diabetes mellitus
Reading File
Asking for Preferences
Finding Sources
Finding Sources
Searching PubMed
"diabetes mellitus" AND "management"
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Searching the Web
ADA 2025 standards of care diabetes mellitus guidelines key updates
Reading File
Reading File
Loading Image
Loading Image
Diabetes Mellitus — Comprehensive Medical Student Review
1. Definition & Epidemiology
Diabetes mellitus (DM) is the most common endocrine disease — a heterogeneous group of hyperglycemic disorders characterized by elevated serum glucose and disturbances of carbohydrate, fat, and protein metabolism arising from defects in insulin secretion, insulin action, or both. Approximately 9–10% of the US adult population is affected, and ~33% have prediabetes. Globally, prevalence is rising sharply, especially in Southeast Asia and the Western Pacific.
2. Classification (ADA)
The ADA recognizes four clinical types:
| Type | Core Mechanism |
|---|---|
| Type 1 DM (T1DM) | Autoimmune β-cell destruction → absolute insulin deficiency |
| Type 2 DM (T2DM) | Progressive β-cell failure on a background of insulin resistance; ~90–95% of all cases |
| Gestational DM (GDM) | Diabetes diagnosed in pregnancy, not clearly overt prior to gestation |
| Other specific types | Monogenic (MODY, neonatal DM), exocrine pancreas disease (cystic fibrosis, pancreatitis), drug/chemical-induced (glucocorticoids, HIV therapy, post-transplant) |
- Creasy & Resnik's Maternal-Fetal Medicine, p. 1425
- Harrison's Principles of Internal Medicine 22E, Chap. 416
3. Pathophysiology
Type 1 DM
T1DM is a chronic autoimmune disease in which T-lymphocytes and autoantibodies target pancreatic β cells in the islets of Langerhans, resulting in their destruction and absolute insulin deficiency. Autoimmune markers include:
- Islet cell autoantibodies (ICA)
- Anti-insulin autoantibodies (IAA)
- Anti-glutamic acid decarboxylase-65 (anti-GAD65)
- Islet antigen-2 (IA-2) autoantibodies
Genetic susceptibility (>60 loci, especially HLA-DR3/DR4) combined with environmental triggers (enteroviruses are prime suspects) initiates the autoimmune cascade. The result is near-complete loss of insulin secretion:
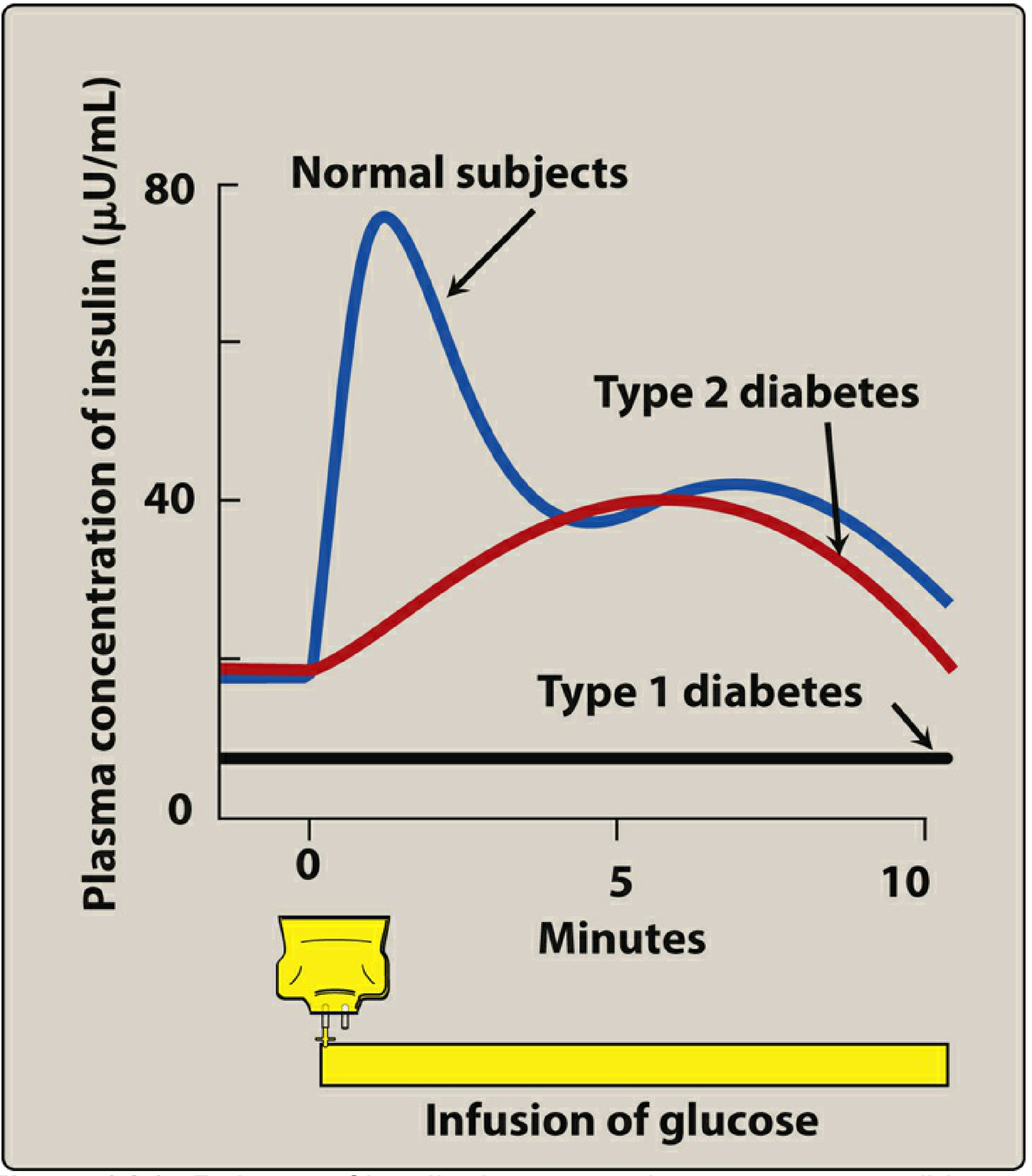
- Lippincott Illustrated Reviews: Pharmacology, p. 795
Type 2 DM
Two central defects operate simultaneously:
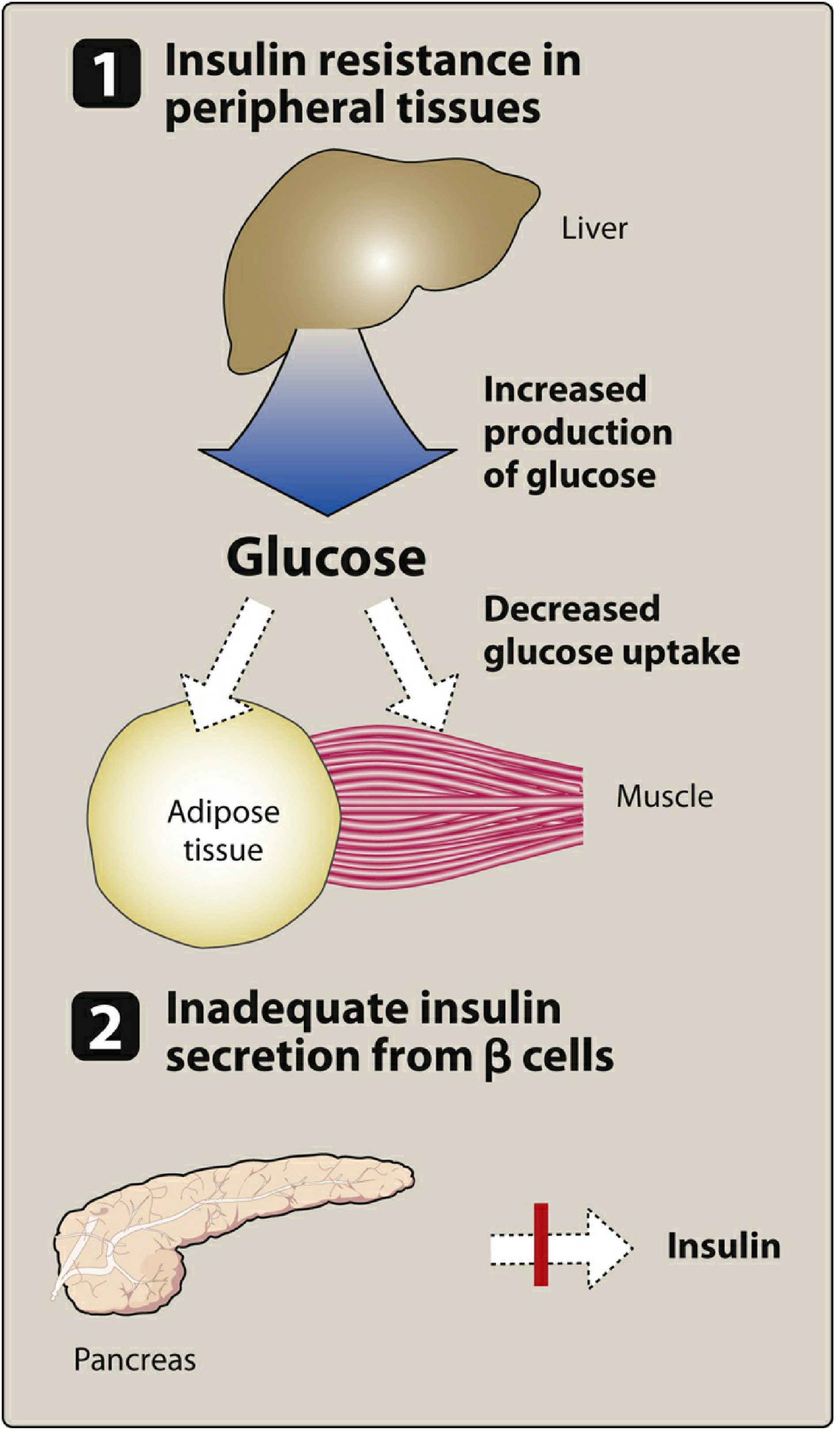
- Peripheral insulin resistance — decreased insulin sensitivity in liver (↑ gluconeogenesis), muscle (↓ glucose uptake), and adipose tissue (↑ lipolysis)
- β-cell secretory failure — initially compensatory hyperinsulinemia, but progressive β-cell apoptosis (driven by glucotoxicity and lipotoxicity) eventually produces relative insulin deficiency
Obesity, especially visceral fat accumulation, is the main driver of insulin resistance. Excess free fatty acids from visceral adipose impair insulin signaling via IRS-1/PI3K pathways and activate inflammatory mediators (TNF-α, IL-6).
- Guyton & Hall Textbook of Medical Physiology, p. 973–974
- Lippincott Illustrated Reviews: Biochemistry 8e, p. 797
4. Clinical Presentation
Classic "3 Ps" (from hyperglycemia/osmotic diuresis)
- Polyuria — glucose-driven osmotic diuresis
- Polydipsia — compensatory response to volume/osmotic loss
- Polyphagia — cellular starvation despite hyperglycemia (especially T1DM)
Additional features
- Weight loss (catabolism of protein and fat)
- Fatigue and weakness
- Blurred vision (osmotic lens changes)
- Frequent superficial infections (vaginitis, fungal skin infections)
- Slow wound healing
- T2DM may be asymptomatic at diagnosis; often discovered incidentally on screening
5. Diagnosis
Based on any one of the following (ADA criteria), confirmed on repeat testing unless unequivocal symptoms present:
| Test | Diabetes | Prediabetes |
|---|---|---|
| Fasting plasma glucose (FPG) | ≥126 mg/dL (7.0 mmol/L) | 100–125 mg/dL (IFG) |
| 2-hr glucose (75 g OGTT) | ≥200 mg/dL (11.1 mmol/L) | 140–199 mg/dL (IGT) |
| HbA1c | ≥6.5% | 5.7–6.4% |
| Random glucose + symptoms | ≥200 mg/dL | — |
HbA1c reflects average glucose over the prior ~3 months. The relationship is: eAG (mg/dL) = 28.7 × HbA1c – 46.7
(e.g., A1C 7% ≈ eAG 154 mg/dL)
(e.g., A1C 7% ≈ eAG 154 mg/dL)
- Tietz Textbook of Laboratory Medicine 7e, p. 2407
- Lippincott Illustrated Reviews: Pharmacology, p. 796
6. Management
Glycemic Targets (ADA)
| Parameter | Target |
|---|---|
| Fasting plasma glucose | 80–130 mg/dL |
| 2-hr postprandial glucose | <180 mg/dL |
| HbA1c (most patients) | <7% |
| HbA1c (gestational DM) | <6% |
| HbA1c (frail elderly/multiple comorbidities) | <8% |
A. Non-pharmacological
- Diet: Individualized macronutrient composition (~45% carbohydrate, 25–35% fat, 10–35% protein). Mediterranean-style eating improves glycemic control and reduces CVD risk. Limit refined carbohydrates.
- Exercise: Improves insulin sensitivity; recommended ≥150 min/week of moderate aerobic activity.
- Weight loss: Critical in T2DM — even 5–10% reduction significantly improves glucose, BP, and lipids.
- Patient education: Self-monitoring, hypoglycemia recognition, insulin adjustment for meals and exercise, sick-day management.
B. Pharmacotherapy — Type 1 DM
Insulin is mandatory. Goal: mimic physiologic secretion with a basal-bolus strategy.
| Insulin Type | Onset | Peak | Duration | Examples |
|---|---|---|---|---|
| Rapid-acting | <15 min | 0.5–1.5 hr | 3–5 hr | Aspart, Lispro, Glulisine |
| Short-acting | 30–60 min | 2–3 hr | 4–8 hr | Regular insulin |
| Inhaled | <15 min | 1–2 hr | ~3 hr | Inhaled human insulin |
| Intermediate | 2–4 hr | 4–10 hr | 10–16 hr | NPH |
| Long-acting | 2–4 hr | Flat | 20–24 hr | Glargine, Detemir |
| Ultra-long | 1–9 hr | Flat | ~42 hr | Degludec |
Delivery methods include multiple daily injections (MDI), continuous subcutaneous insulin infusion (CSII/insulin pump), and automated insulin delivery (AID) systems with continuous glucose monitoring (CGM).
ADA 2026 update: AID is now the preferred insulin delivery method — the first time it received the strongest recommendation for T2DM as well as T1DM.
- Harrison's Principles of Internal Medicine 22E, Table 416-1
C. Pharmacotherapy — Type 2 DM
Seven main drug classes are used:
| Class | Mechanism | Key Features |
|---|---|---|
| Metformin | ↓ hepatic gluconeogenesis (AMPK activation) | First-line; weight neutral; no hypoglycemia; cheap; avoid in severe renal/hepatic disease |
| Sulfonylureas (glipizide, glyburide, glimepiride) | ↑ insulin secretion (close K⁺-ATP channels on β cells) | Risk of hypoglycemia; weight gain |
| GLP-1 receptor agonists (semaglutide, liraglutide, dulaglutide) | ↑ glucose-dependent insulin secretion, ↓ glucagon, slow gastric emptying | Weight loss; CV and renal benefits; GI side effects |
| SGLT-2 inhibitors (empagliflozin, dapagliflozin, canagliflozin) | ↑ urinary glucose excretion | Weight loss; CV and renal benefits; risk of UTI/DKA |
| DPP-4 inhibitors (sitagliptin, saxagliptin) | Prolong endogenous GLP-1/GIP action | Weight neutral; well tolerated |
| Thiazolidinediones (pioglitazone) | ↑ peripheral insulin sensitivity (PPARγ agonist) | Weight gain; fluid retention; CV risk with rosiglitazone |
| Insulin | Exogenous replacement | Used when oral agents fail; multiple regimens |
Key evidence milestones:
-
DCCT (1993, T1DM): Intensive glucose control (HbA1c ~7.2%) reduced microvascular complications by ~60%
-
UKPDS (T2DM): Tight control (HbA1c reduced 9.1% → 7%) reduced microvascular complications vs. conventional therapy
-
GLP-1 RAs and SGLT-2 inhibitors now have cardiovascular outcome trial data supporting use in patients with established CVD, HF, or CKD
-
Katzung's Basic & Clinical Pharmacology 16e
-
Goldman-Cecil Medicine
7. Complications
Acute Complications
| Complication | Trigger | Key Features | Treatment |
|---|---|---|---|
| Diabetic Ketoacidosis (DKA) | Insulin deficiency (T1DM primarily; also T2DM) | Hyperglycemia + anion gap metabolic acidosis + ketonemia; N/V, abdominal pain, Kussmaul breathing, fruity breath | Insulin infusion, IV fluids, K⁺ replacement, treat precipitant |
| Hyperglycemic Hyperosmolar State (HHS) | Relative insulin deficiency (T2DM, elderly) | Extreme hyperglycemia (>600 mg/dL), hyperosmolality, NO significant ketoacidosis, profound dehydration | Aggressive IV fluids, insulin, treat precipitant (infection, MI, CVA) |
| Hypoglycemia | Excess insulin / secretagogues, missed meals, exercise | Sweating, tremor, confusion, seizure (<54 mg/dL = severe) | Oral glucose (15-15 rule); IV dextrose or glucagon if unconscious |
Sodium bicarbonate has no demonstrated benefit in DKA treatment and may worsen outcomes.
- Rosen's Emergency Medicine
Chronic Complications
Microvascular (diabetes-specific; caused by chronic hyperglycemia):
- Retinopathy: Non-proliferative → proliferative → vitreous hemorrhage, blindness. Leading cause of adult blindness in the US. Screen annually from T1DM diagnosis + 5 yrs; at T2DM diagnosis.
- Nephropathy (Diabetic Kidney Disease): Microalbuminuria → macroalbuminuria → declining GFR → ESRD. Leading cause of renal failure. ACE inhibitors/ARBs are first-line.
- Neuropathy: Distal symmetric polyneuropathy (most common), autonomic neuropathy (gastroparesis, orthostatic hypotension, sexual dysfunction), mononeuropathy. Treatment: gabapentin, pregabalin, duloxetine.
Macrovascular (shared with general population; accelerated in DM):
- Coronary heart disease / ASCVD — 2–4× higher mortality
- Peripheral arterial disease → foot ulcers → amputation (leading cause of non-traumatic amputation)
- Cerebrovascular disease
Other complications:
-
Infections (impaired neutrophil function, poor circulation): polymicrobial foot infections, Fournier's gangrene
-
Cataracts, glaucoma
-
Gastroparesis
-
Cheiroarthropathy (stiff hand syndrome)
-
Hearing loss, periodontal disease, cognitive impairment
-
Harrison's Principles of Internal Medicine 22E, Chap. 417
-
Robbins, Cotran & Kumar Pathologic Basis of Disease
-
Washington Manual of Medical Therapeutics
8. Monitoring & Targets
| Parameter | Target / Frequency |
|---|---|
| HbA1c | <7% every 3 months until stable, then every 6 months |
| Blood pressure | <130/80 mmHg |
| LDL cholesterol | <100 mg/dL (<70 in high CVD risk) |
| Foot examination | Annually (monofilament, vibration, pulses, inspection) |
| Ophthalmology | Annually |
| Urine albumin/creatinine ratio | Annually |
| eGFR | Annually |
| Depression/psychosocial screening | At diagnosis and periodically |
9. Special Situations
- Gestational DM: Screen all pregnancies at 24–28 weeks with 1-hr 50 g GCT (if ≥140 mg/dL → 3-hr OGTT). GDM carries risk of fetal macrosomia, birth complications, neonatal hypoglycemia, and maternal T2DM later in life. Treatment: diet first, insulin or metformin if needed.
- Prediabetes: IFG + IGT → lifestyle intervention (weight loss 7%, 150 min/week exercise) reduces progression to T2DM by 58% (DPP trial).
Summary Comparison: T1DM vs. T2DM
| Feature | T1DM | T2DM |
|---|---|---|
| Mechanism | Autoimmune β-cell destruction | Insulin resistance + β-cell failure |
| % of DM | 5–10% | 90–95% |
| Typical age | Childhood/teens (peak 10–14 yr) | >30 yr (but rising in youth) |
| Body habitus | Normal/thin | Often overweight/obese |
| Ketosis | Common | Rare |
| Autoantibodies | Present (GAD65, ICA, IAA) | Absent |
| First-line Rx | Insulin (mandatory) | Lifestyle + Metformin |
| Genetic | HLA-DR3/DR4 | Polygenic; strong lifestyle component |
Key landmark trials to know: DCCT (T1DM, microvascular benefit of tight control), UKPDS (T2DM, tight glycemic and BP control), EMPA-REG OUTCOME and LEADER (CV/renal benefits of SGLT-2i and GLP-1 RAs respectively).
Recent guideline note (ADA 2026): Automated Insulin Delivery (AID) systems have received the strongest recommendation for both T1DM and T2DM patients requiring insulin — a significant upgrade from previous years.
Sources: Harrison's Principles of Internal Medicine 22E (2025) · Lippincott Illustrated Reviews: Pharmacology & Biochemistry · Guyton & Hall Textbook of Medical Physiology · Katzung's Basic & Clinical Pharmacology 16e · Rosen's Emergency Medicine · Tietz Textbook of Laboratory Medicine 7e · Washington Manual of Medical Therapeutics · Robbins Pathologic Basis of Disease · Creasy & Resnik's Maternal-Fetal Medicine
This is a shared conversation. Sign in to Orris to start your own chat.