Aortic regurgitation
Reading File
Finding Sources
Finding Sources
Searching PubMed
"aortic regurgitation"[MeSH Terms] AND management
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
aortic regurgitation echocardiography color Doppler
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive information from multiple authoritative textbooks. Here is a complete clinical overview of aortic regurgitation.
Aortic Regurgitation (AR)
Definition
Aortic regurgitation (also called aortic insufficiency, AI) is diastolic backflow of blood from the aorta into the left ventricle (LV) through an incompetent aortic valve. It can be acute or chronic, and the two present in fundamentally different ways.
Etiology
AR arises from either leaflet pathology or aortic root dilatation (causing malcoaptation of otherwise normal leaflets):
| Leaflet Causes | Aortic Root Causes |
|---|---|
| Bicuspid aortic valve (most common congenital) | Marfan syndrome / connective tissue disease |
| Infective endocarditis | Hypertension-induced annuloaortic ectasia |
| Rheumatic heart disease | Aortic dissection |
| Dopamine/serotonin receptor agonists | Syphilitic aortitis |
| — | Ankylosing spondylitis / psoriatic arthritis |
Acute AR is most often caused by infective endocarditis or aortic dissection.
— Goldman-Cecil Medicine, p. 694
Pathophysiology
Chronic AR — Volume Overload + Afterload Excess
AR imposes volume overload on the LV: the ventricle must pump both the forward stroke volume from the LA and the regurgitant volume returning from the aorta. Compensation occurs through eccentric hypertrophy (LV dilatation + wall thickening), allowing the chamber to accommodate the extra volume at a lower filling pressure and maintain a large total stroke volume.
Because the increased stroke volume raises systolic pressure, there is also an element of afterload excess — producing a combined eccentric + concentric hypertrophy pattern unique to AR (unlike mitral regurgitation, which is pure volume overload without significant afterload increase).
The widened pulse pressure (high systolic, low diastolic) reflects both the elevated stroke volume and the diastolic runoff back into the LV. The low aortic diastolic pressure reduces coronary perfusion pressure, and the elevated LV diastolic pressure compresses subendocardial vessels — together these cause coronary ischemia even without obstructive CAD.
— Guyton and Hall Medical Physiology, p. 293; Goldman-Cecil Medicine, p. 694
Acute AR — Unprepared Ventricle
In acute AR, the previously normal (non-dilated, non-hypertrophied) LV is suddenly overwhelmed by the regurgitant volume. The result is a precipitous fall in forward output with a sharp rise in LV filling pressure → pulmonary oedema and haemodynamic collapse. Reflex vasoconstriction increases peripheral vascular resistance, worsening the situation. The reduced coronary perfusion gradient accelerates ischaemia and deterioration.
The mitral valve preclosure sign — high diastolic LV pressure closing the mitral valve before systole — is an especially ominous echocardiographic finding in acute AR.
Clinical Features
Chronic AR
Symptoms typically appear after years of compensated disease:
- Exertional dyspnoea, orthopnoea, fatigue (left heart failure)
- Angina (less common than in AS; can occur with flushing)
- Awareness of heartbeat; carotid artery pain
Signs — the hyperdynamic large-stroke-volume state generates an array of classic eponymous findings:
| Sign | Description |
|---|---|
| Corrigan's (water-hammer) pulse | Sharp upstroke, rapid collapse of carotid pulse |
| de Musset sign | Head bobbing with each heartbeat |
| Duroziez sign | Systolic + diastolic bruits over compressed femoral artery |
| Quincke's pulse | Nail-bed pulsations (systolic plethora, diastolic blanching) |
| Hill sign | Femoral systolic BP ≥40 mmHg above brachial — most reliable sign of severity |
| Austin Flint murmur | Mid-diastolic rumble at apex; regurgitant jet causes mitral leaflet vibration |
Auscultation:
- High-pitched, blowing early diastolic murmur at lower left sternal border (valvular disease) or upper right sternal border (aortic root disease)
- Heard best with patient sitting forward, in full expiration
- Progressively longer murmur as severity worsens (pansystolic in severe AR)
- Apex: hyperactive, displaced downward and to the left
Acute AR
Signs are far more subtle — the compensatory eccentric hypertrophy is absent. Key clues:
- Short early diastolic murmur
- Soft S1 (mitral valve preclosure mutes the mitral component)
- Pulmonary oedema / shock may dominate
Investigations
ECG
- Left ventricular hypertrophy (nonspecific but almost universal)
Chest X-Ray
- Cardiomegaly (LV enlargement)
- Uncoiling and dilatation of the aortic root
Echocardiography (primary imaging modality)
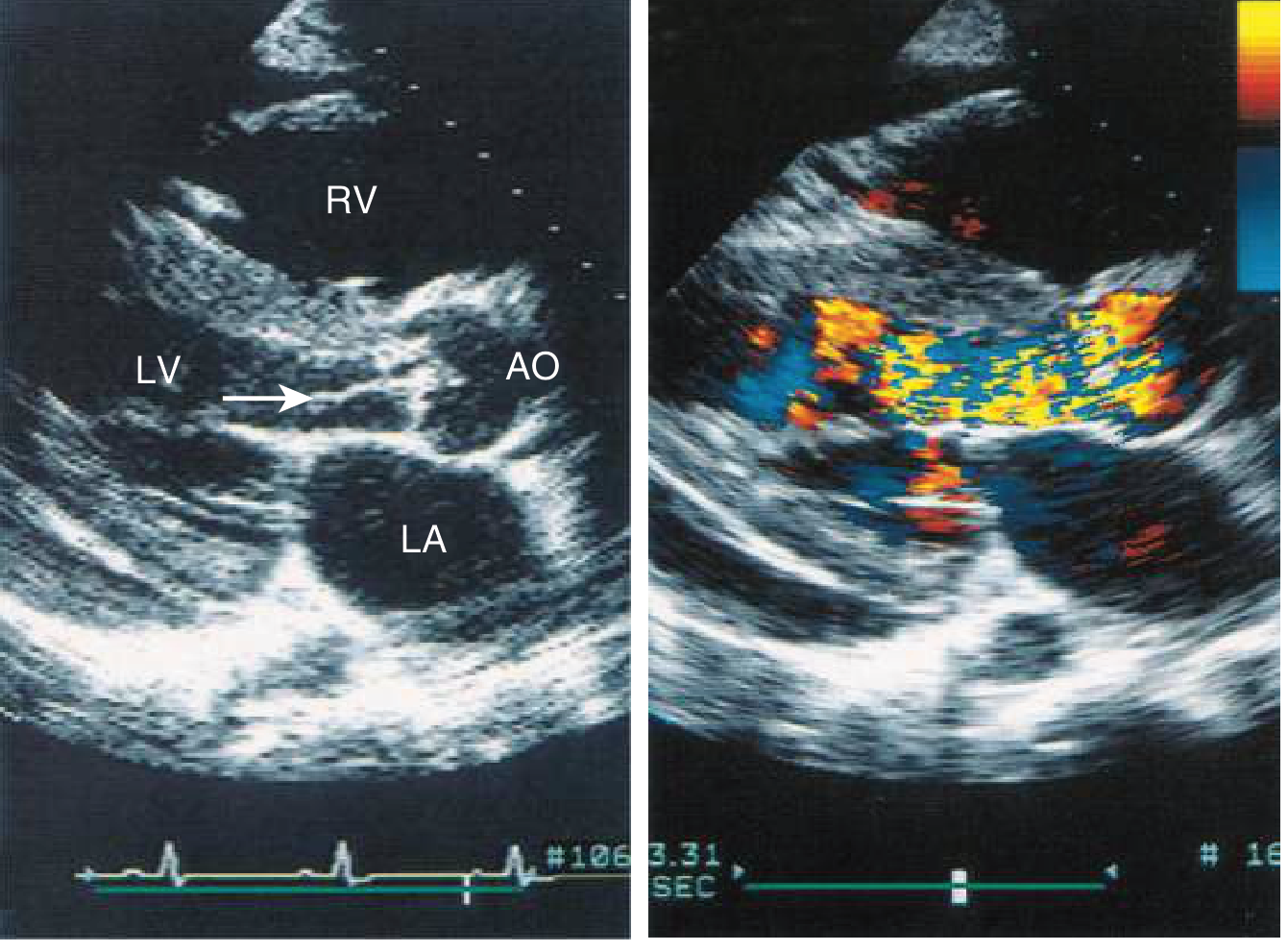
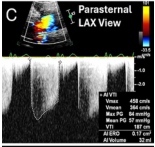
Key echo measurements:
- LV end-diastolic dimension (LVEDD) and end-systolic dimension (LVESD)
- LVEF — triggers surgery if <50%
- Vena contracta — most reliable single colour Doppler measure of severity (>6 mm = severe)
- Jet width:LVOT ratio — correlates with angiographic severity
- Pressure half-time (PHT) — short PHT (<200 ms) indicates severe AR
- Holodiastolic flow reversal in descending aorta — marker of severe AR
- Aortic root and ascending aorta measurements
Cardiac MRI (CMR)
Increasingly preferred for serial follow-up: more accurate for detecting small changes in LV volumes/function, and can quantify regurgitant fraction accurately in 3D. — Harrison's Principles of Internal Medicine 22E
Grading Severity (Echocardiographic Criteria — Severe AR)
| Parameter | Severe AR |
|---|---|
| Regurgitant volume | ≥60 mL/beat |
| Regurgitant fraction | ≥50% |
| Effective regurgitant orifice (ERO) | ≥0.30 cm² |
| Vena contracta | >0.6 cm |
| PHT | <200 ms |
| Holodiastolic aortic flow reversal | Present (descending aorta) |
Natural History
- Severe AR can remain asymptomatic for 10+ years in chronic disease
- Once significant symptoms develop: expected survival ~5 years without valve replacement
- Older patients develop heart failure symptoms and LV dysfunction earlier
- Left ventricular dilation, reduced EF, and moderate-or-greater pulmonary hypertension all predict higher mortality
- Surgery threshold indicators: LVESD >50 mm (or approaching 45 mm by some data), LVEDD >70 mm, LVEF <50%, calculated regurgitant orifice >30 mm²
— Braunwald's Heart Disease; Morgan & Mikhail's Clinical Anesthesiology
Management
Acute Severe AR — Surgical Emergency
- Urgent aortic valve replacement (AVR) is the definitive treatment
- Sodium nitroprusside (vasodilator) temporarily improves haemodynamics pre-operatively but is never a substitute for surgery
- Intra-aortic balloon pump is contraindicated (worsens regurgitation)
- Even in active infective endocarditis, surgery should not be withheld purely for a short duration of antibiotics — valve re-infection rate post-replacement is low (0–10%)
- Mitral valve preclosure on echo = especially urgent indication for surgery
Chronic Asymptomatic AR
- Vasodilators (nifedipine, ACE inhibitors): improve haemodynamics short-term but do not reliably delay surgery — not routinely recommended as a strategy to defer AVR
- Treat hypertension aggressively per standard guidelines
- Serial monitoring (echo every 1–2 years for severe AR; 3–5 years for mild–moderate)
- Surgery indicated even when asymptomatic if: LVEF <50%, LVESD >50 mm, or patient is undergoing other cardiac surgery
Chronic Symptomatic AR or AR with LV Dysfunction
- AVR as soon as feasible — medical therapy is only for short-term stabilisation
- Most patients require prosthetic valve; some are candidates for valve repair
- TAVR is emerging as an option for high-risk older patients (previously contraindicated for AR due to lack of calcification for anchoring, but newer dedicated devices show promise)
Surgical vs. Transcatheter Options
- Surgical AVR remains the gold standard for most patients
- Valve repair (Ross procedure in young patients; leaflet/root repair) when anatomy is suitable
- TAVR for AR: historically challenging due to absence of calcified annulus for anchoring; newer dedicated devices with promising early results — may become a reliable option in high-risk individuals
Special Situations
Pregnancy: Chronic AR is generally well tolerated in pregnancy as peripheral vasodilation reduces afterload. Acute AR is poorly tolerated and carries high maternal and fetal risk.
Older adults: More likely to develop HF symptoms and LV dysfunction earlier; operative mortality rises steeply as LVEF falls (from <5% with normal function to ~14% for LVEF <35%). — Braunwald's Heart Disease
Key Eponym Summary
| Eponym | Sign |
|---|---|
| Corrigan's pulse | Rapid-rise, rapid-fall carotid pulse |
| Austin Flint murmur | Apical mid-diastolic rumble (differential: mitral stenosis) |
| Hill sign | Femoral BP ≥40 mmHg > brachial (most reliable for severity) |
| de Musset sign | Head nodding |
| Duroziez sign | Femoral bruit on compression |
| Quincke's pulse | Capillary nail-bed pulsations |
| Müller sign | Systolic pulsations of the uvula |
| Traube sign | Pistol-shot sound over femoral artery |
Sources: Goldman-Cecil Medicine (p. 694–695) · Braunwald's Heart Disease · Harrison's Principles of Internal Medicine 22E · Guyton and Hall Medical Physiology (p. 293) · Morgan & Mikhail's Clinical Anesthesiology 7e
This is a shared conversation. Sign in to Orris to start your own chat.