Draw and describe functional unit of kidney
Reading File
Finding Sources
Searching Images
nephron functional unit kidney diagram anatomy
Generating Image
Loading Image
Reading File
Reading File
Reading File
Loading Image
The Nephron — Functional Unit of the Kidney
Diagrams
Generated Diagram:
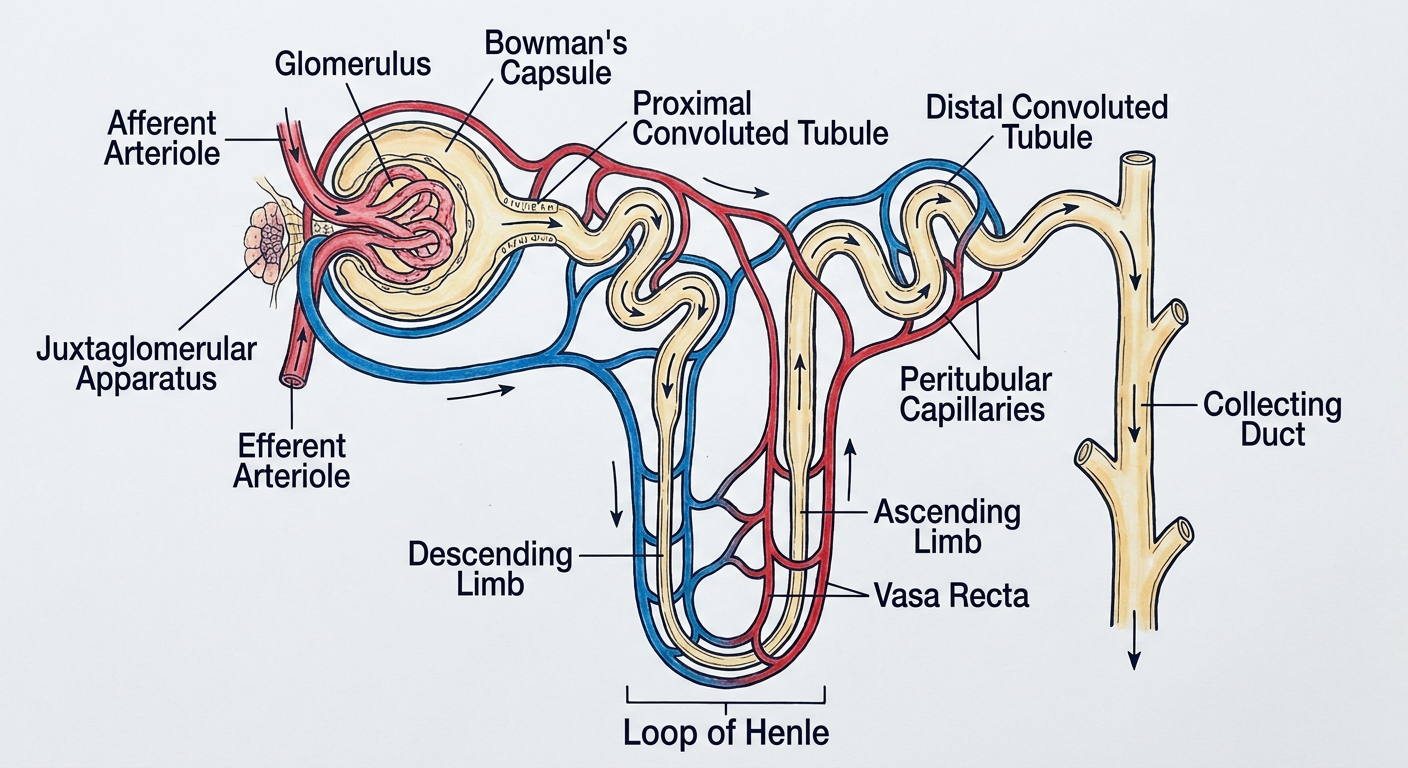

From Harrison's Principles of Internal Medicine:
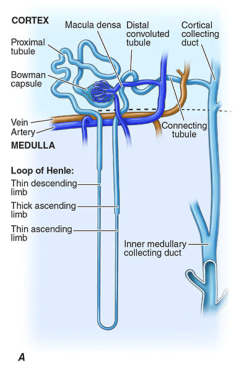

Definition
The nephron is the structural and functional unit of the kidney. Each human kidney contains approximately 900,000 to 1 million nephrons (ranging from 200,000 to >2.5 million per kidney). It consists of two major components:
- Renal corpuscle (Malpighian corpuscle) — glomerulus + Bowman's capsule
- Renal tubule — proximal tubule → loop of Henle → distal tubule → connecting segment
The collecting duct is functionally inseparable but technically not part of the nephron, as it derives embryologically from the ureteric bud rather than the metanephric blastema. — Brenner and Rector's The Kidney
Components and Functions
1. Renal Corpuscle
| Structure | Description |
|---|---|
| Glomerulus | Tuft of fenestrated capillaries; site of ultrafiltration (~180 L/day filtered) |
| Bowman's capsule | Double-walled cup surrounding the glomerulus; collects the filtrate |
| Afferent arteriole | Brings blood into the glomerulus |
| Efferent arteriole | Carries blood out; maintains glomerular filtration pressure |
| Juxtaglomerular apparatus | Macula densa + JG cells; regulates renin secretion and tubuloglomerular feedback |
2. Proximal Convoluted Tubule (PCT)
- Location: Renal cortex
- Function: Reabsorbs >60% of the filtrate — the "workhorse" of the kidney
- Key transporters: Na⁺/H⁺ exchanger (NHE3), Na⁺/glucose cotransporters, amino acid transporters, aquaporin-1
- Water follows isosmotically (AQP1); bicarbonate is reclaimed; NH₄⁺ is secreted
- Luminal brush border (microvilli) increases surface area ~40-fold
3. Loop of Henle
- Location: Descends from cortex into medulla, then returns
- Segments:
- Thin descending limb — permeable to water; concentrates filtrate
- Thin ascending limb — impermeable to water; permeable to solutes
- Thick ascending limb (TAL) — impermeable to water; active transport via NKCC2 (Na⁺-K⁺-2Cl⁻ cotransporter)
- Function: Creates the medullary concentration gradient via countercurrent multiplication — the concentrating segment
- ROMK channel recycles K⁺ into the lumen; NH₄⁺ can substitute for K⁺ on NKCC2
- The medullary interstitium is the only compartment in the body with higher osmolality than serum
4. Distal Convoluted Tubule (DCT)
- Location: Renal cortex (~10–12 mm long, shortest nephron segment)
- Key transporter: NCC (Na⁺-Cl⁻ cotransporter, target of thiazide diuretics)
- Impermeable to water → removes solute without water → diluting segment
- Contains TRPV5 (Ca²⁺ channel on luminal membrane) for calcium reabsorption regulated by PTH
5. Collecting Duct
- Two cell populations:
- Principal cells: Vasopressin-stimulated AQP2 insertion → water reabsorption; aldosterone-stimulated ENaC → Na⁺ reabsorption + K⁺ secretion via ROMK
- α-Intercalated cells: H⁺-ATPase on luminal side → H⁺ secretion (acid-base regulation)
- β-Intercalated cells: H⁺-ATPase on basolateral side + pendrin (Cl⁻/HCO₃⁻ exchanger) → HCO₃⁻ secretion in alkalosis
- Sets final urine composition
6. Vasa Recta
- Straight capillaries running parallel to the loop of Henle
- Preserve the medullary osmotic gradient via countercurrent exchange
- Supply O₂ and nutrients to the medulla
Summary Table
| Segment | Location | Key Function | Water Permeability |
|---|---|---|---|
| Glomerulus | Cortex | Filtration | N/A |
| PCT | Cortex | Bulk reabsorption (>60%) | High (AQP1) |
| Thin descending limb | Medulla | Water reabsorption | High |
| Thin ascending limb | Medulla | Passive solute exit | Low |
| Thick ascending limb | Medulla/cortex | Active NaCl reabsorption | None |
| DCT | Cortex | Dilution, Ca²⁺/Na⁺ fine-tuning | Low |
| Collecting duct | Cortex→medulla | Final concentration (ADH-dependent) | Variable (AQP2) |
Sources: Brenner and Rector's The Kidney; National Kidney Foundation Primer on Kidney Diseases, 8th ed.; Harrison's Principles of Internal Medicine, 21st ed.
countercurrent Mechanism
Finding Sources
Searching Images
countercurrent multiplication mechanism loop of Henle kidney urine concentration diagram
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Loading Image
Here is a comprehensive explanation of the countercurrent mechanism in the kidney.
Countercurrent Mechanism
The kidney uses two distinct but interrelated countercurrent systems to concentrate urine and create the hyperosmotic medullary interstitium:
- Countercurrent Multiplier — Loop of Henle
- Countercurrent Exchanger — Vasa Recta
What Is a Countercurrent System?
A countercurrent system is one in which inflow runs parallel to, counter to, and in close proximity to outflow for some distance. In the kidney, filtrate in the descending limb flows down toward the medulla while filtrate in the ascending limb flows up toward the cortex — in opposite directions, right next to each other. This spatial arrangement is what allows the gradient to be amplified ("multiplied"). — Ganong's Review of Medical Physiology
Part 1: Countercurrent Multiplier (Loop of Henle)
Key Structural Prerequisites
- ~25% of nephrons are juxtamedullary nephrons with long loops that dip deep into the medulla (some reaching the papillary tips)
- Thin descending limb (TDL): highly permeable to water (AQP1); relatively impermeable to solutes
- Thick ascending limb (TAL): actively pumps NaCl out via NKCC2; impermeable to water
- Filtrate flows in opposite directions in the two limbs
Step-by-Step Operation (Hypothetical Steps — Ganong's):
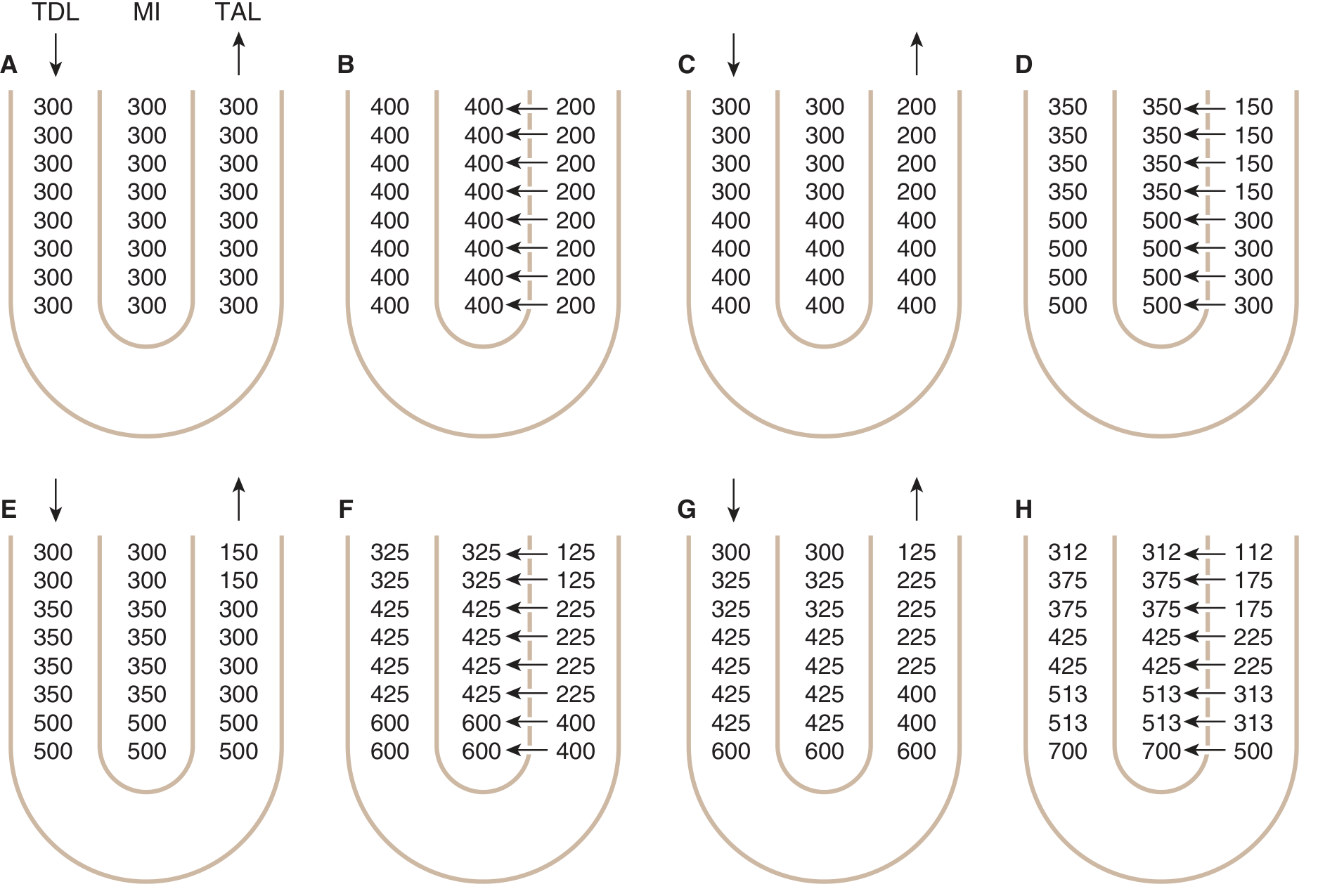
| Step | Event |
|---|---|
| A | All compartments start at 300 mOsm/L |
| B | TAL pumps Na⁺/Cl⁻ into interstitium → interstitium rises to 400 mOsm/L; TAL lumen drops to 200 mOsm/L |
| C | Isotonic fluid (300 mOsm/L) continuously enters from proximal tubule; the TDL equilibrates with the hypertonic interstitium — water exits the TDL |
| D | Continued pumping: fluid at the apex of the loop becomes increasingly concentrated; fluid leaving the TAL becomes increasingly dilute |
| Final state | A corticomedullary osmotic gradient is established — ~300 mOsm/L at the cortex, rising to 1200–1400 mOsm/L at the papillary tip |
"The pump in the TAL is capable of establishing about a 200 mOsm/L concentration gradient between the tubular lumen and interstitial fluid. Because the TAL is impermeable to water, solutes pumped out are not followed by osmotic flow of water — solutes accumulate in excess of water." — Guyton & Hall Medical Physiology
Role of Juxtamedullary Nephrons
- Long loops of Henle spread the gradient over a greater distance → higher osmolality at the tip
- The thin ascending limb (present only in juxtamedullary nephrons) is impermeable to water but permeable to NaCl → passive countercurrent multiplication adds to the gradient in the inner medulla
Part 2: Role of Urea
Urea contributes 40–50% of the inner medullary osmolality (~500–600 mOsm/L of the 1200 total):
- Urea is not reabsorbed in the proximal tubule, loop of Henle, or early collecting duct
- As ADH drives water reabsorption in the cortical collecting duct, urea concentrates in the lumen
- In the inner medullary collecting duct, vasopressin activates UT-A1 and UT-A3 urea transporters → urea diffuses into the medullary interstitium
- Some urea re-enters the loop of Henle via UT-A2 in the thin descending limb → urea recycling
- This urea recycling amplifies the medullary gradient without requiring additional energy
A high-protein diet increases urea production → greater medullary urea → better concentrating ability. A low-protein diet reduces it. — Ganong's
Part 3: Countercurrent Exchanger (Vasa Recta)
The Renal Concentrating Mechanism — Transport proteins:
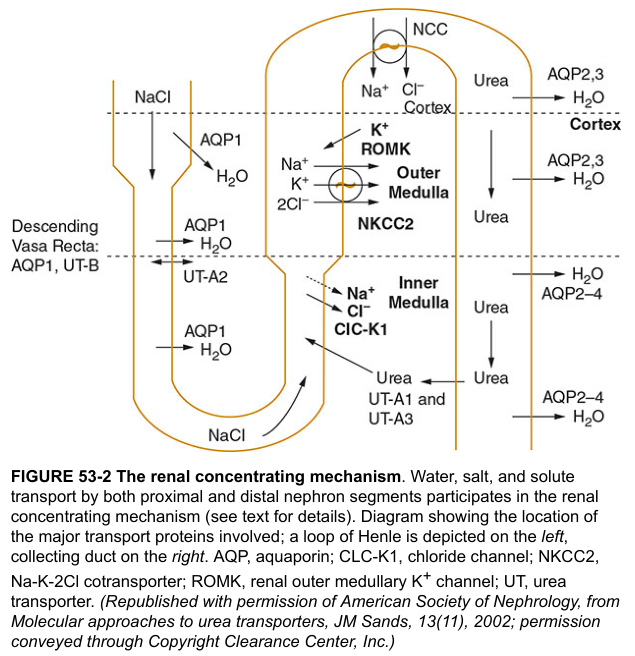
The vasa recta are straight peritubular capillaries that loop into the medulla alongside the loop of Henle. Their role is to supply the medulla with nutrients and oxygen without washing out the gradient.
| Vessel | What Happens |
|---|---|
| Descending vasa recta | Blood enters the medulla; water exits (AQP1) and solutes (NaCl, urea via UT-B) enter → blood becomes progressively hyperosmotic |
| Ascending vasa recta | Blood returns toward cortex; water re-enters and solutes exit → blood osmolality falls back toward normal |
Because NaCl and urea diffuse out of the ascending limb and back into the descending limb, the solutes are recycled within the medulla rather than being carried away. This preserves the gradient that the loop of Henle worked to build.
Part 4: ADH and Final Urine Concentration
The gradient is built by the loop of Henle but is only used when ADH (vasopressin) is present:
| ADH Level | Collecting Duct | Result |
|---|---|---|
| High | AQP2 inserted into luminal membrane → water reabsorbed → urine equilibrates with medullary interstitium | Concentrated urine: up to 1200 mOsm/L |
| Low/absent | Collecting duct impermeable to water | Dilute urine: as low as 50–100 mOsm/L |
Tubular Osmolality Journey (Summary)
Glomerular filtrate: ~290 mOsm/L (isosmotic with plasma)
End of PCT: ~290 mOsm/L (isosmotic reabsorption)
Bottom of loop (TDL): ~1200 mOsm/L (water exits → concentrated)
End of TAL: ~100–150 mOsm/L (NaCl pumped out → dilute)
End of DCT: ~50–100 mOsm/L (diluting segment)
Final urine (with ADH): up to 1200 mOsm/L
Final urine (no ADH): ~50–100 mOsm/L
Key Transporters Summary
| Transporter | Location | Role |
|---|---|---|
| AQP1 | TDL, descending vasa recta | Water reabsorption |
| NKCC2 | TAL | Active NaCl reabsorption (no water) |
| ROMK | TAL | K⁺ recycling into lumen |
| CLC-K1 | Thin ascending limb | Cl⁻ exit (passive) |
| UT-A1/A3 | Inner medullary CD | Urea into interstitium (ADH-regulated) |
| UT-A2 | Thin descending limb | Urea recycling |
| UT-B | Descending vasa recta | Urea trapping |
| AQP2 | Collecting duct (apical) | Water reabsorption (ADH-regulated) |
| AQP3/4 | Collecting duct (basolateral) | Water exit to interstitium |
Sources: Ganong's Review of Medical Physiology, 26th ed.; Guyton & Hall Textbook of Medical Physiology; National Kidney Foundation Primer on Kidney Diseases, 8th ed.; Harrison's Principles of Internal Medicine, 21st ed.
Illustrate counter current mechanisms for urine acidification
Finding Sources
Finding Sources
Searching Images
urine acidification renal tubular H+ secretion bicarbonate reabsorption collecting duct intercalated cells diagram
Finding Sources
Finding Sources
Loading Image
Loading Image
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Loading Image
Generating Image
Countercurrent Mechanism for Urine Acidification
Important clarification: The term "countercurrent mechanism for urine acidification" refers specifically to NH₃/NH₄⁺ countercurrent cycling — the medullary ammonia gradient that enables the kidney to excrete large amounts of acid as ammonium (NH₄⁺) in the final urine. This is distinct from the urine concentration countercurrent mechanism, but uses the same anatomical architecture.
Overview Diagram
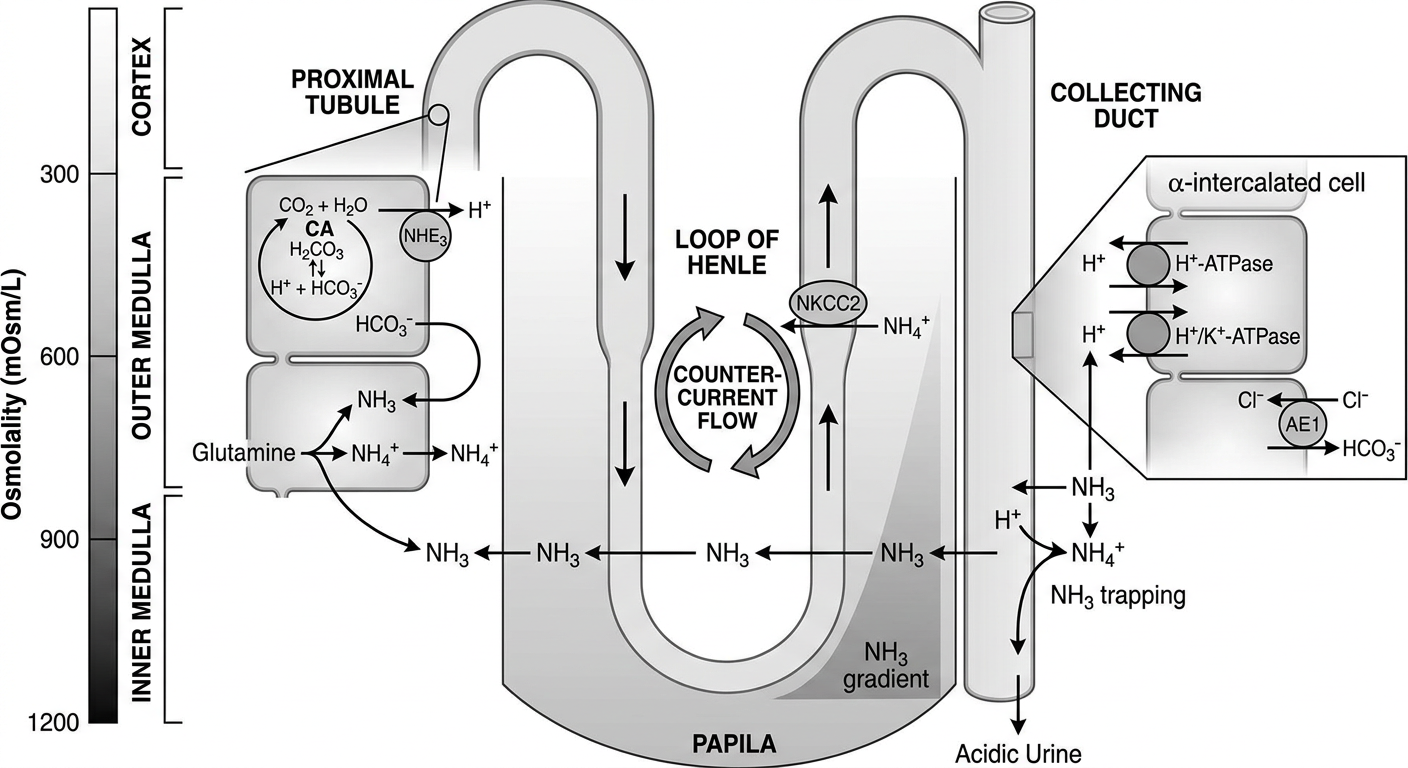
The Three Pillars of Renal Acid Excretion
The kidney excretes acid via three coordinated processes:
| Process | Where | Amount |
|---|---|---|
| HCO₃⁻ reabsorption | Proximal tubule (~85%), TAL, DCT | Reclaims filtered buffer |
| Titratable acid excretion (H₂PO₄⁻) | Distal nephron | ~30–40 mEq/day |
| NH₄⁺ excretion (ammoniagenesis) | PCT synthesis + medullary cycling | ~40–60 mEq/day (dominant) |
Part 1: Proximal Tubule — Bicarbonate Reclamation & NH₄⁺ Generation
HCO₃⁻ Reabsorption
Lumen: H⁺ + HCO₃⁻ → H₂CO₃ → H₂O + CO₂ (luminal CA IV)
Cell: CO₂ + H₂O → H₂CO₃ → H⁺ + HCO₃⁻ (cytosolic CA II)
H⁺ secreted via NHE3 (Na⁺/H⁺ exchanger) into lumen
HCO₃⁻ exits basolaterally via NBC1 (Na⁺/3HCO₃⁻ cotransporter)
Ammoniagenesis from Glutamine
Glutamine → 2NH₄⁺ + 2HCO₃⁻
(in PCT mitochondria)
NH₄⁺ secreted into lumen via NHE3 (substituting for H⁺)
HCO₃⁻ returns to blood → "new bicarbonate" is generated
- This is the most important source of net acid excretion
- Stimulated by acidosis, hypokalaemia, and cortisol
Part 2: The NH₃/NH₄⁺ Countercurrent Cycling — The True "Countercurrent" for Acidification
This is the heart of the mechanism. The medulla acts as an NH₃ trap, concentrating ammonia toward the papilla to allow massive NH₄⁺ excretion in collecting duct urine.
Step-by-Step Cycling:
CORTEX
PCT: Glutamine → NH₄⁺ secreted into lumen via NHE3
↓ (filtrate flows down)
OUTER MEDULLA
Thick Ascending Limb (TAL):
NH₄⁺ reabsorbed via NKCC2 (NH₄⁺ substitutes for K⁺)
→ NH₄⁺ enters medullary interstitium
→ dissociates: NH₄⁺ ⇌ NH₃ + H⁺
→ NH₃ accumulates in medullary interstitium
↓
INNER MEDULLA
NH₃ diffuses from interstitium into collecting duct lumen
In the acidic lumen: NH₃ + H⁺ → NH₄⁺ (TRAPPED — cannot back-diffuse)
NH₄⁺ excreted in urine
Why "Countercurrent"?
- NH₄⁺ flows down in the tubular lumen (descending)
- TAL reabsorbs it and deposits it in the interstitium
- This creates an increasing NH₃ gradient from cortex → papilla (just like NaCl in urine concentration)
- The collecting duct descends through this NH₃-rich interstitium, "picking up" NH₃ which becomes trapped as NH₄⁺ by the H⁺ secreted by intercalated cells
"NH₄⁺ can substitute for K⁺ on the NKCC2 in the TAL. NH₄⁺ is absorbed by NKCC2 in the K⁺ position. Once in the cell, NH₄⁺ dissociates to NH₃ because of the higher intracellular pH. NH₃ diffuses into the interstitium through the Rhesus glycoprotein channel (RhCG)." — National Kidney Foundation Primer on Kidney Diseases, 8th ed.
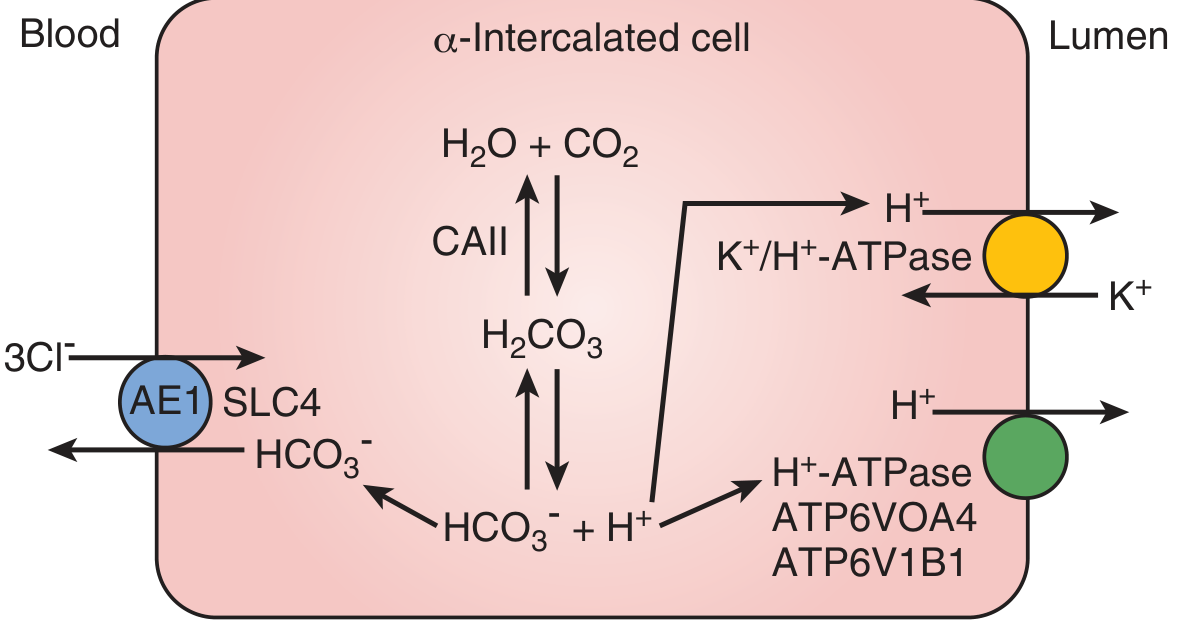
Part 3: Collecting Duct — Final Urine Acidification
α-Intercalated Cells (Type A)
| Membrane | Transporter | Action |
|---|---|---|
| Apical (lumen) | H⁺-ATPase (V-type; ATP6V0A4/ATP6V1B1) | Pumps H⁺ into lumen → acidifies urine |
| Apical (lumen) | H⁺/K⁺-ATPase | Secretes H⁺, reabsorbs K⁺ |
| Basolateral | AE1/SLC4A1 (Cl⁻/HCO₃⁻ exchanger) | HCO₃⁻ exits to blood (new bicarbonate) |
| Cytoplasm | Carbonic anhydrase II | CO₂ + H₂O → H₂CO₃ → H⁺ + HCO₃⁻ |
In the α-intercalated cell:
CO₂ + H₂O →[CA II]→ H₂CO₃ → H⁺ + HCO₃⁻
H⁺ → secreted into lumen (H⁺-ATPase / H⁺K⁺ATPase)
HCO₃⁻ → exits to blood via AE1
The lumen pH can fall as low as 4.5 (minimum urine pH achievable).
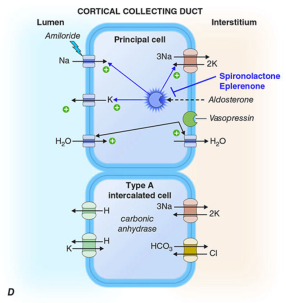
β-Intercalated Cells (Type B)
- Mirror image of α cells
- H⁺-ATPase on the basolateral side
- Pendrin (SLC26A4) on apical side secretes HCO₃⁻ into lumen
- Active during alkalosis to excrete excess base
Harrison's diagram of B-IC and PC cells:
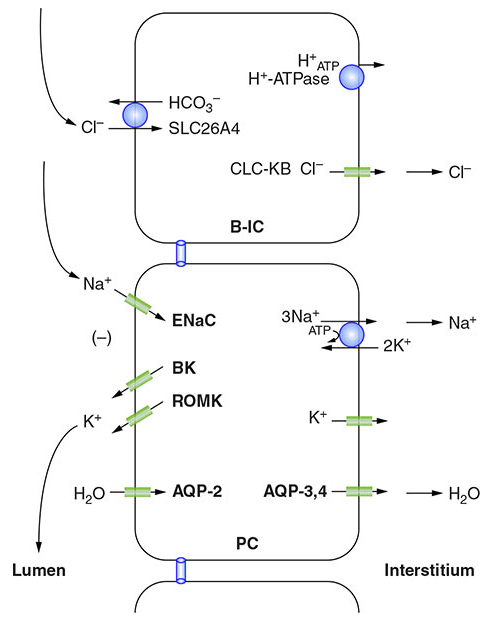

Part 4: Titratable Acid — Phosphate Buffering
In the lumen, secreted H⁺ combines with filtered buffers:
H⁺ + HPO₄²⁻ → H₂PO₄⁻ (titratable acid, pKa 6.8)
H⁺ + NH₃ → NH₄⁺ (ammonia buffering, pKa 9.0)
- NH₃ is the dominant buffer because its pKa (9.0) is far above urine pH → nearly complete trapping
- Phosphate contributes ~30–40 mEq/day; NH₄⁺ contributes ~40–60 mEq/day
Integrated Summary: The Full Countercurrent Picture
CORTEX (pH ~7.4)
┌─────────────────────────────────────────────────────┐
│ PCT: Glutamine → NH₄⁺ (lumen) + HCO₃⁻ (blood) │
│ NHE3: H⁺ secreted → HCO₃⁻ reclaimed │
└──────────────────┬──────────────────────────────────┘
↓ filtrate descends
OUTER MEDULLA
┌─────────────────────────────────────────────────────┐
│ TAL: NKCC2 absorbs NH₄⁺ from lumen │
│ → NH₃ builds up in interstitium │
│ → Interstitial [NH₃] increases toward papilla │
└──────────────────┬──────────────────────────────────┘
↓
INNER MEDULLA (highest [NH₃])
┌─────────────────────────────────────────────────────┐
│ Collecting duct descends through NH₃-rich milieu │
│ α-IC cells: H⁺-ATPase → H⁺ into lumen (pH → 4.5) │
│ NH₃ (interstitium) → lumen + H⁺ → NH₄⁺ (TRAPPED) │
│ AE1: HCO₃⁻ → blood ("new bicarbonate") │
└─────────────────────────────────────────────────────┘
↓
URINE: NH₄⁺ + H₂PO₄⁻ + pH 4.5–6.0
Clinical Relevance: Renal Tubular Acidosis (RTA)
| RTA Type | Defect | Urine pH | NH₄⁺ Excretion |
|---|---|---|---|
| Type 1 (Distal) | H⁺-ATPase or AE1 mutation → α-IC cell fails | >5.5 (cannot acidify) | Low |
| Type 2 (Proximal) | NHE3 / CA II defect → HCO₃⁻ wasting | <5.5 at steady state | Low |
| Type 4 | Hypoaldosteronism → reduced NH₄⁺ synthesis | <5.5 | Very low |
Defects in ATP6V0A4 or ATP6V1B1 (H⁺-ATPase subunits) or SLC4A1 (AE1) cause distal RTA by disrupting the final acidification step. — Campbell-Walsh-Wein Urology
Sources: National Kidney Foundation Primer on Kidney Diseases, 8th ed.; Brenner and Rector's The Kidney; Harrison's Principles of Internal Medicine, 21st ed.; Campbell-Walsh-Wein Urology
This is a shared conversation. Sign in to Orris to start your own chat.