Endometriosis gynaecology
Reading File
Finding Sources
Searching PubMed
endometriosis management treatment
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content covering all major aspects of endometriosis. Let me compile the full clinical summary:
Endometriosis — Gynaecology
Definition
Endometriosis is the presence of endometrial-like tissue (glands and/or stroma) outside the uterus. It affects approximately 10% of women of reproductive age and is found in nearly 50% of women with infertility or chronic pelvic pain.
Types
| Type | Description |
|---|---|
| Superficial peritoneal endometriosis | Implants on peritoneal surfaces |
| Ovarian endometriosis (endometrioma) | "Chocolate cysts" — cysts filled with hemosiderin-laden old blood |
| Deep infiltrating endometriosis (DIE) | Nodules ≥5 mm beneath peritoneum; cul-de-sac, rectosigmoid, bladder; highest malignant transformation risk |
Pathogenesis
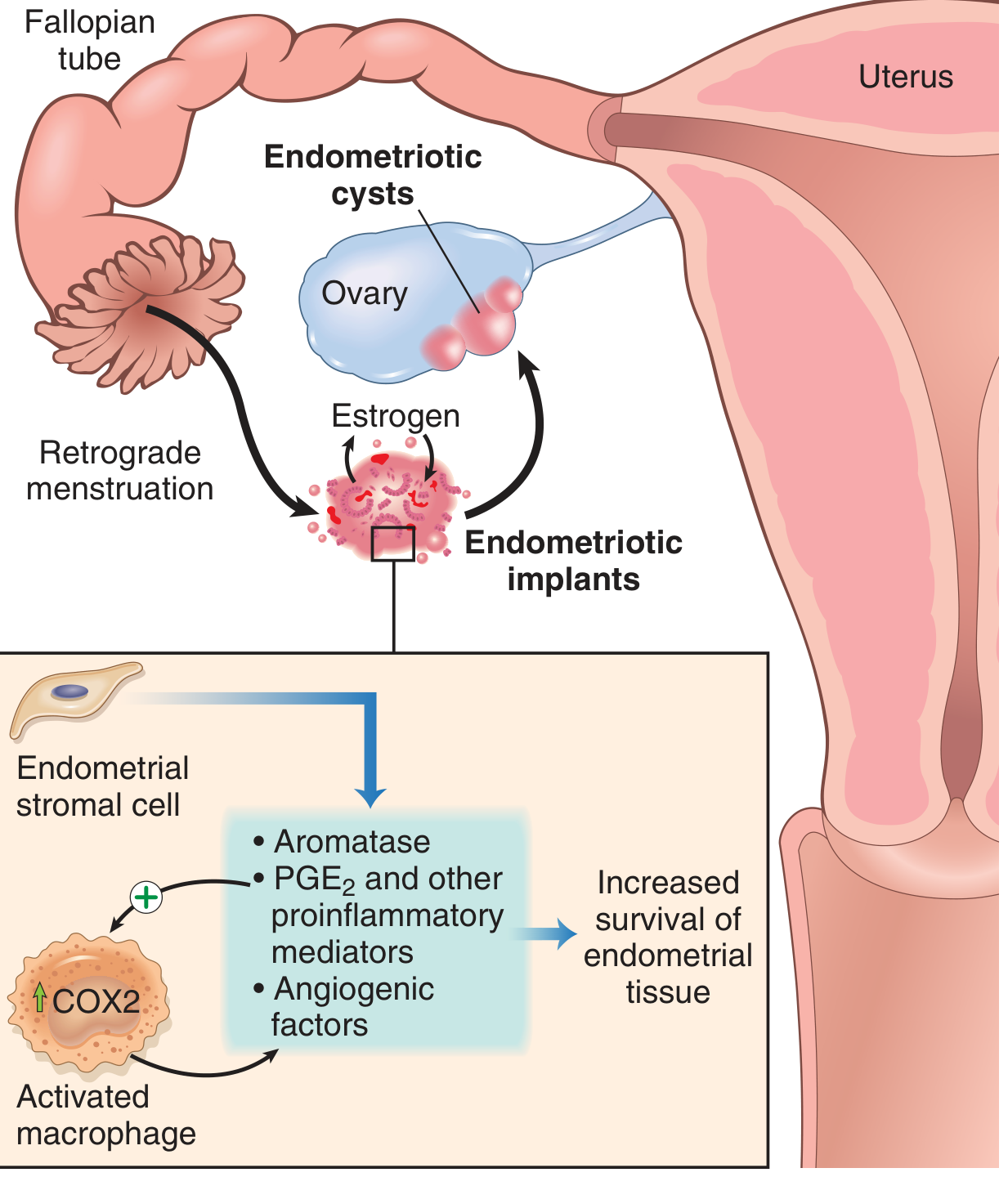
Three main theories are proposed — no single theory explains all cases:
-
Transplantation/Regurgitation theory (Sampson, 1920s) — most widely accepted. Retrograde menstruation through the fallopian tubes deposits endometrial cells onto pelvic peritoneum. Retrograde menstruation occurs in 70–90% of women but only some develop endometriosis, implying additional host factors are required.
-
Coelomic metaplasia theory — Pelvic mesothelium (sharing embryonic origin with Müllerian ducts) undergoes metaplastic transformation into endometrial tissue.
-
Induction theory / Extrauterine stem cell theory — Bone marrow-derived stem/progenitor cells differentiate into endometrial tissue at ectopic sites.
Why implants persist: Endometriotic stromal cells upregulate:
- Aromatase → increased local oestrogen production
- PGE₂ and other proinflammatory mediators
- VEGF and angiogenic factors
- MMPs (released by recruited macrophages)
This creates a self-sustaining pro-inflammatory, pro-oestrogenic microenvironment that promotes implant survival. Endometriosis is fundamentally an oestrogen-dependent disease.
Epidemiology & Risk Factors
- Prevalence: ~10% reproductive-age women; up to 50% in women with unexplained infertility
- Economic burden comparable to diabetes mellitus, Crohn's disease, and rheumatoid arthritis (WERF EndoCost study)
Risk factors: Early menarche, short menstrual cycles, heavy flow, nulliparity, Müllerian anomalies, outflow obstruction, positive family history, DES exposure
Protective factors: Multiparity, prolonged lactation, late menarche, oral contraceptive use
Common Sites
Endometriosis follows peritoneal fluid currents and predominantly affects the left hemipelvis:
- Ovaries (most common)
- Pouch of Douglas / posterior cul-de-sac
- Uterosacral ligaments
- Posterior uterine surface
- Fallopian tubes, broad ligaments
- Rectosigmoid colon (most common extrapelvic site)
Extrapelvic sites (rare): Diaphragm (right side), pleura, lung (catamenial pneumothorax/haemoptysis), bladder/ureter, umbilicus, laparotomy scars
Morphology (Pathology)
- Macroscopically: red-brown nodules (active) or white fibrotic plaques (inactive); range from microscopic to 1–2 cm
- Ovarian endometriomas: cysts 3–5 cm, filled with dark "chocolate fluid" (degenerated blood/haemosiderin)
- Chronic bleeding → fibrous adhesions obliterating the pouch of Douglas and distorting anatomy
- Histology (diagnostic standard): endometrial glands + stroma at ectopic site
Clinical Features
| Symptom | Notes |
|---|---|
| Dysmenorrhea (cyclical pelvic pain) | Most common; progressively worsening |
| Dyspareunia (deep) | Uterosacral/cul-de-sac involvement |
| Chronic pelvic pain | Non-menstrual |
| Infertility | 30–40% of patients; presenting complaint |
| Dyschezia | Rectosigmoid involvement |
| Dysuria / haematuria | Bladder/ureteral involvement |
| Catamenial pneumothorax / haemoptysis | Thoracic endometriosis |
| Cyclic shoulder pain | Diaphragmatic endometriosis |
Examination: Uterosacral nodularity, fixed retroverted uterus, adnexal mass (endometrioma), cervical lateral displacement, painful rectovaginal septum
Diagnosis
Clinical
- Empirical medical treatment (e.g., hormonal therapy) may precede formal laparoscopic confirmation in patients with classic symptoms and no suspicion of malignancy (ESHRE guideline)
Imaging
- Transvaginal ultrasound (TVS): First-line; excellent for ovarian endometriomas (ground-glass echogenicity, homogeneous). Limited for peritoneal disease.
- MRI: Best for deep infiltrating endometriosis — bowel, bladder, and rectovaginal septum involvement
Laparoscopy
- Gold standard for diagnosis. Allows visual inspection of entire pelvis and histologic confirmation.
- Positive histology confirms; negative histology does not exclude the diagnosis.
- Histologic confirmation recommended for endometriomas >4 cm and deep disease (to exclude malignancy)
Biomarkers
- CA-125 is elevated in some women but has poor sensitivity/specificity — not recommended for routine diagnosis
- Elevated CRP, IL-6, TNF-α in peripheral blood (research tools; not yet standard)
Classification / Staging
rASRM (American Society for Reproductive Medicine)
Staged I–IV (Minimal → Severe) based on:
- Size, appearance, depth of peritoneal/ovarian implants
- Adnexal adhesions
- Cul-de-sac obliteration
Lesion colour classification: Red (active), White (fibrotic), Black (old haemorrhagic)
Limitation: Correlates poorly with pain severity or fertility outcomes.
Endometriosis Fertility Index (EFI)
- Predicts non-IVF pregnancy rates after surgical staging
- Recommended when future fertility is a concern
ENZIAN Score
- Supplements rASRM for deep endometriosis morphology
- Useful for surgical planning
Medical Management
The goal is suppression of ovarian oestrogen production and/or progesterone opposition:
| Drug Class | Examples | Notes |
|---|---|---|
| NSAIDs / COX-2 inhibitors | Ibuprofen, celecoxib | First-line for pain |
| Combined oral contraceptives (COCs) | Continuous or cyclic | Equally effective for pain; low cost |
| Progestins | Norethisterone, medroxyprogesterone, dienogest, LNG-IUS | First-line; dienogest has strong evidence |
| GnRH agonists | Leuprolide, nafarelin, goserelin | Highly effective; add-back therapy needed for bone protection |
| GnRH antagonists | Elagolix | Oral; dose-dependent oestrogen suppression |
| Aromatase inhibitors | Letrozole, anastrozole | Used in refractory/postmenopausal disease |
| Danazol | (Androgenic steroid) | Rarely used now due to side effects |
Key principle: Hormonal drug classes are equally effective for pain relief; selection is guided by side-effect profile, cost, and contraceptive needs. Suppression of ovarian function does NOT improve fertility — medical therapy is not used for infertility management.
Surgical Management
Peritoneal endometriosis
- Ablation or excision equally effective
- Excision preferred when histologic confirmation is desired
- Ablation + adhesiolysis in minimal/mild disease improves fertility vs diagnostic laparoscopy alone
Ovarian endometrioma
- Cystectomy (excision) preferred over drainage + ablation — lower recurrence of pain, endometrioma, and need for reoperation; higher spontaneous pregnancy rates (ESHRE Grade A)
- Intraovarian suture for haemostasis is superior to bipolar coagulation in reducing post-surgical adhesions
Deep infiltrating endometriosis
- Complete surgical excision in a single-stage procedure (multidisciplinary team — gynaecologist, colorectal surgeon, urologist)
- Referral to specialist centre strongly recommended
Definitive surgery
- Total hysterectomy ± bilateral salpingo-oophorectomy (BSO) for women who have completed their family
- BSO reduces recurrence but is not curative in all cases
Endometriosis and Fertility
| Stage | Effect on Fertility |
|---|---|
| Minimal/Mild (I–II) | Controversial; possible subtle effects on peritoneal environment |
| Moderate/Severe (III–IV) | Clear mechanical effect — blocked tubo-ovarian motility, impaired ovum pickup |
- Spontaneous abortion rate: Not clearly increased; evidence contradictory
- Assisted reproduction (IVF): Recommended when other treatments fail; ovarian reserve may be reduced by disease and prior surgery
Malignant Transformation
- Risk mainly confined to deep infiltrating endometriosis and ovarian endometriomas
- Associated malignancies: endometrioid carcinoma and clear cell carcinoma of the ovary
- Lifetime risk ~1% in endometrioma patients
Key Takeaways (ESHRE Guidelines)
- Empirical hormonal therapy is acceptable without surgical diagnosis in classic presentations
- Laparoscopy is the gold standard for definitive diagnosis
- Hormonal drug classes are equally effective for pain — choice depends on side effects and patient preference
- Medical therapy does not improve fertility — surgical removal of lesions does (in minimal/mild disease)
- Cystectomy is preferred over drainage for endometriomas ≥3 cm
- Deep DIE requires specialist multidisciplinary surgical management
Sources: Berek & Novak's Gynecology (Berek & Novak's Gynecology, pp. 617–660) | Robbins & Kumar Basic Pathology, p. 689
D c dutta can you use?
Finding Sources
Finding Sources
Unfortunately, DC Dutta's Textbook of Gynaecology is not available in the library. The gynaecology/obstetrics titles currently in the collection are:
- Berek & Novak's Gynecology — the standard international reference I used in the previous answer
- Creasy & Resnik's Maternal-Fetal Medicine — focused on obstetrics/high-risk pregnancy
DC Dutta is a very popular South Asian textbook (widely used in India, Bangladesh, Sri Lanka, etc.), but it's not licensed in this library.
For endometriosis topics, Berek & Novak covers the same content in considerable depth — it's essentially the Western equivalent and aligns well with ESHRE and ASRM guidelines. If you have a specific chapter or topic from DC Dutta you'd like me to address, I can pull the relevant content from Berek & Novak and map it to what Dutta would typically cover.
This is a shared conversation. Sign in to Orris to start your own chat.