
Finding Sources
Finding Sources
Searching PubMed
"precocious puberty"[MeSH Terms] AND treatment
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive, authoritative content from multiple major textbooks. Here is the detailed answer:
Precocious Puberty — Types, Features, Investigations & Treatment
Definition
Precocious puberty is the development of secondary sexual characteristics before:
- Girls: age 8 years (breast development, pubic hair, menarche)
- Boys: age 9 years (testicular enlargement >4 mL, pubic hair, phallic growth)
Note: The Lawson Wilkins Pediatric Society (1999) proposed lowering thresholds to 7 years (White girls) and 6 years (Black girls), but this remains controversial and may lead to underdiagnosis.
Precocious puberty is 20× more common in girls than boys. In ~90% of girls the cause is idiopathic; in boys, a structural CNS cause is found in ~50–66%.
Classification
The fundamental classification is based on whether the process is GnRH-dependent (central/hypothalamic-pituitary axis activated) or GnRH-independent (peripheral, autonomous sex steroid secretion):
| Feature | Central (True) CPP | Peripheral (Pseudo) PPP |
|---|---|---|
| Mechanism | Premature HPG axis activation | Autonomous steroid secretion |
| GnRH dependence | Dependent | Independent |
| LH/FSH | Elevated, pubertal pattern | Suppressed / prepubertal |
| Sex steroids | Elevated | Elevated |
| GnRH stimulation test | Pubertal response | Blunted/absent response |
| Progression | Usually progressive | Variable |
Additionally:
- Isosexual precocity: development consistent with phenotypic sex
- Heterosexual precocity: estrogenic features in boys or virilization in girls
Type 1: Central (True / Gonadotropin-Dependent) Precocious Puberty
Mechanism
Premature activation of the hypothalamic–pituitary–gonadal (HPG) axis → pulsatile GnRH secretion → ↑ LH and FSH → ↑ gonadal sex steroid production.
Causes
Girls (90% idiopathic)
| Cause | Details |
|---|---|
| Idiopathic | Most common in girls; no structural abnormality found |
| Hypothalamic hamartoma | 2–28% of CPP; most common structural cause; congenital mass of ectopic GnRH neurons; presents with extreme precocity (<3 yrs), gelastic (laughing) seizures, behavioral disturbances; isodense on imaging, does not enhance with contrast |
| CNS tumors | Optic glioma, arachnoid cysts, astrocytoma, ependymoma, tuberous sclerosis, craniopharyngioma |
| Hydrocephalus, septo-optic dysplasia, craniostenosis | Congenital malformations |
| Inflammatory/infectious lesions | Encephalitis, meningitis |
| hCG-secreting germinomas | Hypothalamic or pineal tumors; activate LH receptor |
| Genetic mutations | Activating mutations in kisspeptin (KISS1), kisspeptin receptor (KISS1R), or MKRN3 (makorin ring finger protein 3) gene |
| Neurofibromatosis type 1 | Documented association with CPP |
Boys (~2/3 have CNS disease)
- Hypothalamic tumors, cysts, inflammatory lesions, seizure disorders
- Idiopathic cases account for <10% in boys
Clinical Features
- Breast development, pubic/axillary hair, body odor
- Acceleration of linear growth and bone maturation
- Menarche (girls), testicular/phallic enlargement (boys), facial hair
- Emotional lability, acne
- Risk of short adult stature due to premature epiphyseal closure
- Increased long-term risks: breast and endometrial cancer, cardiovascular disease, type 2 diabetes, hypertension (girls)
Type 2: Peripheral (Pseudo / Gonadotropin-Independent) Precocious Puberty
Mechanism
Autonomous sex steroid secretion from gonads, adrenals, or ectopic sources — independent of LH/FSH stimulation.
Causes
| Cause | Sex | Key Features |
|---|---|---|
| McCune-Albright syndrome | Mainly girls | GNAS1 mutation → constitutive Gα activation → autonomous ovarian cysts → estrogen production. Classic triad: polyostotic fibrous dysplasia + café au lait spots ("coast of Maine" margins) + GnRH-independent precocity. Other endocrinopathies (hyperthyroidism, Cushing, acromegaly, hyperparathyroidism) may coexist |
| Congenital adrenal hyperplasia (CAH) | Both (virilization in girls) | 21-hydroxylase deficiency most common (CYP21A2 mutations). Classic form: virilization + growth acceleration + advanced bone age. Non-classic/late-onset: premature adrenarche, acne. 5–10% of premature adrenarche have late-onset CAH |
| Testotoxicosis (familial male-limited precocious puberty) | Boys | Autosomal dominant activating mutations in LH receptor → uncontrolled testosterone secretion. Signs of puberty at ~2 years of age |
| Adrenal tumors | Both | Androgen or estrogen-secreting adenoma/carcinoma |
| Gonadal tumors | Both | Granulosa-theca cell tumors (most common estrogen-secreting ovarian neoplasm, usually benign); Leydig cell tumors (testosterone-secreting); hCG-secreting germ cell tumors |
| Primary hypothyroidism | Both | High TSH has intrinsic FSH-like activity → ovarian cysts, breast development, vaginal bleeding. Unique: associated with delayed bone age (unlike all other causes). Responds to levothyroxine |
| Exogenous sex steroids | Both | Inadvertent exposure to estrogen/androgen-containing products |
| Functional ovarian cysts | Girls | Usually transient; resolve spontaneously |
Incomplete (Partial) Forms
These are isolated pubertal signs without full progression:
| Form | Features | Management |
|---|---|---|
| Premature thelarche | Isolated uni/bilateral breast enlargement, no nipple/areolar development, no other sexual maturation. Usually occurs by age 2, rarely after 4. Caused by breast sensitivity to low estrogen or transient follicular cysts. Benign, self-limited | Reassurance + follow-up every 3–6 months; no treatment needed. Uterine volume measurement is the most sensitive discriminator from early CPP |
| Premature adrenarche/pubarche | Isolated pubic/axillary hair without other maturation. Increased sensitivity to adrenal androgens. Must exclude late-onset CAH | Follow-up; screen for NCAH (17-OHP), watch for PCOS/insulin resistance. Risk of PCOS, hyperinsulinemia, dyslipidemia in adolescence especially if low birth weight |
| Isolated premature menarche | Rare; vaginal bleeding without other pubertal signs | Investigate for vaginal pathology, foreign body, estrogen exposure |
Investigations
Step 1 — Clinical Assessment
- Complete history: timing, rapidity of progression, family history, birth history, CNS symptoms
- Physical examination: Tanner staging, measurement of height/weight/growth velocity
- Check for associated signs: café au lait spots, abdominal mass, neurological signs
Step 2 — Bone Age
- X-ray of left hand and wrist (Greulich-Pyle atlas)
- Advanced bone age in most forms of precocious puberty
- Delayed bone age only in hypothyroid-induced precocious puberty
Step 3 — Hormone Panel (First-Line)
| Test | Interpretation |
|---|---|
| Basal LH & FSH | LH >5 IU/L (or >0.6 IU/L by fluorometric assay, >0.3 IU/L by ICMA) → pubertal pattern → CPP likely |
| Estradiol (E₂) | Elevated in isosexual precocity in girls |
| Testosterone | Elevated in boys or virilized girls |
| DHEAS | Elevated in adrenarche/adrenal causes |
| 17-hydroxyprogesterone (17-OHP) | Screen for CAH (21-hydroxylase deficiency): early morning sample; <200 ng/dL rules out NCAH; 200–500 ng/dL → ACTH stimulation test; >1000 ng/dL diagnostic for CAH |
| TSH + T4 | Rule out primary hypothyroidism |
| hCG | Elevated in hCG-secreting tumors (germinomas) |
Step 4 — GnRH Stimulation Test (Gold Standard for CPP)
- IV bolus of 100 µg GnRH → measure LH and FSH at 0, 30, 60 minutes
- Pubertal response: LH peak ≥8 IU/L
- LH/FSH ratio >0.66–1.0 in girls is diagnostic of CPP
- In GnRH-independent PP: LH and FSH remain suppressed after GnRH stimulation
- Also used to monitor efficacy of GnRH agonist therapy
Step 5 — Imaging
| Imaging | Indication |
|---|---|
| MRI brain (gadolinium) | All boys with CPP; girls <6 years or with neurological signs. Localizes hypothalamic hamartomas, tumors, structural lesions |
| Pelvic/abdominal ultrasound | Assess ovarian cysts, adrenal tumors, uterine volume |
| Adrenal CT | If elevated testosterone + DHEAS → exclude adrenal tumor |
| Testicular ultrasound | Exclude Leydig cell tumor if elevated testosterone without ↑17-OHP or DHEAS |
| Breast ultrasound | Distinguish premature thelarche from fibroadenoma/cysts |
Diagnostic Flowchart (Phenotypic Females)
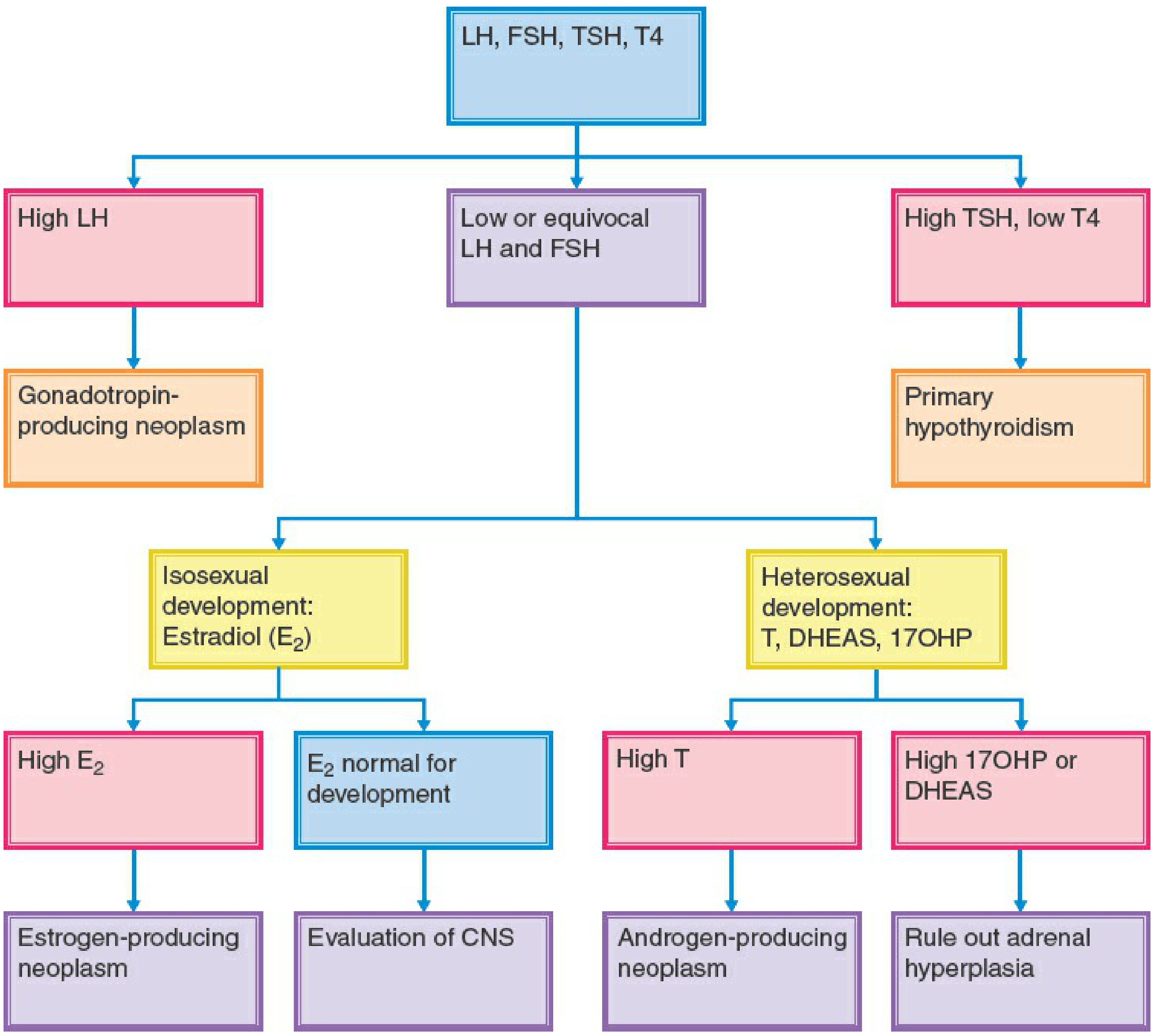
Treatment
Central (GnRH-Dependent) Precocious Puberty
GnRH Agonist Therapy — First-Line
GnRH agonists cause receptor downregulation → pituitary desensitization → suppression of LH, FSH, and sex steroids.
Goals:
- Halt pubertal progression
- Delay accelerated bone maturation and prevent premature epiphyseal closure
- Improve final adult height (most effective if started before age 6)
- Mitigate psychosocial consequences
- Does not cause osteoporosis
Key agents and dosing:
| Drug | Formulation | Dose |
|---|---|---|
| Leuprolide acetate | 3-month depot IM | 11.25 mg or 30 mg q3 months |
| Triptorelin | 6-month depot IM | 22.5 mg every 6 months |
| Other GnRH analogues (histrelin subcutaneous implant, nafarelin intranasal) | Various | Per formulary |
Monitoring: GnRH stimulation test used to monitor treatment efficacy (suppression of pubertal LH/FSH response confirms adequate suppression).
When to stop: Discontinuation at chronologic age ~11 years and bone age ~12 years is associated with maximum adult height gain. No consensus on optimal withdrawal age; 11–11.5 years has been widely recommended.
Puberty resumes after discontinuation. Counseling is an essential component of treatment.
Structural Cause (CNS Lesion / Tumor)
- Surgery or radiation therapy to remove/treat the causative lesion if feasible
- GnRH analogue therapy in addition if needed
Peripheral (GnRH-Independent) Precocious Puberty
Treatment is cause-specific:
| Cause | Treatment |
|---|---|
| CAH (21-hydroxylase deficiency) | Glucocorticoids: hydrocortisone 15 mg/m²/day (suppresses ACTH and adrenal androgen production) |
| Testotoxicosis (familial male-limited PP) | Ketoconazole 10–20 mg/kg/day (steroidogenesis inhibitor) ± antiandrogen: spironolactone 5–7 mg/kg/day or bicalutamide 2 mg/kg/day |
| McCune-Albright syndrome | GnRH agonists ineffective. Tamoxifen (SERM) — multicenter trial showed ↓ vaginal bleeding, ↓ growth rate, ↓ bone age advancement. Aromatase inhibitors (letrozole, testolactone) used as adjuncts |
| Primary hypothyroidism | Levothyroxine → all features (cysts, galactorrhea, precocity) resolve within months |
| Adrenal/gonadal tumors | Surgical resection |
| Ovarian granulosa cell tumor | Surgery (>2/3 are benign) |
| Functional ovarian cysts | Observation; most resolve spontaneously |
| Exogenous steroids | Remove source of exposure |
Additional agents for GnRH-independent PP (general):
- Aromatase inhibitors (testolactone, letrozole): used adjunctively for familial male-limited PP, CAH, and McCune-Albright
- Abiraterone (potent CYP17 inhibitor): novel but not yet systematically evaluated in children
- Ketoconazole: broad steroidogenesis inhibitor
Incomplete/Partial Forms
| Form | Treatment |
|---|---|
| Premature thelarche | No treatment; reassurance, 3–6 month follow-up |
| Premature adrenarche | No treatment unless CAH confirmed; treat PCOS/insulin resistance if present; long-term follow-up for metabolic complications |
| Premature menarche | Investigate and observe |
Long-Term Consequences (Untreated)
- Short stature: premature bone age advancement → early epiphyseal fusion
- Psychosocial distress: mismatch between physical and emotional maturity
- Girls: ↑ risk of breast cancer, endometrial cancer, cardiovascular disease, type 2 diabetes
- Boys: psychosocial difficulties, behavioral problems
Sources:
- Goldman-Cecil Medicine International Edition (2 Vol), pp. 348–364
- Harrison's Principles of Internal Medicine 22E (2025), pp. 3152–3153
- Berek & Novak's Gynecology, pp. 346–356
- Tietz Textbook of Laboratory Medicine, 7th Ed, pp. 2295–2314
This is a shared conversation. Sign in to Orris to start your own chat.