Toxocariasis. Fascioliasis. Etiology, epidemiology, pathogenesis, sign and symptoms, diagnosis, differential diagnosis, treatment, prevention
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Loading Image
Reading File
Searching Images
Toxocara larva migrans eosinophilic granuloma histology
Loading Image
Loading Image
Searching Images
Fasciola hepatica liver CT scan track lesions acute phase
Loading Image
Here is a comprehensive review of both conditions.
TOXOCARIASIS
Etiology
Toxocariasis is caused by Toxocara species — nematode roundworms of dogs (Toxocara canis) and cats (Toxocara cati). Most human cases are due to T. canis. Humans are accidental (dead-end) hosts who cannot sustain the full lifecycle of these parasites. — Goldman-Cecil Medicine, Red Book 2021
Epidemiology
- Seroprevalence: ~5% of the US population ≥6 years shows serologic evidence of prior infection
- Visceral larva migrans (VLM): primarily affects children aged 2–7 years
- Ocular larva migrans (OLM): typically older children and adolescents; majority of US OLM cases are in southern states
- Risk factors: dog/cat ownership, poverty, hot/humid regions (eggs survive longer in warm, moist soil)
- Transmission: ingestion of embryonated eggs from soil contaminated by dog/cat feces — sandboxes, playgrounds. Direct animal contact is not necessary because freshly shed feces are not immediately infective
- Egg maturation: eggs become infective after 2–4 weeks in the environment
— Red Book 2021, Goldman-Cecil Medicine
Pathogenesis
The lifecycle in the definitive (animal) host mirrors Ascaris lumbricoides in humans: larvae penetrate the intestinal wall → travel via vasculature to the lungs → ascend the bronchial tree → are swallowed → develop into adults in the gut.
In the human accidental host, the released larvae cannot develop into adults. Instead, they migrate through organs (liver, lungs, CNS, eyes) continuously but never mature. Eventually, dying larvae induce significant:
- Immediate-type hypersensitivity reactions
- Delayed-type hypersensitivity reactions
- Formation of eosinophilic granulomas
Notably, VLM and OLM are generally mutually exclusive — heavy systemic larval burden tends to lead to VLM, while low-burden infection in older/more immune hosts tends to result in OLM. — Goldman-Cecil Medicine
Clinical Manifestations
1. Covert Toxocariasis
The most common form — persistent eosinophilia with minimal or no symptoms, representing ongoing larval migration.
2. Visceral Larva Migrans (VLM)
- Low-grade fever, cough, wheezing (pulmonary involvement)
- Hepatosplenomegaly → right upper quadrant pain
- Symptoms appear gradually and resolve over 4–8 weeks
- Less common: myocarditis, nephritis, rash
3. Neurotoxocariasis
- Eosinophilic meningoencephalitis, space-occupying CNS lesions
- Myelitis, cerebral vasculitis
- Can present with seizures, encephalopathy, neuropsychiatric symptoms
4. Ocular Larva Migrans (OLM)
- Unilateral visual impairment, often without systemic signs
- Strabismus may accompany it
- Can cause uveitis, endophthalmitis, retinal granulomas
- Difficult to distinguish from retinoblastoma or intraocular TB
Laboratory abnormalities: leukocytosis, marked eosinophilia, hypergammaglobulinemia, elevated isohemagglutinin titers to A/B blood group antigens (visceral disease)
— Goldman-Cecil Medicine, Red Book 2021
Diagnosis
| Test | Details |
|---|---|
| ELISA (Toxocara antibodies) | Test of choice; available through CDC; less sensitive for OLM |
| Immunoblot confirmation | Used for positive or equivocal ELISA results |
| Liver imaging (US/CT/MRI) | Diffuse nodular lesions <2 cm; multiple hypodense foci |
| Liver biopsy | Rarely needed; low sensitivity/specificity; shows eosinophilic granulomas |
| PCR | Not currently available clinically |
| OLM: fluorescein angiography / CT | Differentiate from retinoblastoma; elevated anti-Toxocara Abs in vitreous/aqueous fluid are supportive |
Key caveat: A positive antibody test does not distinguish active from past infection. — Goldman-Cecil Medicine, Red Book 2021
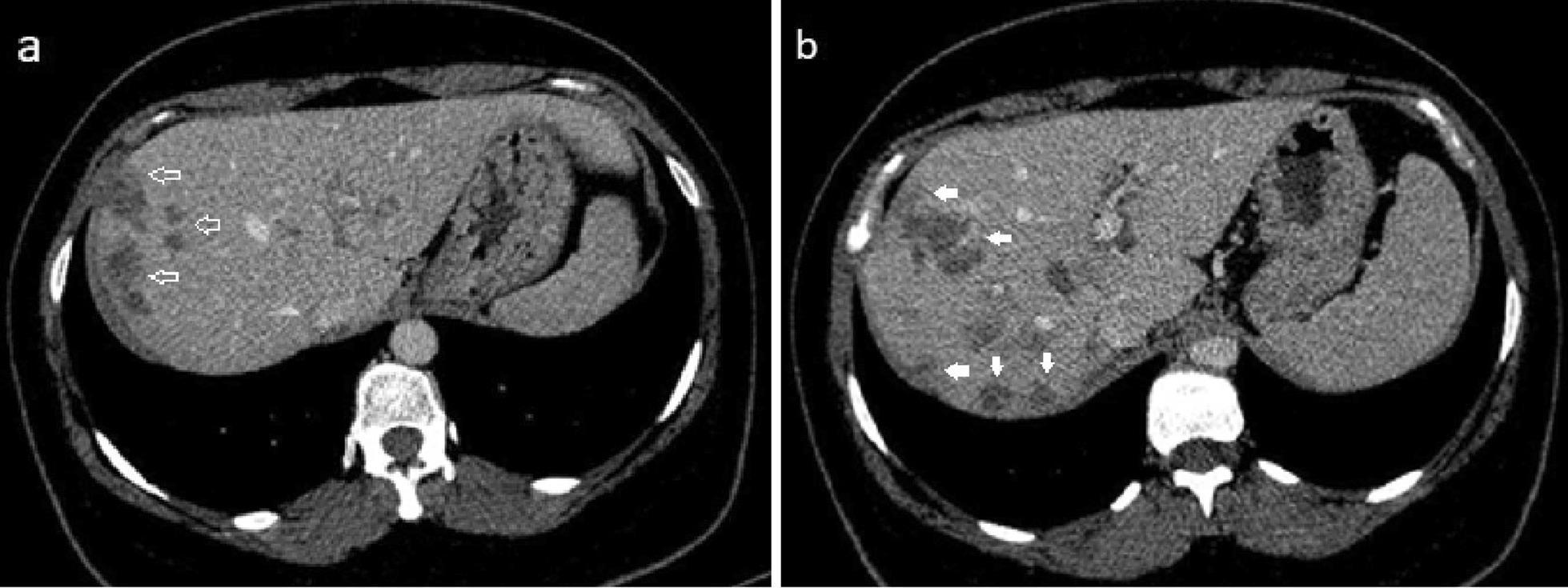
CT abdomen: Hepatic toxocariasis — multiple small hypodense nodules scattered through the liver (left, acute phase); calcific residual lesions after treatment (right, 1 year follow-up)
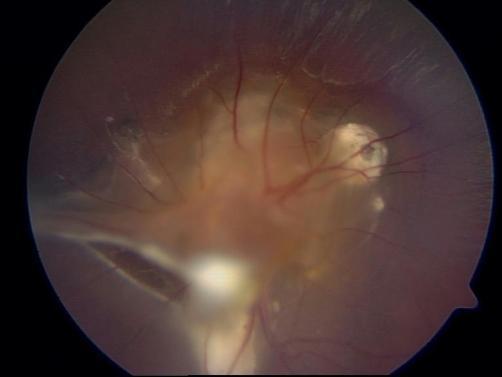
Ocular larva migrans: fundus photograph showing a central yellowish-white retinal granuloma, satellite granulomas, and epiretinal fibrous traction bands extending toward the periphery
Differential Diagnosis
- Visceral form: other causes of eosinophilia with organomegaly — Ascaris, trichinellosis, schistosomiasis, strongyloidiasis; hepatic metastases (imaging may mimic)
- Ocular form: retinoblastoma, intraocular TB, endophthalmitis from other causes, Coats disease
- Neurotoxocariasis: other causes of eosinophilic meningitis (angiostrongyliasis), CNS lymphoma/granuloma
- Cutaneous larva migrans, sparganosis (for migratory skin nodules) — Dermatology 2-Volume Set 5e
Treatment
| Drug | Regimen | Indication |
|---|---|---|
| Albendazole (first-line) | 400 mg twice daily × 5 days | VLM and OLM |
| Mebendazole | Alternative | VLM |
| Corticosteroids (prednisone 60 mg/day × 5 days) | Adjunct | Severe pulmonary, cardiac, or neurologic VLM |
| Corticosteroids (oral/topical) | Control intraocular inflammation | OLM |
| Surgical therapy | Vitreoretinal intervention | Complicated OLM (vitreoretinal traction) |
Albendazole is FDA-approved (though not specifically for this indication) and is safe in children as young as 1 year. — Goldman-Cecil Medicine, Red Book 2021
Prevention
- Proper disposal of dog and cat feces
- Regular deworming of dogs/cats (especially puppies/kittens)
- Cover sandboxes when not in use
- Handwashing after contact with soil
- No specific post-exposure prophylaxis is recommended
- No person-to-person transmission
— Red Book 2021
FASCIOLIASIS
Etiology
Fascioliasis is caused by the liver flukes Fasciola hepatica (adult: 30 × 13 mm; up to 3 cm long) and F. gigantica (adult: up to 75 × 20 mm; up to 7.5 cm long). Both are trematodes (flukes) with herbivores (cattle, sheep, goats) as natural definitive hosts. Humans are incidental hosts. — Goldman-Cecil Medicine, Tietz Textbook of Laboratory Medicine
Epidemiology
- Global burden: estimated 2.4–2.6 million people infected in >70 countries; approximately 10,635 new cases in 2010 (90,041 DALYs)
- Geographic distribution: F. hepatica — Americas, Europe, Oceania, Africa, Asia; F. gigantica — Africa and Asia only
- Highest prevalence: Peru and Bolivia (>60%)
- Women have higher incidence, with more severe infections and complications
- Risk groups: consumers of raw freshwater plants — watercress, water lettuce, alfalfa, mint, parsley, khat
- In the USA, cases are primarily imported (immigrants, returning travelers); rare autochthonous cases reported
— Goldman-Cecil Medicine, Tietz Textbook of Laboratory Medicine
Life Cycle & Pathogenesis
- Eggs passed in feces → deposited in water
- Miracidia hatch in 9–14 days → infect freshwater snails (Lymnaea spp.) — the first intermediate host
- In snails (4–7 weeks): sporozoites → rediae → cercariae
- Free-swimming cercariae encyst on aquatic plants as metacercariae — the second intermediate host
- Humans ingest contaminated raw vegetables; metacercariae excyst in the duodenum
- Larvae penetrate the intestinal wall → peritoneal cavity → penetrate Glisson's capsule of the liver (~4 weeks)
- Larvae migrate through liver parenchyma → causing ongoing inflammatory tracks → reach bile ducts after 3–5 months
- Adults mature in hepatic and common bile ducts (and sometimes the gallbladder); eggs shed into bile → stool
- Adults can live in bile ducts up to 13 years
Histopathology: liver contains dilated, thick-walled, calcareous bile ducts with yellow-brown bile and marked periductal fibrosis. Migration tracks visible on imaging or histology. — Goldman-Cecil Medicine
Clinical Manifestations
Acute (Invasive/Migratory) Phase
During larval migration through liver parenchyma:
- Prolonged fever, hepatomegaly, right upper quadrant pain
- Significant eosinophilia (hallmark of this phase)
- Anorexia, weight loss, nausea, vomiting
- Cough, urticaria, lymphadenopathy, arthralgias
- Brief acute diarrhea (2–5 days) may precede liver invasion
- Hyperbilirubinemia is notably absent in this phase
- Rarely: subcapsular liver hematoma (intense hemorrhage), ectopic migration to subcutaneous tissue, pancreas, eye, brain, stomach wall
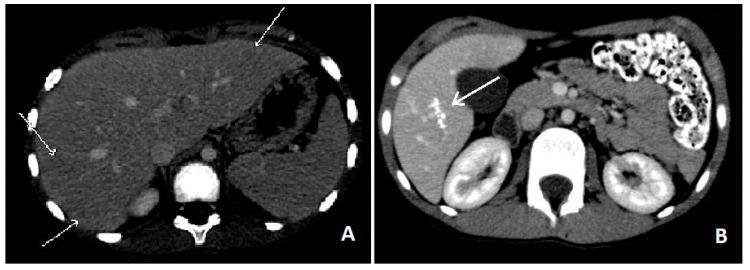
CT findings (acute): multiple hypodense lesions resembling metastases, but changing in position, attenuation, and shape over time (the hallmark distinguishing feature). Track-like lesions, subcapsular hematoma, enhancement of Glisson's capsule.
Chronic (Obstructive/Biliary) Phase
When flukes reside in bile ducts:
- Colicky right upper quadrant pain and epigastric pain (biliary obstruction)
- Eosinophilia absent in ~50% of chronic cases
- Elevated alkaline phosphatase (biliary obstruction)
- Complications: cholangitis, gallstones, hemobilia, liver abscess, liver fibrosis, biliary stricture
- Adult flukes visible on ERCP
— Goldman-Cecil Medicine
Diagnosis
Acute Phase
| Test | Notes |
|---|---|
| Serology (Fas2 ELISA / cathepsin L1-based ELISA) | Sensitivity 92%, specificity 84%; test of choice in acute phase; antibodies detectable within 2–4 weeks of infection |
| CT/MRI/US liver | Track-like, shifting hypodense lesions; useful but may resemble metastases |
| Stool exam | Negative in acute phase (eggs not yet shed — adults not established) |
| Liver biopsy | Only if serology and imaging unavailable; flukes may be seen migrating through parenchyma |
Chronic Phase
| Test | Notes |
|---|---|
| Stool microscopy (Lumbreras rapid sedimentation + Kato-Katz) | Eggs shed into stool after adult establishment; ≥3 stool samples preferred |
| Serology | Helpful if stool negative; takes >1 year to resolve after cure |
| ERCP | Directly visualizes and can retrieve adult flukes from bile duct |
| US/CT | Low sensitivity in chronic phase |
Egg morphology: Large oval, yellow-brown, thin-shelled, 130–150 µm × 63–90 µm with small operculum. F. gigantica eggs up to 200 µm. Eggs of F. hepatica and Fasciolopsis buski are nearly indistinguishable — clinical correlation required. Spurious passage of eggs occurs if infected animal liver is ingested. — Tietz Textbook of Laboratory Medicine, Goldman-Cecil Medicine
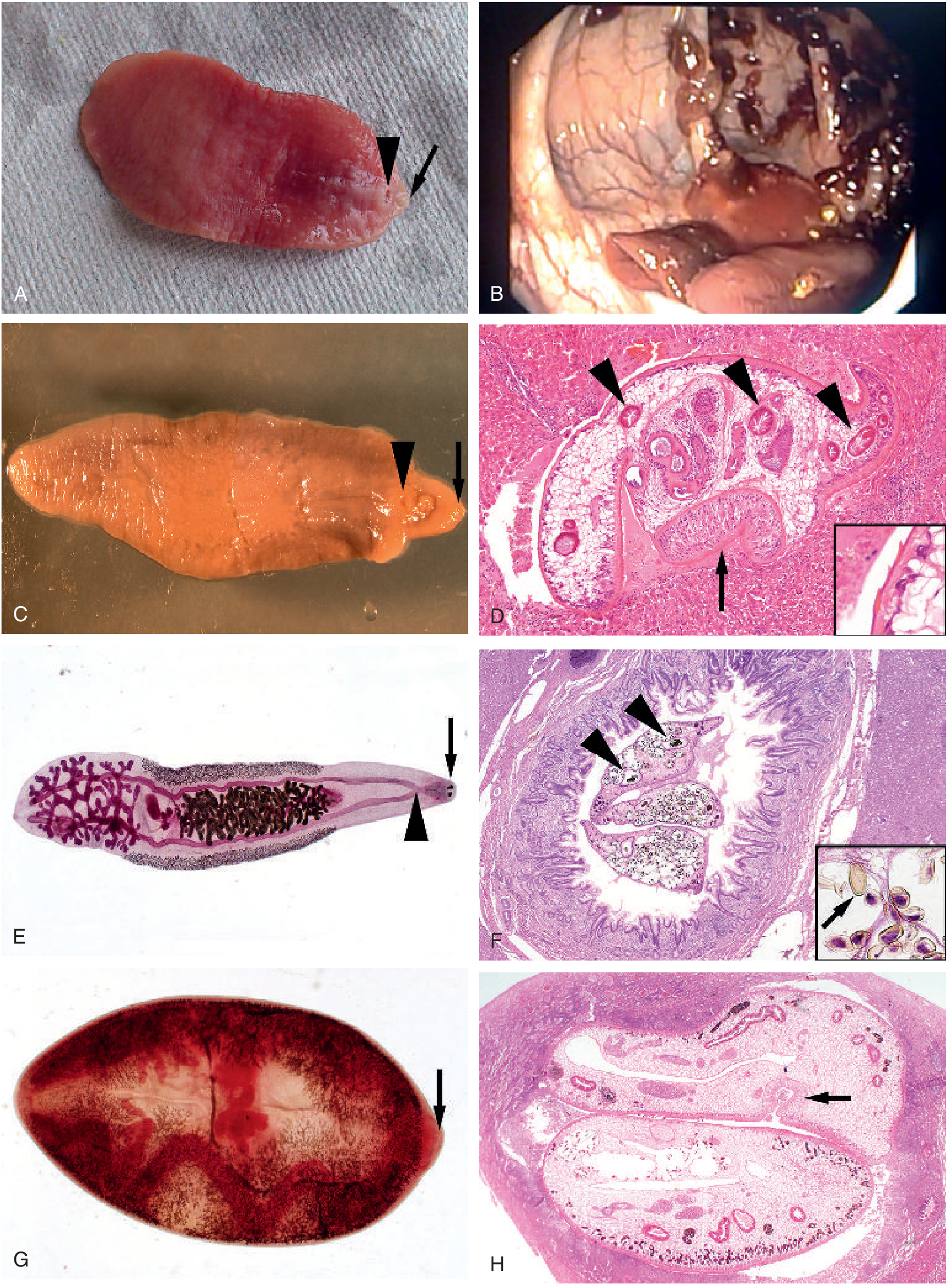
Fasciola hepatica adult (C) showing oral sucker (arrow) and ventral sucker (arrowhead) with characteristic cone-shaped anterior end; (D) F. hepatica migrating through liver parenchyma showing intestinal branches in cross-section (H&E, ×100)
CT abdomen: Hepatic fascioliasis (acute phase) — multiple ill-defined hypodense lesions in the right lobe with characteristic "tunnels and caves" appearance representing larval migration tracks (white arrows)
Differential Diagnosis
Acute phase
- Acute cholecystitis (clinically similar, but fascioliasis has eosinophilia and absent hyperbilirubinemia)
- Hepatic metastases / liver abscess (imaging may mimic; metastases don't shift position over time)
- Liver cell carcinoma
- Other causes of hepatomegaly + eosinophilia: visceral toxocariasis, schistosomiasis, echinococcosis, ascariasis
- Viral hepatitis (no eosinophilia)
Chronic phase
- Cholangitis, choledocholithiasis from other causes
- Cholangiocarcinoma
- Primary sclerosing cholangitis
- Other liver flukes (Clonorchis, Opisthorchis — but respond to praziquantel, unlike Fasciola which is resistant)
— Goldman-Cecil Medicine
Treatment
| Drug | Regimen | Notes |
|---|---|---|
| Triclabendazole (TCZ) — first-line | 10 mg/kg orally on 2 consecutive days | Cure rate >90%; only drug effective against both immature and adult flukes |
| Praziquantel | NOT effective against Fasciola | Effective for other liver flukes (Clonorchis, Opisthorchis) |
| Multiple TCZ rounds | Used for treatment failure | Some infections are resistant to TCZ |
| Surgical/ERCP | For biliary obstruction | Dead parasites may cause acute obstruction in chronic phase |
Serology takes >1 year to resolve after cure. Stool exams should remain negative for ≥3 months to confirm cure. — Goldman-Cecil Medicine
Prevention
- Avoid consumption of raw freshwater aquatic plants (watercress, water lettuce, etc.) in endemic regions
- Wash and cook vegetables thoroughly
- Avoid drinking untreated water from streams or ponds
- Livestock management: control of Lymnaea snail habitat; anthelmintic treatment of cattle/sheep
- Public health education in endemic areas (Peru, Bolivia, Egypt, Iran)
- No vaccine available
Summary Comparison Table
| Feature | Toxocariasis | Fascioliasis |
|---|---|---|
| Causative agent | Toxocara canis / T. cati (nematode) | Fasciola hepatica / F. gigantica (trematode) |
| Primary host | Dogs, cats | Cattle, sheep, goats |
| Route of infection | Ingestion of embryonated eggs from soil | Ingestion of metacercariae on raw aquatic plants |
| Intermediate host | None (eggs directly infective) | Lymnaea snail (1st), aquatic plants (2nd) |
| Key finding | Eosinophilia, eosinophilic granulomas | Eosinophilia, liver track lesions |
| Diagnosis | ELISA (antibodies); liver US/CT | Fas2/cathepsin L1 ELISA (acute); stool eggs (chronic) |
| Treatment | Albendazole 400 mg BID × 5 days | Triclabendazole 10 mg/kg × 2 days |
| Praziquantel | Not used | Not effective (unlike other flukes) |
| Prevention | Deworm pets; cover sandboxes; hygiene | Avoid raw watercress; cook plants; snail control |
Sources: Goldman-Cecil Medicine (9780323930345); Red Book 2021 Report of the Committee on Infectious Diseases (9781610025218); Tietz Textbook of Laboratory Medicine, 7th ed. (9780323775724); Sleisenger and Fordtran's Gastrointestinal and Liver Disease (9780323609623)
This is a shared conversation. Sign in to Orris to start your own chat.