Staphylococcus in detail
Reading File
Finding Sources
Searching PubMed
Staphylococcus aureus AND pathogenesis
Searching Images
Staphylococcus aureus gram stain microscopy
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Reading File
Finding Sources
Reading File
Loading Image
I now have comprehensive material. Let me compile the full detailed answer on Staphylococcus.
Staphylococcus — Comprehensive Review
1. Taxonomy and General Characteristics
The genus Staphylococcus contains >80 species and subspecies, most of which inhabit skin and mucous membranes of humans. The three clinically most important are:
| Species | Key Feature | Main Diseases |
|---|---|---|
| S. aureus | Coagulase positive | Invasive infections, toxin syndromes |
| S. epidermidis | Coagulase negative, slime layer | Foreign-body infections, endocarditis |
| S. saprophyticus | Coagulase negative, novobiocin resistant | UTI in young women |
Gram stain: Gram-positive cocci, 0.5–1.5 µm diameter, arranged in characteristic grape-like clusters on agar (though single cells, pairs, or short chains are common in clinical specimens).
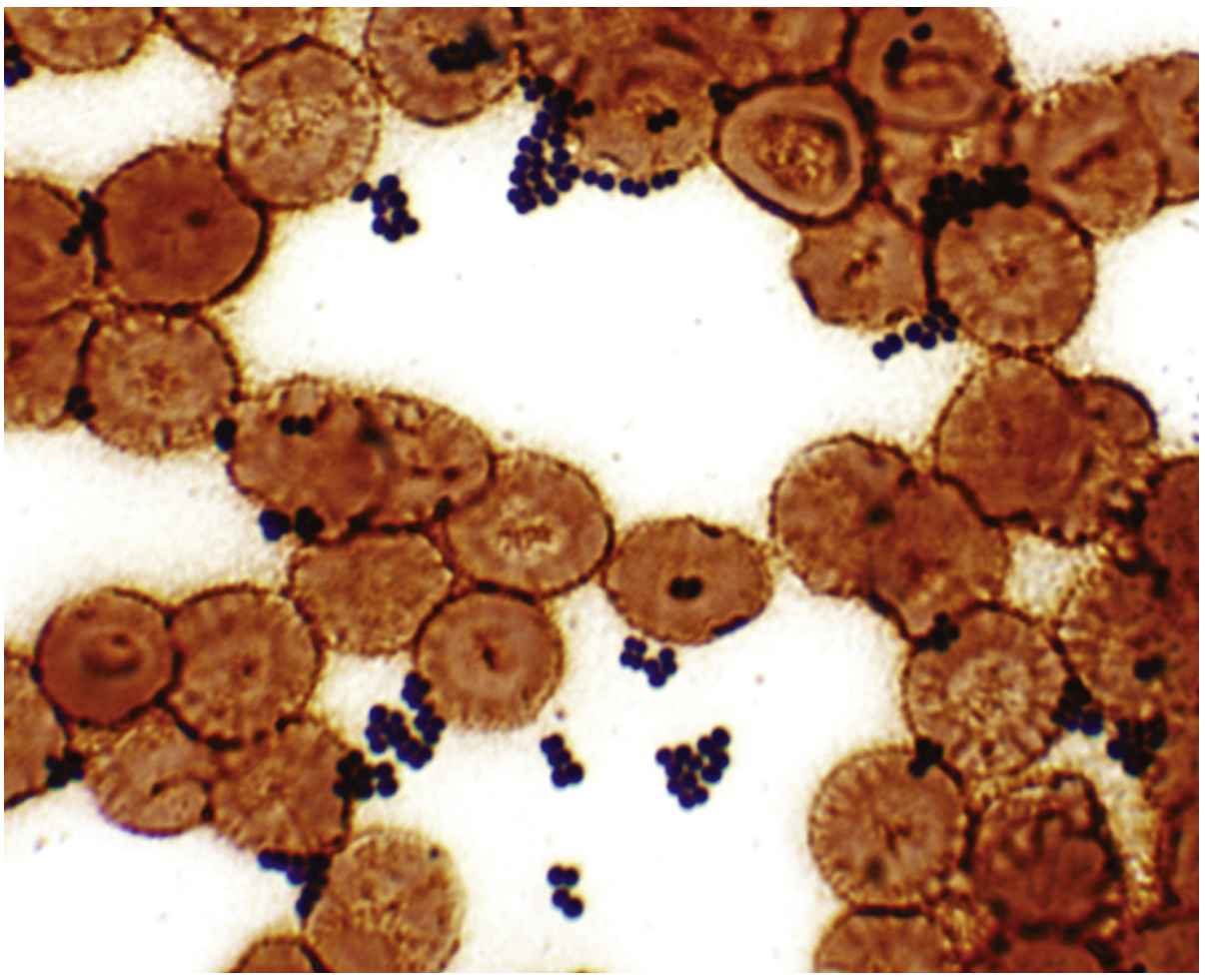
Gram stain of Staphylococcus in a blood culture — dark blue clusters amid erythrocytes (Medical Microbiology 9e, Fig. 18.1)
Biochemical identity markers:
- Catalase positive (distinguishes from Streptococcus)
- Coagulase positive = S. aureus (distinguishes from all CoNS)
- Grows aerobically AND anaerobically
- Tolerates 10% NaCl and temperatures 18–40°C
- Protein A present (IgG Fc binding)
2. Microbiology of S. aureus
Culture
- Forms large, smooth, β-hemolytic colonies on blood agar within 24 hours
- Golden-yellow pigment (carotenoid) on prolonged incubation
- Selective media: mannitol-salt agar (7.5–10% NaCl) or chromogenic agar
- MALDI-TOF mass spectrometry and coagulase testing for definitive ID
- PCR/NAAT used for MSSA/MRSA nasal screening
Cell Structure
| Component | Function |
|---|---|
| Peptidoglycan (thick) | Endotoxin-like: triggers cytokines, activates complement, aggregates platelets; allows survival on dry surfaces |
| Protein A | Binds IgG Fc → antiphagocytic; B-cell superantigen → supraclonal expansion + apoptosis |
| Polysaccharide capsule | Antiphagocytic |
| Teichoic acids | Adherence to mucosal cells |
| MSCRAMMs | Adhesins binding fibronectin, fibrinogen, collagen, laminin |
3. Virulence Factors
S. aureus produces >50 virulence factors encoded on the core chromosome, mobile genetic elements (bacteriophages, pathogenicity islands), and plasmids.
Toxins
| Toxin | Mechanism | Disease |
|---|---|---|
| α-toxin (alpha-hemolysin / Hla) | Forms pores in cell membranes; lyses RBCs, leukocytes, platelets | Tissue necrosis, pneumonia severity |
| β, γ, δ toxins | Additional hemolysins; membrane disruption | Synergistic tissue damage |
| Panton-Valentine Leukocidin (PVL) | Bi-component toxin; lyses neutrophils and macrophages | Skin abscesses; necrotizing community-acquired pneumonia |
| Phenol-soluble modulins (PSMs) | Lytic peptides; provoke host inflammatory responses | Community-associated MRSA severity |
| Exfoliative toxins A & B (ETA, ETB) | Serine proteases that cleave desmoglein-1 in the granular layer of the epidermis | Staphylococcal Scalded Skin Syndrome (SSSS) |
| Toxic Shock Syndrome Toxin-1 (TSST-1) | Superantigen: crosslinks MHC II with T-cell receptor → massive cytokine storm (IL-1, IL-2, TNF) | Toxic Shock Syndrome (TSS) |
| Enterotoxins A–Q | Heat-stable superantigens; resist boiling for 30 min; stimulate vagal afferents → vomiting | Food poisoning |
| Leukotoxins (Luk) | Leukocyte lysis | Enhanced immune evasion |
Enzymes
| Enzyme | Action |
|---|---|
| Coagulase (bound + free) | Converts fibrinogen → fibrin; fibrin coat protects from phagocytosis |
| Staphylokinase | Fibrinolysin — dissolves clots for dissemination |
| Hyaluronidase | Degrades hyaluronic acid → tissue spread |
| Lipases | Degrade lipids → skin invasion |
| Nucleases (DNase) | Degrade DNA in neutrophil extracellular traps |
| Beta-lactamase | Hydrolyzes penicillin ring → resistance |
Regulation of Virulence
Expression is coordinated by the agr (accessory gene regulator) quorum-sensing system. Small colonies with few neighbors → surface adhesins predominate (colonization phase). Dense colonies → secreted toxins and enzymes upregulated (invasion/spread phase).
4. Epidemiology
-
Colonization: 20–30% of healthy adults are persistent nasal carriers; ~60% are intermittent carriers. Primary niche: anterior nares, with secondary sites at oropharynx, perineum, axilla, groin.
-
Higher carriage rates: atopic dermatitis, insulin-dependent diabetes, dialysis, HIV infection, injection drug use
-
Transmission: predominantly direct contact (hand carriage); environmental surface contamination plays a secondary role; aerosol transmission is minimal.
-
MRSA: ~50% of S. aureus infections in US healthcare settings are MRSA. Two distinct epidemiologic strains:
- HA-MRSA (healthcare-associated): restricted to healthcare environments; resistant to multiple drug classes
- CA-MRSA (community-associated): circulates in community AND hospitals; frequently carries PVL; causes primary skin abscesses and necrotizing pneumonia; typically susceptible to more non-beta-lactam agents
-
S. aureus is responsible for millions of infections per year in the US; ~5–10% are invasive, with ¾ of those involving bacteremia.
5. Clinical Diseases
Toxin-Mediated Diseases
Staphylococcal Scalded Skin Syndrome (SSSS / Ritter Disease)
- Predominantly in neonates and children (<5 years); rare in adults (renal failure, immunosuppression)
- Exfoliative toxins A/B cleave desmoglein-1 → intraepidermal split at the granular layer
- Presentation: abrupt perioral erythema → generalized tender erythroderma → bullae → large sheet-like desquamation (Nikolsky sign positive)
- No organisms in the blisters (toxin acts remotely from distant focus, e.g., nasopharynx)
- Treat with anti-staphylococcal antibiotics + supportive care
Staphylococcal Food Poisoning
- Preformed heat-stable enterotoxin (most commonly A) in contaminated food (salted ham, cream, dairy)
- Incubation: 1–6 hours (short — toxin is preformed)
- Explosive onset: nausea, profuse vomiting, abdominal cramps ± diarrhea
- Resolves spontaneously within 24–48 hours; treatment is supportive
Toxic Shock Syndrome (TSS)
- Caused by TSST-1 (accounts for 75% of cases) or staphylococcal enterotoxins acting as superantigens
- Two settings: menstrual TSS (tampon use, vaginal colonization) and non-menstrual TSS (wound infections, surgery, nasal packing)
- Classic triad: fever ≥38.9°C + diffuse macular erythroderma + hypotension
- Multi-system involvement (≥3 organ systems): renal failure, hepatic dysfunction, thrombocytopenia, CNS encephalopathy, desquamation (especially palms/soles, 1–2 weeks after onset)
- Management: fluid resuscitation, anti-staphylococcal antibiotics + clindamycin (to suppress toxin synthesis), source control
Suppurative/Pyogenic Infections
| Disease | Key Features |
|---|---|
| Impetigo | Superficial skin vesicle on erythematous base; honey-colored crusting |
| Folliculitis | Impetigo involving hair follicles |
| Furuncles (boils) | Large painful pus-filled nodules involving deeper dermis |
| Carbuncles | Coalescence of furuncles into subcutaneous tissue; fever, chills, bacteremia |
| Cellulitis | Spreading dermal/subcutaneous infection |
| Wound infections | Surgical site infections; erythema, purulent discharge |
| Bacteremia | From any focus; high 30-day mortality; may seed distant sites |
| Endocarditis | Both tricuspid (IV drug users) and mitral/aortic (native/prosthetic valves); most common cause of acute IE worldwide; PVL-carrying strains adhere to endothelial and prosthetic surfaces |
| Pneumonia | Nosocomial (MRSA) > community-acquired; post-influenza necrotizing pneumonia (PVL strains); cavitation, pneumatoceles, empyema, septic shock |
| Osteomyelitis | Hematogenous; metaphysis of long bones in children; vertebrae in adults |
| Septic arthritis | Most common bacterial cause; any joint; requires drainage |
| Meningitis | Rare; post-neurosurgery or trauma |
| Spinal epidural abscess | Most common cause; back pain + fever + neurological deficits |
| Brain abscess | Post-trauma or hematogenous |
6. Coagulase-Negative Staphylococci (CoNS)
S. epidermidis
- Normal skin flora, usually non-pathogenic
- Virulence: biofilm ("slime layer") production → adherence to foreign bodies + resistance to phagocytosis and antibiotics
- Causes: subacute endocarditis (particularly prosthetic valves), infected IV catheters, CSF shunts, prosthetic joint infections, urinary catheters
- Treatment: vancomycin ± rifampin; removal of the foreign body is often required
S. saprophyticus
- Predilection for urinary tract of young sexually active women
- Causes dysuria, pyuria; rarely causes asymptomatic colonization
- Treatment: TMP-SMX or nitrofurantoin; responds rapidly; reinfection uncommon
7. Laboratory Diagnosis
| Test | Details |
|---|---|
| Gram stain | Clusters of Gram-positive cocci; useful for pyogenic infections, less for bacteremia or toxin syndromes |
| Culture | Nonselective blood agar (24h colonies); selective: mannitol-salt agar, chromogenic agar |
| Coagulase test | Slide (bound coagulase/clumping factor) + tube (free coagulase); positive = S. aureus |
| Catalase test | Positive (distinguishes from Streptococcus) |
| MALDI-TOF MS | Rapid, accurate species identification from colonies |
| PCR/NAAT | MRSA/MSSA nasal screening; detection from positive blood cultures |
| Antibiotic susceptibility testing | Disk diffusion, E-test, broth microdilution; oxacillin/cefoxitin disk for MRSA |
8. Antimicrobial Resistance — MRSA
Mechanism: mecA gene (on mobile SCCmec cassette) encodes PBP2a (penicillin-binding protein 2a), which has very low affinity for all β-lactam antibiotics → resistance to all penicillins, cephalosporins, and carbapenems.
Detection: Cefoxitin disk diffusion (surrogate for mecA); PCR for mecA gene.
9. Treatment
MSSA Infections
- Drug of choice: Nafcillin/oxacillin (antistaphylococcal penicillins) IV for serious infections
- Cefazolin: equivalent efficacy, less toxicity — preferred in many institutions; 2 g IV q8h for bacteremia/endocarditis
- Oral (mild-moderate SSTI): dicloxacillin, cephalexin
MRSA Infections
| Syndrome | MRSA Treatment |
|---|---|
| Skin/soft tissue (mild) | TMP-SMX, doxycycline, clindamycin (oral) |
| Bacteremia/endocarditis | Vancomycin 15–20 mg/kg IV q8–12h (target AUC:MIC >400) OR Daptomycin 8–10 mg/kg IV qd |
| Pneumonia (HAP/VAP) | Vancomycin OR linezolid (linezolid preferred by some guidelines for lung penetration) |
| CNS/meningitis | Vancomycin (linezolid as alternative) |
Recent evidence update: A 2025 systematic review and meta-analysis (PMID 40349971) found cefazolin non-inferior or superior to antistaphylococcal penicillins for MSSA bacteremia with a more favorable tolerability profile. A 2025 meta-analysis (PMID 39290168) found oral step-down therapy is reasonable for select patients with S. aureus bacteremia/endocarditis after initial IV stabilization.
Alternative/Salvage Agents
- Linezolid (600 mg IV/PO q12h) — useful for vancomycin MIC creep (MIC ≥2 µg/mL), pulmonary MRSA
- Daptomycin — avoid for pulmonary infections (inactivated by surfactant)
- Ceftaroline — 5th-generation cephalosporin active against MRSA; bactericidal
- Tedizolid, oritavancin, dalbavancin — newer agents for MRSA SSTI
- Tigecycline — broad-spectrum alternative; not for bacteremia
- Fosfomycin, fusidic acid — combination use for MRSA
10. Prevention and Control
- Hand hygiene — single most effective intervention (healthcare workers)
- MRSA surveillance screening: nasal swabs (NAAT) for high-risk patients on admission
- Decolonization: intranasal mupirocin + chlorhexidine baths for high-risk patients (pre-operatively or prior to dialysis)
- Proper wound care, aseptic catheter insertion, and maintenance bundles
- Covering exposed skin; isolation precautions (contact precautions) for MRSA
- No licensed S. aureus vaccine as of 2026 (multiple attempts failed in phase 3 trials)
Summary Table: S. aureus vs CoNS
| Feature | S. aureus | CoNS (S. epidermidis, S. saprophyticus) |
|---|---|---|
| Coagulase | Positive | Negative |
| Protein A | Present | Absent |
| Pigment | Golden-yellow | White/cream |
| Hemolysis | β-hemolytic | Variable |
| Primary virulence | Toxins + invasins | Biofilm (SlE) |
| Typical infections | Acute, aggressive | Subacute, foreign-body related |
| MRSA rate | High | High (MR-CoNS) |
Sources: Medical Microbiology 9e; Goldman-Cecil Medicine; Henry's Clinical Diagnosis and Management by Laboratory Methods; Robbins, Cotran & Kumar Pathologic Basis of Disease; Harrison's Principles of Internal Medicine 22E; Washington Manual of Medical Therapeutics; Fishman's Pulmonary Diseases.
Streptococcus in detail of each classified bacteria
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.