Microbiology diagnosis of Intestinal and urogenital protozoa (Entamoeba histolytica (Amebae), Balantidium coli (Ciliates), Giardia lamblia, Trichomonas vaginalis (Flagellates), Cryptosporidium parvum and Isospora belli (Sporozoa)
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive material from all relevant textbooks. Let me compile the complete, well-organized answer.
Microbiology Diagnosis of Intestinal and Urogenital Protozoa
Overview of Diagnostic Approaches
Four main methods are used across all these organisms:
- Microscopy — wet mount, concentration techniques, permanent stained smears (trichrome, iron hematoxylin, modified acid-fast)
- Culture — selected organisms only
- Antigen detection — EIA/immunochromatographic assays
- Molecular methods — NAATs/PCR (increasingly the gold standard)
1. Entamoeba histolytica (Amebae)
Morphology
- Trophozoite: 12–60 µm; single nucleus with central karyosome and fine peripheral chromatin; pseudopods; motile. The hallmark diagnostic feature is ingested erythrocytes within the cytoplasm (distinguishes E. histolytica from the morphologically identical E. dispar).
- Cyst: 10–20 µm; round; 1–4 nuclei; chromatoid bars with rounded ends; glycogen vacuole.
Specimen Collection
- Stool (primary): fresh liquid stools (trophozoites) or formed stools (cysts). Fix immediately in PVA or SAF to preserve fragile trophozoites.
- Liquid diarrheal specimens or sigmoidoscopic aspirates/biopsies may yield trophozoites.
- Liver abscess aspirate: reddish-brown "anchovy paste" fluid; trophozoites found at the periphery, not in the necrotic center.
Diagnostic Methods
| Method | Notes |
|---|---|
| Wet mount | Motile trophozoites with unidirectional pseudopod movement; cysts detected by iodine preparation |
| Concentration (formalin-ethyl acetate) | Increases yield for cysts |
| Permanent stain (trichrome / iron hematoxylin) | Definitive identification of nuclear morphology |
| Stool antigen EIA | Sensitive and E. histolytica-specific; cannot detect E. dispar |
| Serology (IHA, ELISA) | Detects antibodies; valuable for extraintestinal (hepatic abscess) disease; 90%+ sensitivity in invasive disease |
| PCR / NAAT | Most sensitive and specific; differentiates E. histolytica from E. dispar; used by reference labs |
| Culture | Robinson's medium; rarely used clinically |
Key diagnostic point: Microscopy alone cannot distinguish E. histolytica from E. dispar. Only erythrophagocytosis (trophozoites with ingested RBCs), antigen testing, or PCR can confirm true E. histolytica. Serology is the test of choice for amebic liver abscess. — Sherris & Ryan's Medical Microbiology, 8th ed.
2. Balantidium coli / Balantioides coli (Ciliates)
Morphology
- Trophozoite: the largest intestinal protozoan (~50–100 µm × 40–70 µm); entire surface covered with cilia; prominent kidney-bean-shaped macronucleus (for identification); smaller micronucleus beside it; cytostome (mouth) at anterior end.
- Cyst: 50–70 µm; spherical; enclosed in a thick double wall; the macronucleus and cilia may still be visible.
Specimen
- Stool (fresh or fixed). Trophozoites are found in liquid/dysenteric stools; cysts in formed stools.
- Intestinal biopsy in invasive cases (colonic ulcers).
Diagnostic Methods
| Method | Notes |
|---|---|
| Direct wet mount | Trophozoites show rapid rotary/tumbling motility due to cilia; prominent macronucleus visible (Fig. 65.12H in Henry's) |
| Concentration | Formalin-ethyl acetate for cysts |
| Permanent stain (iron hematoxylin) | Reveals kidney-bean macronucleus of cysts (Fig. 65.12I) |
| Stool O&P exam | Standard ova and parasite examination; organism is unmistakable due to size |
No antigen or PCR tests are widely available; diagnosis rests on microscopic morphology. The organism is distinctive enough that experienced microscopists rarely miss it. — Henry's Clinical Diagnosis and Management by Laboratory Methods
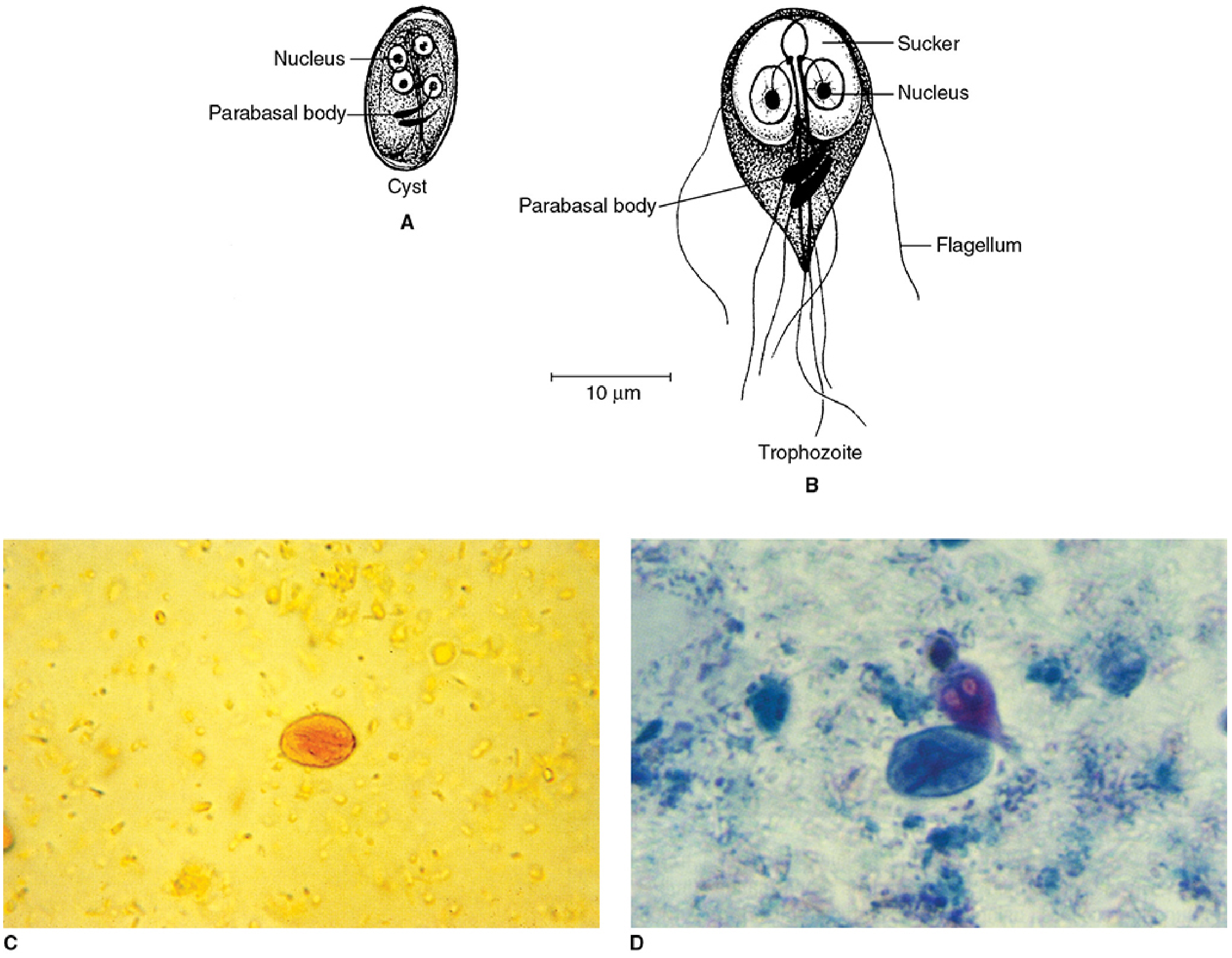
3. Giardia lamblia / duodenalis (Flagellates)
Morphology
- Trophozoite: 9–21 µm × 5–15 µm; stingray/pear-shaped; 2 nuclei resembling spectacled eyes; large ventral sucking disc; 4 pairs of flagella; "falling leaf" tumbling motility in fresh specimens.
- Cyst: 8–19 µm; oval; 4 nuclei (when mature); curved median bodies give a "smiley face" appearance; cytoplasm may be retracted from wall.

Specimen
- Stool (primary): cyst shedding is intermittent — examine 3 specimens collected on separate days for >90% sensitivity.
- Duodenal aspirate or small bowel biopsy (when stool exams negative but clinical suspicion high).
- Duodenal string test (Enterotest capsule): swallowed capsule retrieves bile-stained duodenal fluid.
Diagnostic Methods
| Method | Notes |
|---|---|
| Wet mount + iodine | Detects cysts and motile trophozoites |
| Concentration + permanent stain (trichrome) | Definitive morphologic identification |
| Stool antigen EIA / ELISA | Routine in most labs; detects Giardia-specific antigens; highly sensitive and specific (>95%) |
| Rapid immunochromatographic (point-of-care) assay | Sensitivity 80–90%; newer formats achieve >95% |
| NAAT / PCR | Most sensitive; included in multiplex stool GI panels (e.g., BioFire FilmArray) |
| Duodenal biopsy | Reveals trophozoites attached to brush border; also shows any associated pathology (e.g., nodular lymphoid hyperplasia) |
Barium studies are not useful and impair subsequent stool examination. — Yamada's Textbook of Gastroenterology, 7th ed.
4. Trichomonas vaginalis (Flagellates)
Morphology
- Trophozoite only (no cyst stage): 10–23 µm; pear-shaped; 4 anterior flagella + 1 posterior flagellum forming the outer edge of an undulating membrane that extends only halfway along the body (differentiates it from Pentatrichomonas hominis); prominent axostyle protrudes posteriorly; single nucleus.
- Motility: tumbling/rotary in fresh wet mounts.
Specimen
- Female: vaginal swab (highest yield), cervical swab, urine sediment.
- Male: urethral swab, prostatic fluid, urine sediment (first-catch).
Diagnostic Methods
| Method | Sensitivity | Notes |
|---|---|---|
| Direct wet mount | ~51–65% | Rapid, low cost; requires immediate examination of fresh specimen; motile organisms confirm diagnosis |
| Culture (Diamond's medium / InPouch TV pouch) | ~75–96% | Former gold standard; 5–7 days incubation |
| Antigen detection (OSOM Trichomonas rapid test) | ~83–90% | Point-of-care; usable in clinic |
| Papanicolaou smear / Giemsa stain | ~50–60% | Incidental finding; confirm positives with NAAT |
| NAAT (PCR, LAMP — e.g., Xpert TV) | >95–99% | Current gold standard; FDA-cleared for both male and female specimens (Xpert TV); included in multiplex STI panels |
NAAT is now the recommended method per CDC 2021 STI guidelines. Wet mount is acceptable only when NAATs are unavailable. — Henry's Clinical Diagnosis and Management by Laboratory Methods
5. Cryptosporidium parvum (Sporozoa / Coccidia)
Morphology
- Oocyst (the diagnostic stage in stool): small, 4–6 µm; round/oval; contains 4 sporozoites (no sporocyst); passed fully sporulated and immediately infectious.
- Trophozoites, merozoites, and other stages are found within the brush-border vacuoles of intestinal epithelial cells on biopsy.
Specimen
- Stool (primary): formed or watery stool in 10% formalin or SAF. Multiple samples increase sensitivity.
- Intestinal biopsy in immunocompromised patients with heavy burden.
- Bile/respiratory specimens in disseminated immunocompromised infection.
Diagnostic Methods
| Method | Notes |
|---|---|
| Modified acid-fast stain (Kinyoun / Ziehl-Neelsen) | Oocysts stain pink-red against a blue or green background; variable (some unstained "ghost forms"); standard diagnostic method |
| Auramine-O fluorescent stain | More sensitive than modified acid-fast; oocysts fluoresce bright yellow |
| DFA (direct fluorescent antibody) | Detects oocysts with fluorescein-labeled monoclonal antibodies; highly sensitive and specific |
| Stool antigen EIA / ELISA | Widely used; detects Cryptosporidium antigen; sensitivity >90% |
| Rapid immunochromatographic assay | Point-of-care; combined kits for Giardia + Cryptosporidium |
| NAAT / PCR | Most sensitive; included in multiplex GI panels; especially valuable in low-oocyst-burden states |
| Histopathology (PAS, H&E, Giemsa) | Biopsy shows small spherical organisms in apical brush border of enterocytes; PAS-positive |
Routine O&P exam often misses Cryptosporidium — laboratories must perform modified acid-fast or EIA specifically when requested. — Yamada's Textbook of Gastroenterology, 7th ed.
6. Cystoisospora belli (formerly Isospora belli) (Sporozoa / Coccidia)
Morphology
- Oocyst: large, ~12 × 30 µm; elongated ellipse; when freshly passed, contains a single sporoblast (unsporulated); after 24–48 h at room temperature → 2 sporocysts, each containing 4 sporozoites (total: 8 sporozoites). This is the largest intestinal coccidian oocyst.
- Eosinophilia may be present (unique among intestinal protozoa).
Specimen
- Stool (primary): oocyst shedding is low-level and intermittent — examine multiple stool samples; oocyst concentration helps.
- Duodenal aspirate or small-bowel biopsy (with electron microscopy) when stool exams repeatedly negative.
Diagnostic Methods
| Method | Notes |
|---|---|
| Direct wet mount | Large oocysts visible but may be sparse; allow specimen to sit 24–48 h for sporulation to confirm morphology |
| Concentration (formalin-ethyl acetate) | Essential given low oocyst burden |
| Modified acid-fast stain (Kinyoun) | Oocysts stain pink-red; more uniformly positive than Cyclospora |
| Hot safranin stain | Provides uniform red staining of oocysts |
| UV fluorescence | Oocysts autofluoresce (like Cyclospora) |
| Intestinal biopsy | Intracellular stages (merozoites, gametocytes) visible with H&E or electron microscopy |
| NAAT | Effective; being incorporated into newer multiplex GI panels |
The large size (~25–30 µm) distinguishes C. belli oocysts from those of Cryptosporidium (4–6 µm) and Cyclospora (8–10 µm). — Harrison's Principles of Internal Medicine, 22nd ed.
Comparative Summary Table
| Organism | Class | Diagnostic Stage | Primary Specimen | Key Stain / Method |
|---|---|---|---|---|
| Entamoeba histolytica | Amoeba | Trophozoite (RBCs inside) + Cyst | Stool, liver aspirate | Trichrome; antigen EIA; PCR |
| Balantidium coli | Ciliate | Trophozoite (ciliated, huge) + Cyst | Stool | Wet mount; iron hematoxylin |
| Giardia lamblia | Flagellate | Cyst + Trophozoite | Stool, duodenal aspirate | Trichrome; antigen EIA; NAAT |
| Trichomonas vaginalis | Flagellate | Trophozoite (no cyst) | Vaginal swab, urine | Wet mount; NAAT (Xpert TV) |
| Cryptosporidium parvum | Sporozoa | Oocyst (4–6 µm) | Stool | Modified acid-fast; DFA; EIA; NAAT |
| Cystoisospora belli | Sporozoa | Oocyst (12×30 µm) | Stool, duodenal aspirate | Modified acid-fast; wet mount; NAAT |
Key Diagnostic Pearls
- E. histolytica vs. E. dispar: Morphologically identical — erythrophagocytosis OR antigen EIA/PCR required for definitive species identification.
- Balantidium coli: Largest intestinal protozoan; the macronucleus and cilia make it unmistakable on wet mount.
- Giardia: Cyst shedding is intermittent; examine 3 stools on separate days. EIA/NAAT now routinely preferred over microscopy.
- Trichomonas vaginalis: No cyst stage — only trophozoites; wet mount is only 51–65% sensitive; NAAT is now the gold standard.
- Cryptosporidium: Must specifically request modified acid-fast stain — not detected on routine O&P; tiny oocysts (4–6 µm) may be overlooked.
- Cystoisospora belli: The largest coccidian oocyst in stool; eosinophilia is a distinguishing clue; low-level shedding mandates multiple specimens.
Sources: Sherris & Ryan's Medical Microbiology, 8th ed. | Henry's Clinical Diagnosis and Management by Laboratory Methods, 24th ed. | Yamada's Textbook of Gastroenterology, 7th ed. | Harrison's Principles of Internal Medicine, 22nd ed. | Tietz Textbook of Laboratory Medicine, 7th ed.
This is a shared conversation. Sign in to Orris to start your own chat.