Oral candidiasis and fluconazole
Reading File
Finding Sources
Finding Sources
Searching PubMed
"oral candidiasis"[MeSH Terms] AND "fluconazole"[MeSH Terms]
Reading File
Reading File
Reading File
Searching Images
oral candidiasis thrush clinical appearance
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive information. Here is a complete, well-organized answer.
Oral Candidiasis & Fluconazole
Overview
Oral candidiasis (thrush) is the most common fungal infection of the oral cavity, caused by overgrowth of Candida species — predominantly C. albicans — from normal oral flora. It represents superficial mucosal infection triggered by disruption of host defenses.
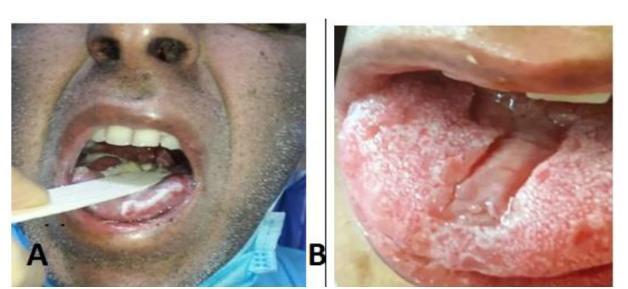
Clinical Forms
There are three distinct presentations (Goldman-Cecil Medicine):
| Form | Appearance | Key Features |
|---|---|---|
| Pseudomembranous (thrush) | White fungal plaques on any oral surface | Can be rubbed off, leaving red or bleeding base |
| Erythematous (atrophic) | Erythema ± atrophy of filiform papillae on dorsal tongue; patchy erythema on palate/buccal mucosa | Associated with oral burning, sensitivity to spicy foods; often linked to salivary hypofunction or dentures |
| Hyperplastic (candidal leukoplakia) | White plaques that do not rub off; fungal hyphae within hyperkeratotic epithelium | Found on anterior buccal mucosa or tongue |
Angular cheilitis (erythema/crusting at labial angles) is usually caused by Candida and typically co-exists with intraoral candidiasis — treat both simultaneously.
Predisposing Factors
- Suppression of bacterial flora by systemic antibiotics
- Chronic salivary dysfunction (Sjögren syndrome, anticholinergic drugs, radiotherapy)
- Uncontrolled diabetes mellitus or anemia
- Immunosuppression — especially HIV/AIDS (low CD4), organ transplant, chemotherapy
- Corticosteroid use (inhaled or systemic)
- Dentures (denture-associated candidiasis)
- Extreme prematurity, neutropenia, indwelling catheters, broad-spectrum cephalosporins/carbapenems
Diagnosis
- Clinical diagnosis is usually sufficient — white plaques that scrape off with an erythematous base
- KOH preparation or Gram stain of scrapings shows yeast cells and pseudohyphae
- Culture is rarely needed for mucocutaneous disease but is recommended for all clinically relevant isolates to assess azole susceptibility (particularly given C. auris and C. glabrata resistance concerns)
Fluconazole — Mechanism of Action
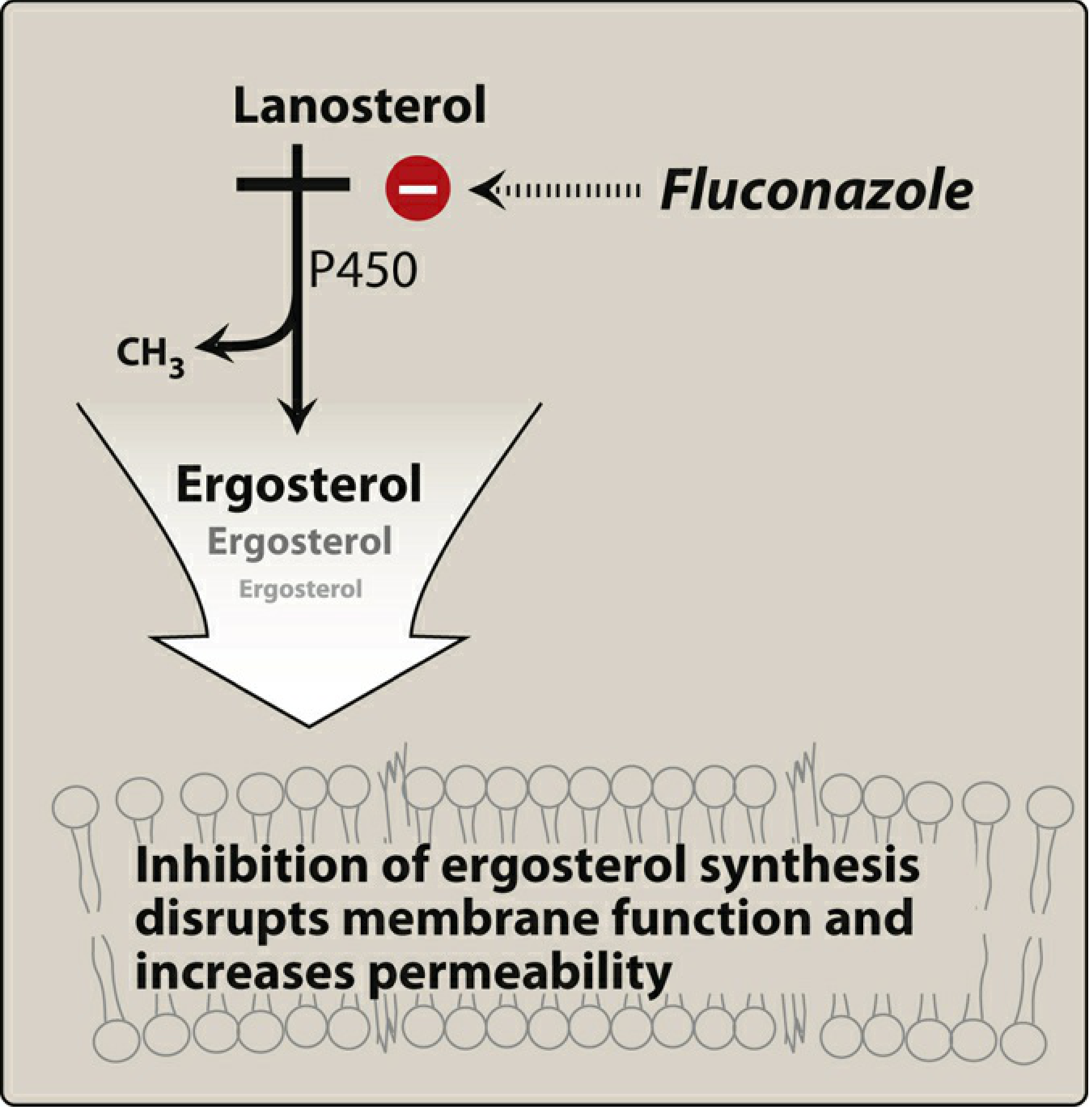
Fluconazole is a triazole antifungal that is predominantly fungistatic. It inhibits 14-α-demethylase (a CYP450 enzyme), thereby blocking the conversion of lanosterol → ergosterol. Ergosterol depletion disrupts fungal membrane structure and permeability, inhibiting cell growth. (Lippincott Pharmacology)
Fluconazole — Pharmacology
| Property | Detail |
|---|---|
| Formulations | Oral tablets/suspension, IV |
| Bioavailability | Excellent oral absorption (~90%); oral = IV efficacy |
| Distribution | Widely distributed; penetrates CSF, saliva, urine |
| Elimination | Primarily renal (>80% excreted unchanged in urine) — dose reduce in renal impairment |
| Spectrum | C. albicans, C. parapsilosis, C. neoformans, Coccidioides — active against most mucocutaneous Candida |
| Intrinsic resistance | C. krusei (intrinsically resistant); C. glabrata (often resistant) |
Treatment of Oral Candidiasis
Immunocompetent patients — first-line topical options
- Nystatin oral suspension
- Clotrimazole troches (not for infants)
- Miconazole mucoadhesive buccal tablets
Fluconazole may be more effective than nystatin/clotrimazole troches and is considered when topical therapy fails. (Red Book 2021)
Fluconazole dosing for oral/oropharyngeal candidiasis
- Standard regimen: 200 mg loading dose on day 1, then 100 mg/day for 2 weeks
- Immunocompromised patients (HIV/AIDS): fluconazole is first-line
Special situations
| Situation | Recommendation |
|---|---|
| Salivary hypofunction (e.g., Sjögren) | Systemic fluconazole may be ineffective (insufficient saliva to deliver drug to mucosa); prefer nystatin or miconazole vaginal tablets dissolved slowly in the mouth |
| Denture-associated candidiasis | Antifungal therapy + disinfect and refit the denture |
| Angular cheilitis | Topical clotrimazole 0.75% cream + concurrent intraoral/systemic antifungal |
| Esophageal candidiasis | Oral fluconazole (14–21 days); IV fluconazole/echinocandin if unable to swallow |
| Recurrent infections | Suppressive fluconazole (3× weekly) |
| Chronic mucocutaneous candidiasis | Fluconazole or itraconazole (lifelong intermittent therapy) |
Fluconazole-Refractory Disease
Resistance develops particularly in patients with advanced HIV/low CD4 counts on prolonged fluconazole prophylaxis. Mechanisms include:
- Mutations in 14-α-demethylase gene → reduced azole binding
- Efflux pumps expelling the drug from the cell
- Reduced ergosterol content
Alternatives for refractory disease:
- Itraconazole solution
- Voriconazole
- Posaconazole
- Amphotericin B deoxycholate oral suspension
- IV echinocandins (caspofungin, micafungin)
C. auris — an emerging multidrug-resistant pathogen — may be pan-resistant; suspect with prior antifungal exposure in healthcare settings.
Fluconazole — Adverse Effects & Drug Interactions
Common adverse effects: nausea, vomiting, headache, skin rash; reversible alopecia with prolonged use; ~10% develop elevated AST. Hepatotoxicity increases at doses >1200 mg/day.
Pregnancy: Fluconazole at standard doses during pregnancy increases cardiac birth defects ~3-fold; avoid in pregnancy unless benefit clearly outweighs risk.
Key drug interactions (fluconazole inhibits CYP3A4 and CYP2C9):
| Drug | Effect |
|---|---|
| Warfarin | ↑ INR (increased bleeding risk) |
| Phenytoin | ↑ phenytoin levels |
| Cyclosporine / tacrolimus | ↑ calcineurin inhibitor levels |
| Sulfonylureas (glipizide, glyburide) | ↑ hypoglycemia risk |
| Rifampin | ↓ fluconazole levels by ~25% |
| Cisapride, quinidine | ↑ QT prolongation risk |
Sources: Goldman-Cecil Medicine; Lippincott Illustrated Reviews: Pharmacology; Red Book 2021 (Committee on Infectious Diseases)
For uncontrolled diabetic patients
Finding Sources
Finding Sources
Searching PubMed
"oral candidiasis"[MeSH Terms] AND "diabetes mellitus"[MeSH Terms]
Reading File
Reading File
Finding Sources
Searching PubMed
"candidiasis" AND "diabetes mellitus" AND "oral"
Reading File
Finding Sources
Reading Article
Finding Sources
Finding Sources
I now have all the information needed. Here is a focused, clinically detailed answer.
Oral Candidiasis in Uncontrolled Diabetic Patients
Why Diabetes Increases Risk
Uncontrolled diabetes creates a highly permissive environment for oral Candida overgrowth through several converging mechanisms:
1. Immune Dysfunction from Hyperglycemia
Chronic hyperglycemia directly impairs innate immune defenses (Fitzpatrick's Dermatology):
- Impaired leukocyte chemotaxis, adherence, and phagocytosis — especially pronounced during hyperglycemia and diabetic ketoacidosis
- Reduced cutaneous T-cell function and blunted antigen challenge response
- Deficient IL-17 signaling at mucosal barriers, which is the primary defense against mucocutaneous Candida
2. High Glucose in Oral Secretions
- Elevated salivary glucose acts as a direct nutrient source for Candida, promoting rapid proliferation
- Reduced salivary flow (common in diabetics) further concentrates glucose and removes a key mechanical defense
3. Altered Oral Microbiome
Dysbiosis from hyperglycemia and frequent antibiotic use (for diabetic infections) shifts the oral flora, reducing bacterial competition against Candida
4. Neuropathy and Vascular Disease
Peripheral neuropathy and microangiopathy impair local tissue immunity and wound response
Clinical Patterns in Diabetic Patients
In mildly immunocompromised patients such as those with diabetes, infection typically remains superficial (pseudomembranous, erythematous, or angular cheilitis) — but risk of spread to deeper structures increases with worsening glycemic control or additional immunosuppression. (Robbins & Kumar Basic Pathology)
Diabetics are also at elevated risk for:
- Recurrent vulvovaginal candidiasis in women — recurrence should prompt clinicians to screen for underlying diabetes or HIV (Goldman-Cecil Medicine)
- Esophageal candidiasis — can occur when cellular immunity is impaired by poorly controlled diabetes
- Mixed-species infections — a 2023 study (PMID: 37721911) found mixed oral candidiasis (C. albicans co-infecting with C. glabrata, C. krusei, C. tropicalis, others) in ~13% of type 2 diabetic patients with oral candidiasis (HbA1c ≥7%). Treatment failure is more likely when the etiological agent is not species-identified.
Fluconazole in Uncontrolled Diabetics — Special Considerations
⚠️ Critical Drug Interaction: Fluconazole + Sulfonylureas
This is the most clinically important concern in diabetic patients. Fluconazole inhibits CYP2C9, the enzyme responsible for metabolizing sulfonylureas. This leads to:
| Sulfonylurea | Effect |
|---|---|
| Glipizide | ↑↑ plasma levels → severe hypoglycemia |
| Glyburide | ↑↑ plasma levels → severe hypoglycemia |
| Tolbutamide | ↑ plasma levels |
Goldman-Cecil Medicine explicitly lists glipizide and glyburide among drugs with "significant increases in blood level" caused by fluconazole.
Management: If fluconazole is prescribed in a sulfonylurea-treated patient:
- Warn the patient about hypoglycemia symptoms
- Monitor blood glucose closely during the course
- Consider dose reduction of the sulfonylurea
- Consider topical antifungals (nystatin, clotrimazole, miconazole) as first-line if infection is mild, to avoid the interaction entirely
Fluconazole Efficacy in Diabetics
- For patients with adequate salivary production, systemic fluconazole (200 mg loading dose → 100 mg/day × 2 weeks) remains the drug of choice (Goldman-Cecil Medicine)
- In patients with salivary hypofunction (common in diabetics due to autonomic neuropathy or anticholinergic antidiabetic medications), systemic fluconazole may be less effective because insufficient saliva is available to deliver drug to the oral mucosa — prefer nystatin vaginal tablets or miconazole buccal tablets dissolved slowly in the mouth
Risk of Resistance with Repeated Courses
Uncontrolled diabetics with recurrent infections often need repeated or prolonged antifungal therapy, raising the risk of azole resistance — including selection of intrinsically resistant species (C. krusei, C. glabrata). Culture and species identification should be performed in recurrent/refractory cases.
Treatment Algorithm for Diabetic Patients
Oral candidiasis in uncontrolled diabetic
│
├─ Assess glycemic control → optimize HbA1c (cornerstone of prevention)
│
├─ Mild-moderate, good salivary flow, NOT on sulfonylurea
│ → Fluconazole 200 mg day 1, then 100 mg/day × 14 days
│
├─ Mild, adequate salivary flow, ON sulfonylurea
│ → Prefer topical first: nystatin suspension or clotrimazole troches
│ If systemic required: monitor glucose closely, reduce SU dose
│
├─ Salivary hypofunction present
│ → Nystatin vaginal tablets or miconazole 50 mg buccal tablets
│ dissolved slowly in mouth (sucrose-free, avoids dental caries)
│
├─ Recurrent / refractory
│ → Culture and species ID
│ If C. glabrata or C. krusei: itraconazole, voriconazole,
│ posaconazole, or echinocandin
│
└─ Denture-associated → disinfect + refit denture + antifungal
Key Management Principles
- Glycemic control is the foundation — hyperglycemia drives both susceptibility and recurrence; antifungals treat symptoms, not the cause
- Screen for diabetes in any patient with recurrent oral or vulvovaginal candidiasis without another obvious cause
- Drug interaction vigilance — fluconazole + sulfonylureas = significant hypoglycemia risk
- Species identification in recurrent/refractory cases in diabetics, as non-albicans species (particularly C. glabrata, C. krusei) are more prevalent and often resistant to fluconazole
- Denture hygiene — diabetic patients wearing ill-fitting dentures have compounded risk; disinfection and refitting are essential adjuncts to antifungal therapy
Sources: Goldman-Cecil Medicine; Fitzpatrick's Dermatology; Robbins & Kumar Basic Pathology; Harrison's Principles of Internal Medicine 22E; Lippincott Pharmacology; Araiza et al., Biomedica 2023 [PMID: 37721911]
This is a shared conversation. Sign in to Orris to start your own chat.