Classify subcutaneous mycoses.discuss mycetoma in 10 marks format for 3rd yr dermatology post graduate theory examination
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
I now have all the information needed for a comprehensive 10-mark answer. Let me compile the complete examination answer.
Classification of Subcutaneous Mycoses & Mycetoma — 3rd Year Dermatology PG Theory (10 Marks)
CLASSIFICATION OF SUBCUTANEOUS MYCOSES
Subcutaneous mycoses (also called mycoses of implantation) are infections caused by fungi or filamentous bacteria introduced directly into the dermis/subcutis through penetrating injury. They are predominantly tropical/subtropical infections designated as Neglected Tropical Diseases (NTDs) by the WHO.
Classification
| Category | Disease | Causative Organism(s) |
|---|---|---|
| True fungi (Eumycetes) | Eumycetoma | Madurella mycetomatis, Scedosporium apiospermum, Fusarium spp., Acremonium spp. |
| Chromoblastomycosis | Fonsecaea pedrosoi, Cladophialophora carrionii, Phialophora verrucosa | |
| Sporotrichosis | Sporothrix schenckii complex | |
| Phaeohyphomycosis | Exophiala spp., Alternaria spp. | |
| Lobomycosis (Lacaziosis) | Lacazia loboi | |
| Filamentous bacteria (Actinomycetes) | Actinomycetoma | Nocardia brasiliensis, Actinomadura madurae, A. pelletieri, Streptomyces somaliensis |
| Zygomycetes/Entomophthorales | Basidiobolomycosis | Basidiobolus ranarum |
| Conidiobolomycosis | Conidiobolus coronatus | |
| Subcutaneous mucormycosis | Mucor, Rhizopus spp. |
Fitzpatrick's Dermatology, 9th ed. — The most common subcutaneous mycoses are sporotrichosis, mycetoma, and chromoblastomycosis. All three are WHO-classified NTDs.
MYCETOMA (10 Marks)
1. Definition
Mycetoma (syn. Madura foot, maduromycosis) is a chronic, granulomatous, progressive subcutaneous infection caused by either true fungi (eumycetoma) or aerobic filamentous bacteria/actinomycetes (actinomycetoma), characterized by the clinical triad of:
- Painless subcutaneous swelling (tumefaction)
- Multiple sinus tracts
- Discharge of characteristic grains (granules)
The term derives from Madura, India, where it was first formally described by Dr. John Gill in 1842.
2. Etiology & Classification
A. Actinomycetoma (caused by filamentous bacteria)
| Organism | Grain color |
|---|---|
| Nocardia brasiliensis | White/pale yellow |
| Nocardia asteroides | White/pale yellow |
| Actinomadura madurae | White/pale (large, up to 5 mm) |
| Actinomadura pelletieri | Red/pink (pathognomonic) |
| Streptomyces somaliensis | Yellow-brown |
| Actinomyces israelii | Yellow ("sulfur granules") |
B. Eumycetoma (caused by true fungi)
| Organism | Grain color |
|---|---|
| Madurella mycetomatis | Black/dark (most common worldwide) |
| Madurella grisea, Trematosphaeria grisea | Black |
| Scedosporium apiospermum | White/pale |
| Fusarium spp., Acremonium spp. | White/pale |
| Aspergillus nidulans | White |
| Neotestudina rosatii | White |
Key rules for grain color:
- Black grains → always eumycetoma (fungi)
- Red grains → always actinomycetoma (bacteria)
- Pale/white grains → either etiology (requires microscopy to distinguish)
3. Epidemiology
- Endemic in the "Mycetoma belt" — between latitudes 15°S and 30°N (arid tropics/subtropics with low annual rainfall)
- Africa: Sudan, Senegal, Somalia (highest burden); Asia: India, Yemen; Americas: Mexico (Nocardia dominant), Venezuela, Argentina
- Most common organism globally: Madurella mycetomatis
- Most common in Central America/Mexico: Nocardia brasiliensis (actinomycetoma)
- Actinomycetoma : Eumycetoma = 3:1 (fortunate, as actinomycetoma is more treatment-responsive)
- Male predominance: M:F = 2:1 to 5:1; typical patient is a barefoot male agricultural worker aged 20–50 years
- Portal of entry: traumatic implantation (thorn prick, splinter, barefoot walking); no person-to-person transmission
- Risk factors: lack of protective footwear, malnutrition, exposed abrasions
4. Pathogenesis
Organisms (soil/plant saprophytes — isolated from Acacia thorns in endemic areas) are introduced subcutaneously through penetrating injury → localized infection → formation of grains (compact aggregates of organisms within suppurative abscesses) → progressive spread by direct contiguity to dermis → subcutaneous tissue → fascia → bone (osteomyelitis). Distant dissemination is exceptionally rare.
The organism evades host defenses via:
- Cell-wall thickening
- Melanin deposition (in pigmented fungi)
- Formation of the grain matrix — protects inner organisms from immune cells
5. Clinical Features
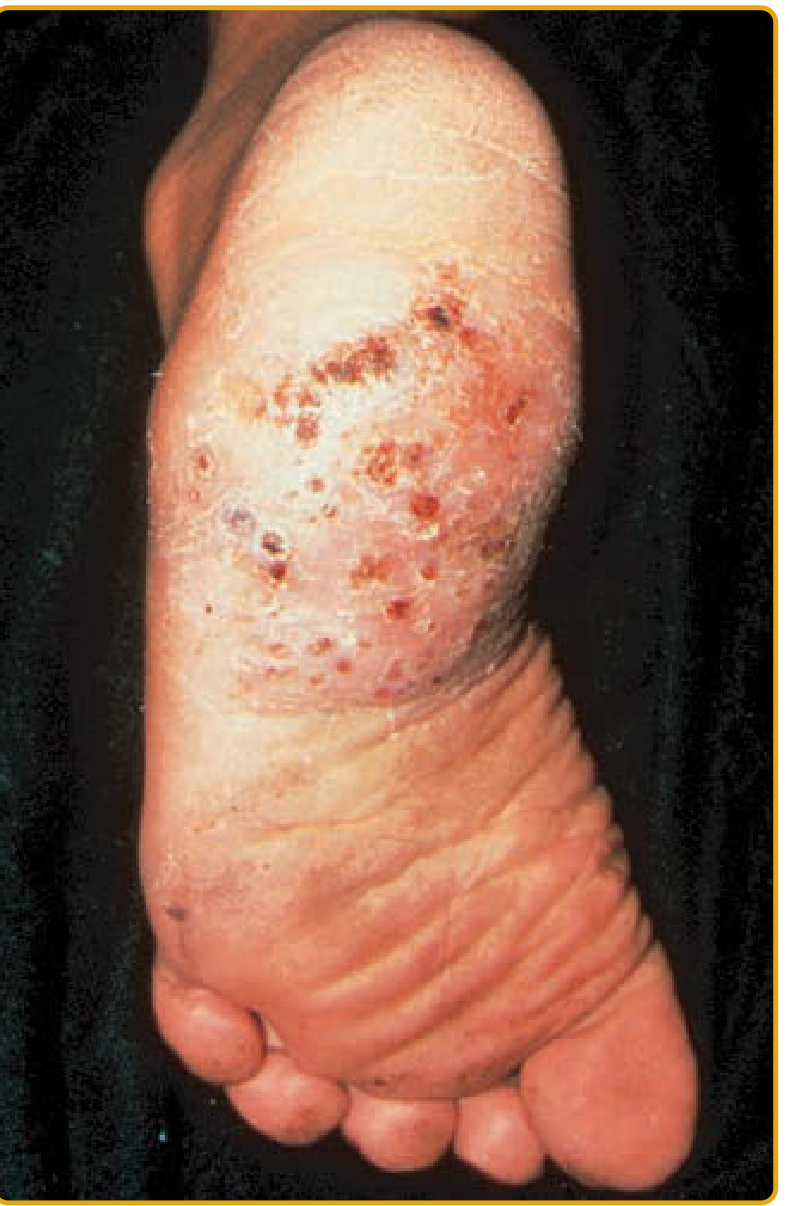
Fitzpatrick's Fig. 162-2: Mycetoma — brawny edema and crusted papules on plantar surface
Classic triad:
- Tumefaction — firm, painless, progressive swelling (begins as a single firm painless papule/nodule)
- Sinuses — multiple draining sinus tracts opening onto the skin surface
- Grains — colored granules discharged in sero-purulent material
Common sites (in order): foot/instep (most common) → lower leg → hand → chest wall (Nocardia) → scalp → buttocks. Lesions on covered areas are almost always actinomycetomas.
Disease stages:
- Early: Firm, painless subcutaneous nodule; skin may appear normal
- Intermediate: Papules, pustules, multiple sinus tracts discharging grains; brawny edema
- Late: Gross deformity, fibrosis, sinus tracts, bone destruction; disease generally remains surprisingly asymptomatic even with advanced involvement
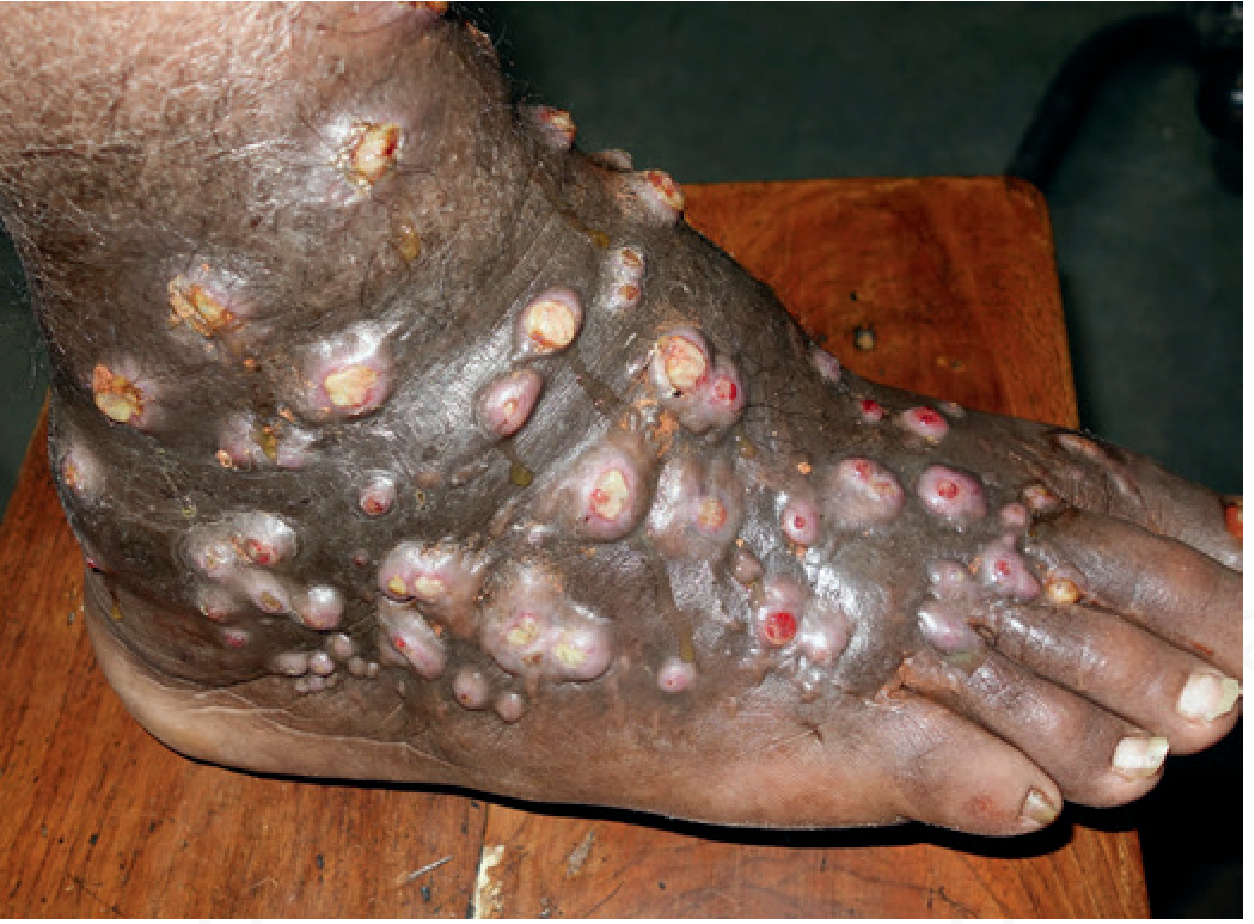
Andrews' Fig. 15.29: Advanced mycetoma — soft tissue swelling with multiple nodular sinus tracts discharging grains
6. Histopathology
- Pseudoepitheliomatous hyperplasia of overlying epidermis
- Suppurative and granulomatous inflammation of dermis and subcutis with fibrosis
- Stellate abscesses containing grains surrounded by neutrophils, giant cells, and fibrosis
- Grains = compact aggregates of organisms (250–1000 μm, visible to naked eye)
- Actinomycetoma grains: fine filaments 1–2 μm diameter; Gram-positive; Gram stain shows thin filaments embedded in Gram-negative matrix; club formation at periphery
- Eumycetoma grains: broader hyphae 2–5 μm; PAS and GMS (Grocott-Methenamine Silver) stain clearly shows fungal hyphae and structures
- Distinguishing rule on H&E: filament diameter relative to nuclei of surrounding inflammatory cells — thick = eumycetoma; thin = actinomycetoma
7. Investigations & Diagnosis
Diagnosis = clinical triad confirmed by grain identification
| Investigation | Findings |
|---|---|
| Grain examination (KOH mount) | Grains 250–1000 μm; microscopy shows filament caliber (fine = actino; broad = eumy) |
| Gram stain of grain | Actinomycetoma: Gram-positive thin filaments (1–2 μm) in Gram-negative matrix |
| PAS / GMS stain | Eumycetoma: thick hyphae (2–5 μm), fungal structures clearly shown |
| Culture | Definitive species identification; use multiple media; incubate at 25°C and 37°C |
| X-ray | Periosteal erosion, periosteal proliferation, lytic (punched-out) lesions in bone — "cavity with sclerotic margin" |
| MRI | "Dot-in-a-circle sign" — pathognomonic; grains appear as low-signal dots surrounded by high-signal halo on T2 |
| Ultrasound | Hyperechoic dots (grains) within hypoechoic cavities |
| PCR | Species identification using specific primers — used in reference centers |
| Serology | Limited utility; helpful in S. somaliensis infection as guide to therapeutic response |
The "dot-in-a-circle" sign on MRI is considered pathognomonic for mycetoma — the low-intensity dots represent grains surrounded by granulomatous tissue.
8. Differential Diagnosis
| Condition | Distinguishing features |
|---|---|
| Botryomycosis | Caused by true bacteria (S. aureus, Pseudomonas); grains present but bacteria on culture |
| Actinomycosis | Actinomyces israelii; typically involves jaw/abdomen/thorax; yellow sulfur granules |
| Chromoblastomycosis | No grains; muriform cells (sclerotic bodies) on histology |
| Chronic osteomyelitis (bacterial/tuberculous) | No surface grains; bacteriological/mycobacterial culture |
| Elephantiasis | No sinuses or grains; filaria serology |
| Kaposi's sarcoma | No grains; characteristic histology |
9. Treatment
Actinomycetoma (more treatment-responsive)
| Regimen | Drugs |
|---|---|
| First-line (Nocardia spp.) | Cotrimoxazole (sulfamethoxazole–trimethoprim) + rifampicin OR streptomycin |
| First-line (A. madurae, A. pelletieri) | Dapsone + streptomycin (Mahgoub's regimen) |
| S. somaliensis | Cotrimoxazole + streptomycin |
| A. israelii | High-dose penicillin (curative) |
| Refractory/severe | Amikacin + imipenem, or moxifloxacin |
| Duration | 6–24 months depending on response |
Eumycetoma (less treatment-responsive)
| Regimen | Details |
|---|---|
| Medical | Itraconazole 200–400 mg/day OR voriconazole 200–400 mg/day OR ketoconazole 200 mg/day × months; responses unpredictable but may slow progression |
| P. boydii / Scedosporium | Voriconazole (drug of choice) or posaconazole ± surgical excision |
| M. mycetomatis | Itraconazole 200 mg/day, may respond |
| Surgery | Wide local excision for early, localized lesions; surgery + antifungals for advanced cases |
| Amputation | Reserved for massive, refractory, advanced eumycetoma — amputation rate historically high (25–30%) |
Key principle: Actinomycetoma = primarily medical therapy; Eumycetoma = combined medical + surgical (and sometimes amputation).
No spontaneous remission occurs in either form.
10. Prognosis & Key Points for Examination
| Feature | Actinomycetoma | Eumycetoma |
|---|---|---|
| Response to treatment | Good (antibiotics) | Poor (antifungals unreliable) |
| Grain filament thickness | 1–2 μm (thin) | 2–5 μm (thick) |
| Typical grain stain | Gram-positive | PAS/GMS positive |
| Bone involvement | Common | Common |
| Amputation risk | Low | High |
High-yield mnemonics:
- "MAD" organisms: Madurella, Actinomadura, Dermatophilus — classic mycetoma agents
- Black grain = Fungus; Red grain = Actino; White grain = Either
- Triad: Tumefaction + Sinuses + Grains
- MRI sign: "Dot-in-a-circle" = pathognomonic
- WHO NTD: Mycetoma, Chromoblastomycosis, and Sporotrichosis (all three = NTDs)
References
- Fitzpatrick's Dermatology, 9th ed., Chapter 162 — Subcutaneous Mycoses
- Andrews' Diseases of the Skin, 13th ed., Chapter 15
- Dermatology (Bolognia/Cerroni), 5th ed., Chapter 77
- Zijlstra EE et al. Mycetoma. Lancet Infect Dis. 2016;16:100
- Van de Sande WW. Global burden of human mycetoma. PLoS Negl Trop Dis. 2013;7:e2550
This is a shared conversation. Sign in to Orris to start your own chat.