Essay answer on CML 10 marks question blood and bone marrow findings in CML from Harrison with proper flowcharts and diagrams??
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Searching Images
CML chronic myeloid leukemia blood smear peripheral blood bone marrow
Loading Image
Searching Images
CML peripheral blood smear left shift myelocytes metamyelocytes basophilia leukocytosis
Reading File
I now have comprehensive content from Harrison's. Let me compile the complete essay answer with flowcharts and diagrams.
Blood and Bone Marrow Findings in Chronic Myeloid Leukemia (CML)
[Harrison's Principles of Internal Medicine, 22nd Ed., Chapter 110 — p. 878–882]
1. Introduction
Chronic Myeloid Leukemia (CML) is a clonal myeloproliferative neoplasm of the pluripotent hematopoietic stem cell, defined by the presence of the BCR::ABL1 fusion gene resulting from the reciprocal translocation t(9;22)(q34.1;q11.2) — the Philadelphia (Ph) chromosome. The disease has a characteristic triphasic natural history: chronic phase → accelerated phase → blast crisis. Peripheral blood and bone marrow findings are central to diagnosis, prognostication, and monitoring.
2. The Philadelphia Chromosome & Pathophysiology
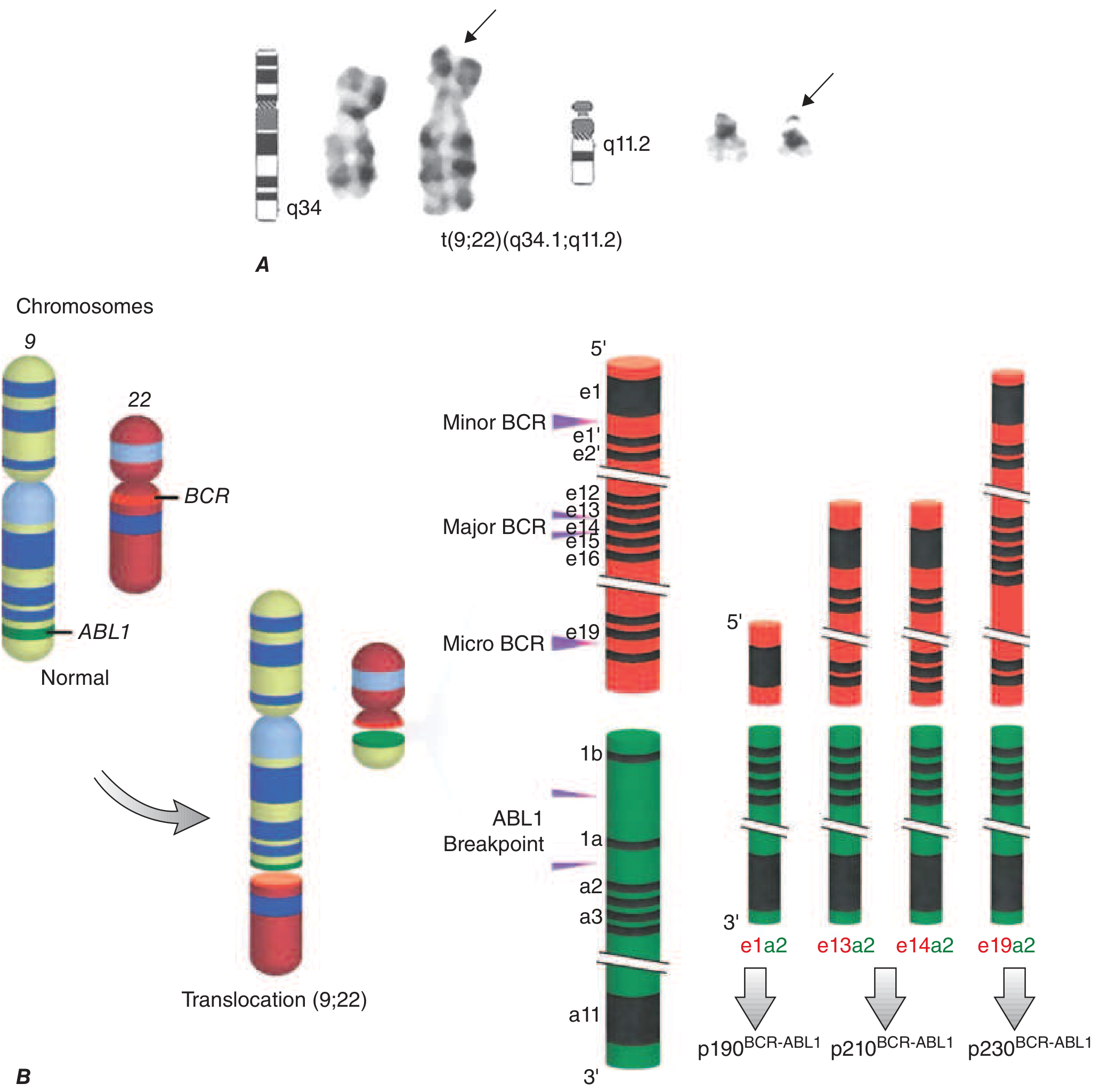
Harrison's Fig. 110-1: A. The Philadelphia chromosome cytogenetic abnormality. B. Breakpoints in chromosome 9 (ABL1) and chromosome 22 (BCR) generate three BCR::ABL1 oncoproteins — p210 (most common in CML), p190 (Ph+ ALL), and p230 (rare, indolent).
The t(9;22)(q34.1;q11.2) is present in >90% of classical CML cases. The hybrid BCR::ABL1 oncogene encodes p210^BCR::ABL1 (major breakpoint), a constitutively active tyrosine kinase that drives:
- ↑ Proliferation (via RAS, MAPK, STAT, PI3K, MYC)
- ↓ Apoptosis
- ↑ Genetic instability → disease progression
3. Peripheral Blood Findings (Chronic Phase)
╔══════════════════════════════════════════════════════════════════╗
║ PERIPHERAL BLOOD FINDINGS IN CML (CHRONIC PHASE) ║
╠══════════════════════════════════════════════════════════════════╣
║ PARAMETER │ FINDING ║
╠════════════════════════╪═════════════════════════════════════════╣
║ WBC Count │ 10–500 × 10⁹/L (leukocytosis) ║
║ Differential │ Left-shifted granulopoiesis ║
║ │ Neutrophils predominate ║
║ │ Bands, Myelocytes, Metamyelocytes ║
║ │ Promyelocytes, Blasts (usually ≤5%) ║
║ Basophils │ INCREASED (≥7% in 10% of patients) ║
║ Eosinophils │ Frequently increased ║
║ Blasts (PB) │ ≥3% in 8–10% of patients ║
║ Platelets │ Thrombocytosis (common) ║
║ │ >450 × 10⁶/L in 30–35% ║
║ │ Thrombocytopenia (rare, poor prognosis) ║
║ Hemoglobin │ Anemia in ~1/3 patients ║
║ │ Hgb <10 g/dL in 10–15% ║
║ Count oscillations │ Cyclic in 10–20% without treatment ║
╚════════════════════════╧═════════════════════════════════════════╝
Key biochemical abnormalities (peripheral blood):
| Parameter | Finding |
|---|---|
| Leukocyte Alkaline Phosphatase (LAP) score | LOW (distinguishes CML from leukemoid reaction) |
| Vitamin B₁₂ | HIGH (due to ↑ transcobalamin from granulocytes) |
| Uric acid | HIGH (hyperuricemia — ↑ cell turnover) |
| Lactic dehydrogenase (LDH) | HIGH |
| Lysozyme | HIGH |
4. The "Left Shift" — Flowchart of CML Peripheral Blood Differential
PERIPHERAL BLOOD DIFFERENTIAL IN CML
══════════════════════════════════════
NORMAL GRANULOCYTE SERIES:
Myeloblast → Promyelocyte → Myelocyte → Metamyelocyte → Band → Neutrophil
(marrow) (marrow) (marrow) (marrow) (blood) (blood)
IN CML (LEFT SHIFT — all stages seen in peripheral blood):
┌─────────────┐
│ Myeloblast │ ≤5% (chronic phase)
└──────┬──────┘
│
┌──────▼──────┐
│Promyelocyte │ Present
└──────┬──────┘
│
┌──────▼──────┐
│ Myelocyte │ Predominant (more than metamyelocytes — KEY FEATURE)
└──────┬──────┘
│
┌──────▼──────────┐
│ Metamyelocyte │ Present
└──────┬──────────┘
│
┌──────▼──────┐
│ Band │ Present
└──────┬──────┘
│
┌──────▼──────┐
│ Neutrophil │ Present (majority of WBC)
└─────────────┘
+
┌─────────────┐ ┌─────────────┐
│ Basophilia │ + │ Eosinophilia│ (Characteristic findings)
└─────────────┘ └─────────────┘
KEY DISTINCTION from Leukemoid Reaction:
CML: Myelocytes > Metamyelocytes + LOW LAP score
Leukemoid: Metamyelocytes > Myelocytes + HIGH LAP score
5. Bone Marrow Findings (Chronic Phase)
╔══════════════════════════════════════════════════════════════════╗
║ BONE MARROW FINDINGS IN CML (CHRONIC PHASE) ║
╠══════════════════════════════════════════════════════════════════╣
║ PARAMETER │ FINDING ║
╠═════════════════════════╪════════════════════════════════════════╣
║ Cellularity │ HYPERCELLULAR (90–100%) ║
║ Myeloid series │ MARKED MYELOID HYPERPLASIA ║
║ M:E Ratio │ 15–20:1 (markedly elevated) ║
║ Blasts (marrow) │ ≤5% (chronic phase) ║
║ │ ≥5% in 5% of patients at presentation ║
║ Basophils (marrow) │ ≥5% in 10–15% of patients ║
║ Megakaryocytes │ Increased, small/dwarf megakaryocytes ║
║ Erythropoiesis │ Relatively suppressed ║
║ Reticulin fibrosis │ Grade 3–4 in 30–40% of patients ║
║ Collagen fibrosis │ RARE at diagnosis ║
║ "Spent phase" │ Myelophthisis/burnt-out marrow (rare ║
║ (myelofibrosis) │ now in TKI era) ║
╚═════════════════════════╧════════════════════════════════════════╝
Important: In 10% of patients, the percentage of marrow blasts and basophils can be significantly higher than in the peripheral blood, conferring worse prognosis or even representing disease transformation.
6. Diagnostic Flowchart for CML
SUSPECTED CML — DIAGNOSTIC ALGORITHM
═══════════════════════════════════════════
Unexplained sustained leukocytosis ± splenomegaly
│
▼
┌────────────────────────┐
│ Peripheral Blood CBC │
│ + Differential │
└───────────┬────────────┘
│
┌─────────▼──────────────────────────┐
│ Left-shifted leukocytosis │
│ + basophilia + low LAP score │
└─────────┬──────────────────────────--┘
│
▼
┌────────────────────────┐
│ Bone Marrow Aspiration│
│ + Trephine Biopsy │
└───────────┬────────────┘
│
┌─────────▼──────────────────────────┐
│ Hypercellular; myeloid hyperplasia │
│ M:E ratio 15–20:1; blasts ≤5% │
└─────────┬──────────────────────────--┘
│
▼
┌────────────────────────┐
│ Cytogenetics (G-banding)│
│ t(9;22)(q34.1;q11.2) │
│ Philadelphia chromosome │
└───────────┬────────────┘
│
┌─────────┴─────────┐
│ │
Ph+ (90%) Ph- (~10%)
│ │
▼ ▼
CML Confirmed FISH / RT-PCR
for BCR::ABL1
│
┌──────────┴──────────┐
│ │
BCR::ABL1 + BCR::ABL1 –
(Ph- CML) Consider atypical CML,
TKI therapy CMML, MDS/MPN
7. CML Phases — Evolving Blood & Marrow Findings
╔══════════════════════════════════════════════════════════════════════════╗
║ CML PHASES: BLOOD AND BONE MARROW PROGRESSION ║
╠══════════════════════════════╦══════════════════╦════════════════════════╣
║ PARAMETER ║ CHRONIC PHASE ║ ACCELERATED/BLAST ║
╠══════════════════════════════╬══════════════════╬════════════════════════╣
║ WBC (×10⁹/L) ║ 10–500 ║ Rising, uncontrolled ║
║ Blasts – Peripheral Blood ║ <5% ║ ≥10% (AP) / ≥20% (BP) ║
║ Blasts – Bone Marrow ║ ≤5% ║ ≥15% (AP) / ≥20% (BP) ║
║ Basophils – Blood ║ ↑ (mild) ║ ≥20% (AP criterion) ║
║ Platelets ║ Often ↑ ║ Persistent ↓ (<100×10⁹)║
║ Basophils – Marrow ║ ≤5–15% ║ ≥20% (AP criterion) ║
║ Marrow cellularity ║ Hypercellular ║ Hypercellular/fibrotic ║
║ M:E ratio ║ 15–20:1 ║ Distorted further ║
║ Reticulin fibrosis ║ 30–40% grade 3–4║ ↑↑ (common) ║
║ Additional cytogenetics ║ Ph only (95%) ║ Double Ph, +8, i(17q) ║
╚══════════════════════════════╩══════════════════╩════════════════════════╝
AP = Accelerated Phase; BP = Blast Phase/Blast Crisis
8. Presenting Signs — Summary Table (Harrison's Table 110-1)
| Parameter | Percentage at Presentation |
|---|---|
| Age ≥60 years | 40–50% (median 55–65 years) |
| Splenomegaly | 30% |
| Hepatomegaly | 5–10% |
| Lymphadenopathy | 5% |
| Hgb <10 g/dL | 10–15% |
| Platelets >450 × 10⁶/L | 30–35% |
| Platelets <100 × 10⁶/L | 3–5% |
| WBC ≥50 × 10⁹/L | 35–40% |
| Marrow blasts ≥5% | 5% |
| Marrow basophils ≥5% | 10–15% |
| PB blasts ≥3% | 8–10% |
| PB basophils ≥7% | 10% |
| Additional chromosomal abnormalities | 4–5% |
9. Cytogenetic and Molecular Findings
CYTOGENETICS & MOLECULAR DIAGNOSIS IN CML
═══════════════════════════════════════════════
G-banding (Karyotype)
↓
t(9;22)(q34.1;q11.2) detected in 90% of cases
↓
Philadelphia (Ph) chromosome = der(22)
┌──────────────────────────────────────────────────────┐
│ Additional chromosome analysis findings in CML: │
│ • Trisomy 8 → adverse prognosis in AP/BP │
│ • Double Ph → disease acceleration │
│ • Isochromosome 17q → poor prognosis │
│ • 17p deletion → TP53 loss │
│ • 20q- → progression signal │
│ • Additional CAs in 4–5% at diagnosis │
└──────────────────────────────────────────────────────┘
FLUORESCENCE IN SITU HYBRIDIZATION (FISH)
BCR::ABL1 dual-color dual-fusion probe
→ Detects Ph+ cells in blood OR marrow
→ More sensitive than G-banding
RT-PCR (Quantitative)
→ Detects BCR::ABL1 transcripts:
e13a2 (b2a2) → p210 (most common CML)
e14a2 (b3a2) → p210 (equally common)
e1a2 → p190 (Ph+ ALL, rare CML, worse prognosis)
e19a2 → p230 (rare, indolent CML)
→ Used for monitoring response to TKI therapy
→ International Scale (IS): reported as %BCR::ABL1
10. CML vs. Leukemoid Reaction — Key Differential
| Feature | CML | Leukemoid Reaction |
|---|---|---|
| WBC | Very high (>50,000) | Elevated (<50,000 usually) |
| Left shift | Myelocytes > Metamyelocytes | Metamyelocytes > Myelocytes |
| Basophilia | Present | Absent |
| LAP score | LOW (<20) | HIGH (>100) |
| Philadelphia chromosome | Present | Absent |
| BCR::ABL1 | Positive | Negative |
| Splenomegaly | Common | Related to cause |
| Cause | Clonal neoplasm | Reactive (infection, etc.) |
11. Summary Flowchart — CML Blood & Bone Marrow at a Glance
CML — BLOOD & BONE MARROW AT A GLANCE
══════════════════════════════════════
BCR::ABL1 (p210) — constitutive tyrosine kinase activity
│
▼
Clonal expansion of pluripotent hematopoietic stem cell
│
┌──────────────┼──────────────┐
│ │ │
┌─────▼──────┐ ┌────▼──────┐ ┌────▼──────────┐
│ Peripheral │ │ Bone │ │ Spleen / │
│ Blood │ │ Marrow │ │ Extramedullary│
└─────┬───────┘ └────┬──────┘ └───────────────┘
│ │
┌─────▼──────────────▼──────────────────────────────┐
│ PERIPHERAL BLOOD BONE MARROW │
│ • Leukocytosis 10–500×10⁹ • Hypercellular 90–100%│
│ • Full myeloid left shift • Myeloid hyperplasia │
│ • Basophilia + Eosinophilia • M:E ratio 15–20:1 │
│ • Blasts ≤5% • Blasts ≤5% │
│ • Thrombocytosis (common) • Dwarf megakaryocytes │
│ • Anemia (1/3) • ↑ Reticulin fibrosis │
│ • LOW LAP score • Ph chromosome+ │
│ • ↑ B12, LDH, uric acid │
└──────────────────────────────────────────────────--┘
│
┌─────────────┼─────────────┐
│ │ │
┌──────▼──────┐ ┌───▼───────┐ ┌───▼──────────┐
│ CHRONIC │ │ACCELERATED│ │ BLAST CRISIS │
│ PHASE │ │ PHASE │ │ │
│ Blasts <5% │ │Blasts │ │Blasts ≥20% │
│ │ │15–19% │ │(Myeloid 70% / │
│ │ │Basophils │ │Lymphoid 30%) │
│ │ │≥20% │ │ │
└─────────────┘ └───────────┘ └───────────────┘
12. Key Points to Remember (10-Mark Essay)
- CML = BCR::ABL1 fusion gene from t(9;22)(q34.1;q11.2) → Philadelphia chromosome
- Peripheral blood: massive leukocytosis (10–500×10⁹/L), full spectrum of myeloid cells ("left shift"), basophilia, eosinophilia, thrombocytosis, anemia in 1/3; blasts usually <5%
- Hallmark of blood smear: myelocytes predominate over metamyelocytes (reverse of leukemoid reaction)
- LAP score is LOW — critical diagnostic distinction from leukemoid reaction
- Bone marrow: markedly hypercellular, myeloid hyperplasia, M:E ratio 15–20:1, blasts ≤5%, dwarf megakaryocytes, reticulin fibrosis (30–40%)
- Diagnosis confirmed by cytogenetics (G-banding), FISH, and RT-PCR for BCR::ABL1
- Biochemistry: ↑ B₁₂, ↑ LDH, ↑ uric acid, ↑ lysozyme, LOW LAP
- Disease progression is marked by ↑ blasts, ↑ basophils, additional cytogenetic abnormalities (trisomy 8, double Ph, i(17q)), worsening cytopenias
- Bone marrow blasts can be higher than peripheral blood in 10% of patients — marrow biopsy is essential
- With TKI therapy (imatinib era), reticulin fibrosis resolves and the "spent phase" of myelofibrosis is now extremely rare
Source: Harrison's Principles of Internal Medicine, 22nd Edition (2025), Chapter 110: Chronic Phase Chronic Myeloid Leukemia, pp. 878–882.
This is a shared conversation. Sign in to Orris to start your own chat.