Focal seizures
Reading File
Finding Sources
Searching PubMed
"focal seizures" OR "partial seizures"
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Finding Sources
Reading File
Reading File
I now have comprehensive information from multiple authoritative sources. Here is a thorough overview of focal seizures.
Focal Seizures
Definition & Classification
Focal seizures (previously called partial seizures) arise from a neuronal network either discretely localized within one brain region or more broadly distributed but still within a single cerebral hemisphere. The 2017 ILAE classification replaced older terms with a system based on two key dimensions:
- Awareness — intact vs. impaired at any point during the seizure
- Onset type — motor vs. nonmotor
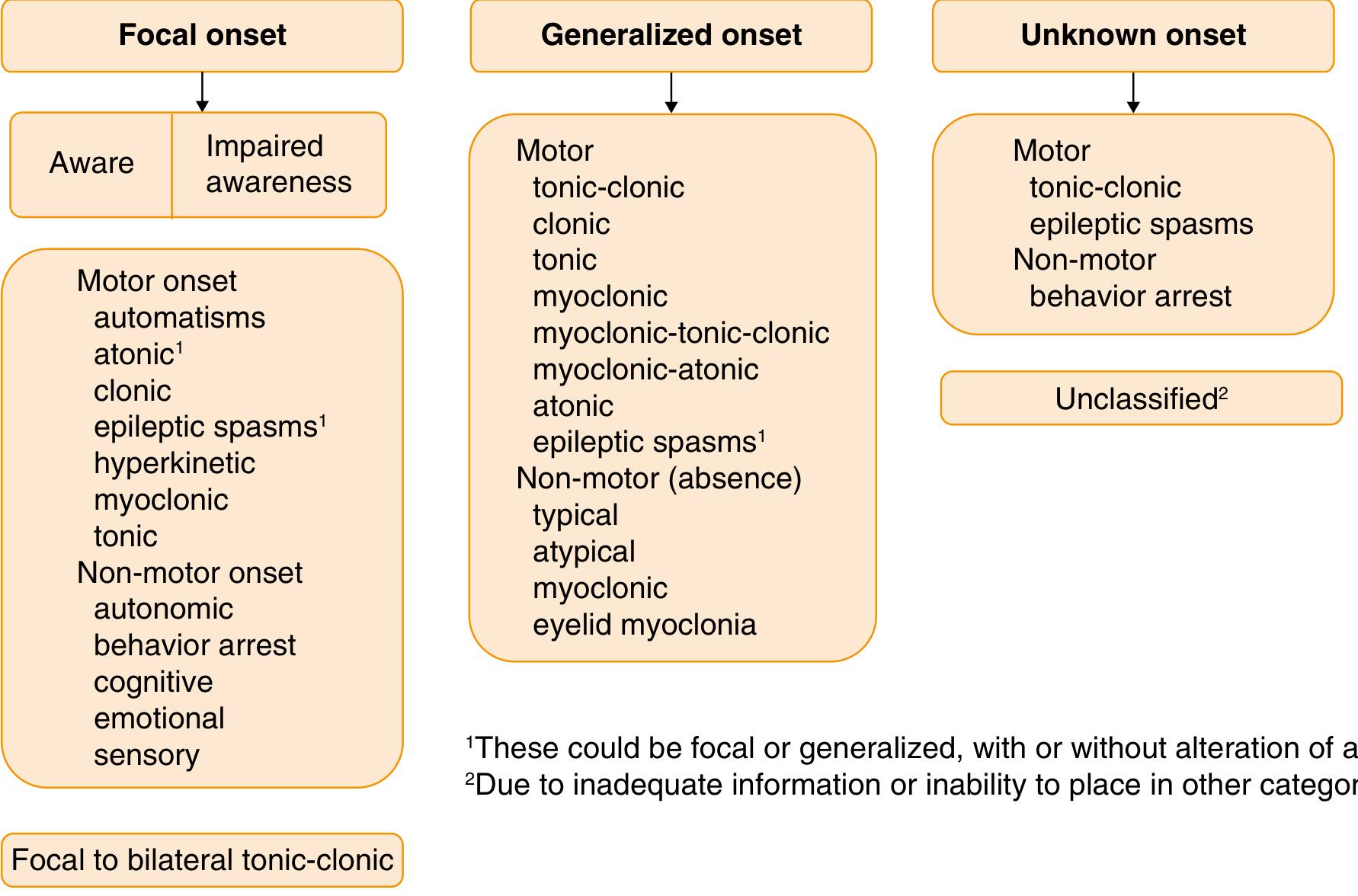
| Old Term (1981) | New Term (2017) |
|---|---|
| Simple partial seizure | Focal aware seizure |
| Complex partial seizure | Focal impaired awareness seizure |
| Secondarily generalized seizure | Focal to bilateral tonic-clonic seizure |
Types
1. Focal Aware Seizures (FAS)
Awareness is fully preserved throughout. Manifestations depend on which cortical region is involved:
- Motor onset: contralateral tonic, clonic, or myoclonic activity
- Sensory: paresthesias, visual phenomena, auditory sounds, vertigo, olfactory (acrid smell) or gustatory (metallic/bitter taste)
- Autonomic: flushing, sweating, piloerection, epigastric rising sensation
- Cognitive/emotional: déjà vu, fear, depersonalization, dreamlike states
When purely subjective, these seizures are commonly called auras. Duration is typically 20–60 seconds. — Goodman & Gilman's Pharmacological Basis of Therapeutics
2. Focal Impaired Awareness Seizures (FIAS)
Awareness is transiently impaired at some point. Key features:
- Begins with a motionless stare
- Automatisms: lip smacking, chewing, swallowing, picking/wringing hand movements, or more elaborate behaviors (emotional display, running)
- Patient is typically disoriented post-ictally; transition to full recovery may take seconds to hours
- Postictal deficits may include anterograde amnesia, aphasia, hemi-neglect, or visual loss from cortical inhibition
- Duration typically 30 seconds to 2 minutes — Harrison's Principles of Internal Medicine 22E
3. Focal to Bilateral Tonic-Clonic Seizures
- A focal seizure spreads to engage both hemispheres, evolving into tonic-clonic activity
- Most common when the focus is in the frontal lobe
- The focal onset is often missed by bystanders, who describe only the dramatic convulsive phase
- Distinguishing this from primary generalized onset has major treatment and prognostic implications
Localization by Seizure Type
| Clinical Type | Localization |
|---|---|
| Jacksonian (focal motor march) | Prerolandic (precentral) gyrus |
| Masticatory, salivation, speech arrest | Amygdaloid nuclei / opercular |
| Head/eye turning, tonic limb extension | Frontal / supplementary motor area |
| Somatosensory aura | Contralateral posterior rolandic cortex |
| Unformed lights, patterns | Occipital lobe |
| Auditory aura | Heschl gyri (superior temporal) |
| Vertigo | Superior temporal |
| Olfactory aura | Mesial temporal |
| Gustatory aura | Insula |
| Déjà vu, formed hallucinations, affective states | Temporal lobe (amygdala-hippocampal complex) |
| Automatisms | Temporal and frontal lobes |
— Adams and Victor's Principles of Neurology, 12th Ed.
Special Phenomena
Jacksonian March
Seizure activity "marches" from a restricted starting region (e.g., fingers) progressively along the motor homunculus to the entire extremity, reflecting the spread of ictal discharge over increasingly larger regions of motor cortex.
Todd's Paralysis
Focal postictal weakness lasting minutes to hours (rarely up to 1–2 days) in the body region involved in the seizure. Reflects postictal inhibition.
Epilepsia Partialis Continua
A rare, continuous focal motor seizure lasting hours to days; often refractory to medical therapy. — Harrison's
Focal Status Epilepticus
A series of focal seizures without recovery between them. Focal impaired awareness status presents as prolonged confused behavior; EEG shows continuous rhythmic discharges. Most common with frontal lobe onset.
EEG Features
- Interictal EEG may be normal or show focal epileptiform spikes/sharp waves
- Ictal EEG: rhythmic localized discharge (often 4–7 Hz), increasing in amplitude and decreasing in frequency as the seizure progresses
- Seizures from medial temporal or inferior frontal regions may be non-localizing on scalp EEG → intracranial electrode placement may be needed
- Focal slowing on EEG suggests localized parenchymal dysfunction — Harrison's / Kaplan & Sadock's Synopsis
Etiologies
Focal seizures imply an underlying focal structural or functional brain abnormality. Common causes include:
- Structural: brain tumor, cortical dysplasia, post-traumatic scar, vascular malformation (AVM, cavernoma), stroke, hippocampal sclerosis
- Metabolic: hyperosmolar states (focal seizures common in hyperosmolar coma), uremia
- Infectious/inflammatory: encephalitis, abscess
- Genetic: certain focal epilepsy syndromes (e.g., KCNT1-related epilepsy)
A focal seizure as a "warning symptom" — an aura before a larger seizure — is itself a focal seizure and points to a focal structural lesion requiring neuroimaging. — Goldman-Cecil Medicine
Treatment
First-Line Anti-Seizure Medications (ASMs) for Focal Epilepsy
| Drug Generation | Agents |
|---|---|
| Conventional | Carbamazepine, phenytoin, valproate, phenobarbital |
| Newer (preferred) | Lamotrigine, levetiracetam, lacosamide, eslicarbazepine, oxcarbazepine, brivaracetam, cenobamate, topiramate, zonisamide, gabapentin, tiagabine, perampanel |
Carbamazepine, lamotrigine, phenytoin, lacosamide, and valproic acid all act by inhibiting high-frequency neuronal firing via sodium channel blockade at therapeutically relevant concentrations. Lamotrigine and levetiracetam are now considered drugs of first choice by many guidelines. — Goodman & Gilman's; Swanson's Family Medicine
Surgical Treatment
When seizures are medically refractory, surgical resection of the epileptogenic focus is an option. The nature of the aura/focal symptoms guides localization of the target zone — a key reason for carefully eliciting the aura history.
Clinical Pearls
- Always ask about a warning — missing the aura/focal onset leads to misclassification as primary generalized epilepsy, with implications for drug choice and workup.
- Bizarre or stereotyped episodic behavior should prompt EEG evaluation before ruling out seizures.
- Focal onset implies structural disease until proven otherwise — MRI brain is mandatory.
- Distinguishing focal from generalized onset is essential: certain ASMs (e.g., carbamazepine) can worsen generalized epilepsies. — Harrison's Principles of Internal Medicine 22E
Sources: Harrison's Principles of Internal Medicine 22E (2025) | Goldman-Cecil Medicine | Adams and Victor's Principles of Neurology, 12th Ed. | Bradley and Daroff's Neurology in Clinical Practice | Goodman & Gilman's Pharmacological Basis of Therapeutics
Alcohol withdrawal
Finding Sources
Searching PubMed
"alcohol withdrawal syndrome" AND management
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
I now have comprehensive material to give a thorough, well-structured answer. Here it is:
Alcohol Withdrawal Syndrome (AWS)
Pathophysiology
Chronic alcohol use causes neuroadaptation: down-regulation of GABA-A receptors (reducing inhibitory tone) and up-regulation of NMDA glutamate receptors (increasing excitatory tone). The brain adapts to the constant presence of alcohol as a CNS depressant.
When alcohol is abruptly reduced or stopped, the underlying excitatory-inhibitory imbalance is unmasked, producing a state of CNS hyperexcitation with surges in catecholamines (elevated plasma, CSF, and urinary catecholamine levels).
Key point: Only a reduction, not necessarily complete cessation, of ethanol intake can trigger withdrawal. — Rosen's Emergency Medicine
Clinical Progression
AWS is a continuum of syndromes with characteristic timing:
| Stage | Features | Typical Onset After Last Drink |
|---|---|---|
| Mild | Tremulousness, anxiety, irritability, nausea/vomiting, diaphoresis, tachycardia, hypertension, insomnia | 6–12 hours |
| Moderate | Perceptual disturbances (alcoholic hallucinosis) | 8–48 hours |
| Severe | Withdrawal seizures | 12–48 hours |
| Life-threatening | Delirium tremens (DTs) | 48–96 hours (typically day 3–4) |
Withdrawal may begin before blood alcohol levels reach zero and can last up to 4–6 months as a protracted abstinence syndrome. — Maudsley Prescribing Guidelines, 15th ed.
Clinical Stages in Detail
1. Minor (Uncomplicated) Withdrawal
- Tremor (hands, tongue, eyelids), irritability, anorexia, nausea, tachycardia, diaphoresis, hypertension, hyperreflexia, vivid dreams
- Usually resolves within 48 hours
- Management: supportive care, hydration, vitamins
2. Alcoholic Hallucinosis
- Visual and auditory hallucinations (more accurately illusions — patients retain reality testing and intact sensorium)
- Onset 8–48 hours after cessation
- Distinguished from DTs by the clear sensorium — The Washington Manual of Medical Therapeutics
3. Withdrawal Seizures
- Typically one or a few brief generalized tonic-clonic convulsions
- Onset 12–48 hours (peak ~24 hours); can occur up to 5 days post-cessation
- May occur in the absence of other signs of withdrawal
- About 2% of individuals with alcohol use disorder experience withdrawal seizures; risk increases with older age, comorbidities, polydrug use, and higher alcohol intake
- Routine AEDs (antiepileptics) are not indicated for typical alcohol withdrawal seizures; treat with benzodiazepines
- Phenytoin does not prevent alcohol withdrawal seizures — Maudsley; Washington Manual
- If seizures occur despite adequate benzodiazepine loading, carbamazepine can be considered
4. Delirium Tremens (DTs)
- Develops in 3–5% of those admitted for withdrawal; appears ~72–96 hours after last drink
- Life-threatening — mortality 10–20% if untreated, ~5% with proper ICU management
- Features: profound confusion + agitation + marked tremor + vivid hallucinations (especially visual and tactile) + severe autonomic hyperactivity:
- Fever > 38.5°C
- BP > 140/90 mmHg
- Tachycardia, diaphoresis
- Paranoid delusions
- Often runs a course of 3–5 days regardless of treatment
Risk factors for DTs: prior DTs or withdrawal seizures, severe alcohol dependence, concurrent illness, multiple prior admissions, low Mg²⁺/K⁺, thiamine deficiency, inadequately treated withdrawal — Maudsley; Harrison's
DSM-5 Diagnostic Criteria
- Cessation or reduction of heavy, prolonged alcohol use
- ≥2 of the following within hours to days:
- Autonomic hyperactivity
- Hand tremor
- Insomnia
- Nausea or vomiting
- Transient hallucinations or illusions
- Psychomotor agitation
- Anxiety
- Generalized tonic-clonic seizures
Specifier: With perceptual disturbances — Rosen's Emergency Medicine / DSM-5
Assessment: CIWA-Ar Scale
The Clinical Institute Withdrawal Assessment for Alcohol, Revised (CIWA-Ar) is the standard 10-item severity scale, completable in ~5 minutes. Domains include:
- Nausea/vomiting
- Tremor
- Paroxysmal sweats
- Anxiety
- Agitation
- Tactile disturbances (formication — "bugs crawling under skin")
- Auditory/visual disturbances
- Headache
- Orientation/clouding of sensorium
Scoring:
- ≤10: mild — monitor, may not need pharmacotherapy
- 10–15: moderate — initiate pharmacotherapy
- >15: severe — urgent pharmacological treatment
- Max possible score: 67
Symptom-triggered dosing based on CIWA-Ar is preferred over fixed-dose schedules (which can lead to over- or undertreatment). — Maudsley; Washington Manual
Management
Step 1 — Immediate Assessment
- Full physical exam: liver disease, GI bleeding, arrhythmias, infection, hypoglycemia, electrolyte imbalances
- Avoid IV fluids unless clear indication (most patients are normohydrated or overhydrated)
Step 2 — Thiamine First
- Thiamine 100–500 mg IM/IV before any glucose (to prevent precipitating Wernicke's encephalopathy)
- Then 100 mg PO daily × ≥1 week
- Multivitamins with folic acid
Step 3 — Pharmacotherapy
Benzodiazepines — First-line
| Drug | Dose | Notes |
|---|---|---|
| Chlordiazepoxide (long-acting) | 25–50 mg PO q4–6h Day 1, taper over 5 days | Preferred for most patients; avoid in severe liver disease |
| Diazepam (long-acting) | 10 mg IV q5–20 min (acute/severe) | Long half-life provides smooth self-tapering |
| Lorazepam (short-acting) | 2–4 mg IV q15–20 min | Preferred in liver failure or brain damage; must be given q4h to avoid blood-level troughs |
| Oxazepam (short-acting) | 15–30 mg PO q6–8h | Renally excreted; best for hepatic failure |
- Long-acting benzodiazepines (diazepam, chlordiazepoxide) are preferred for seizure prophylaxis — their self-tapering pharmacokinetics reduce the risk of breakthrough seizures
- Symptom-triggered protocols are superior to fixed-dose schedules
Delirium Tremens — ICU Management
- High-dose benzodiazepines (up to 800 mg/day chlordiazepoxide reported)
- Refractory DTs: propofol or closely monitored dexmedetomidine as adjuncts
- Dexmedetomidine alone is not adequate — it does not target GABA/glutamate systems, can mask autonomic signs without preventing seizures, and has not improved outcomes in trials
- Antipsychotics are not recommended for AWS (do not address underlying mechanism; lower seizure threshold)
- Correct hypomagnesemia, hypokalemia, hypoglycemia — Washington Manual; Harrison's
Step 4 — Phenobarbital (Adjunct/Alternative)
A 2024 systematic review and meta-analysis (PMID 37923363) found phenobarbital effective for alcohol withdrawal in the ED, offering an alternative especially for benzodiazepine-refractory cases.
Setting of Care
Community/Outpatient Detox — Suitable when:
- Mild symptoms with good social support and 24-hour supervision
- No prior DTs or withdrawal seizures
- Regular consumption <30 units/day, SADQ <30
- Daily medication pickup and professional review feasible
Inpatient Detox — Required when:
- Regular consumption >30 units/day or SADQ >30
- History of seizures or DTs
- Concurrent benzodiazepine use or polysubstance use
- Comorbid physical or psychiatric illness
- Pregnancy, minors, elderly
- Homeless or no social support
- Failed community detox — Maudsley Prescribing Guidelines
Electrolyte & Nutritional Considerations
Alcoholic patients are particularly susceptible to:
- Hypomagnesemia (lowers seizure threshold)
- Hypokalemia
- Hypoglycemia
- Hypophosphatemia
- Thiamine deficiency (→ Wernicke's encephalopathy if glucose given first)
Fluid losses from fever, diaphoresis, and vomiting during DTs can be considerable.
Key Clinical Pearls
- Only reduction in intake is needed to trigger withdrawal — patients can be in AWS while still drinking.
- Previous AWS episodes predict future severity (kindling effect — each withdrawal lowers the seizure threshold).
- The most important risk factor for AWS in hospitalized patients is a prior history of withdrawal episodes.
- Phenytoin is ineffective for alcohol withdrawal seizures — do not use it.
- Antipsychotics treat hallucinations but do not prevent seizures and can lower seizure threshold — use with caution only as adjuncts to benzodiazepines.
- DTs can skip the usual progression and present without prior mild symptoms.
Sources: Harrison's Principles of Internal Medicine 22E (2025) | The Washington Manual of Medical Therapeutics | Maudsley Prescribing Guidelines in Psychiatry, 15th ed. | Rosen's Emergency Medicine | Kaplan & Sadock's Synopsis of Psychiatry | PMID 37923363 — Phenobarbital for AWS: Systematic Review & Meta-Analysis, Acad Emerg Med 2024 | PMID 38747203 — GRACE-4 Practice Guideline for AUD in the ED, Acad Emerg Med 2024
This is a shared conversation. Sign in to Orris to start your own chat.