Using millers anaesthesiology book discuss CRPS - II
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Searching PubMed
"complex regional pain syndrome" type II causalgia
I now have all the content needed for a comprehensive answer. Let me synthesize everything from Miller's Anesthesia and the supplemental sources.
Complex Regional Pain Syndrome Type II (CRPS-II)
Miller's Anesthesia, 10th Edition — with supplemental coverage from Bradley and Daroff's Neurology in Clinical Practice
1. Nomenclature and Historical Background
The term causalgia was first coined by Weir Mitchell in the 1870s to describe severe, progressive, distal limb pain following major nerve injury. In 1946, Evans introduced the term "reflex sympathetic dystrophy" (RSD), later defined by the IASP as continuous pain in part of an extremity after trauma, associated with sympathetic hyperactivity, but without a definable major nerve lesion.
In 1994, the IASP replaced this nomenclature with Complex Regional Pain Syndrome (CRPS):
| Type | Former Name | Key Distinction |
|---|---|---|
| CRPS-I | Reflex Sympathetic Dystrophy (RSD) | No definable nerve lesion |
| CRPS-II | Causalgia | Definable peripheral nerve lesion is present |
CRPS is classified under neuropathic pain in Miller's broader taxonomy of chronic non-cancer pain (CNCP), which includes postherpetic neuralgia, phantom pain, diabetic neuropathy, and HIV-associated neuropathy. — Miller's Anesthesia, 10th ed., p. 6835
2. Epidemiology
- Mean age of CRPS patients: 36–46 years
- Female predominance: 60–81% of cases
- Common precipitating injuries in CRPS overall:
- Fracture: 16–46%
- Strain/sprain: 10–29%
- Post-surgical: 3–24%
- Contusion/crush injury: 8–18%
- In CRPS-II specifically, a major nerve injury is the defining precipitant (e.g., median nerve, sciatic nerve)
— Bradley and Daroff's Neurology in Clinical Practice; Miller's Anesthesia, 10th ed.
3. Pathophysiology
The pathophysiology remains incompletely understood, but several mechanisms are implicated:
a) Sympathetic Nervous System Dysfunction
- An abnormal reflex arc following the sympathetic nervous system, modulated by cortical centres, has been proposed
- Decreased sympathetic outflow to the affected limb is now understood to be the underlying mechanism — autonomic features previously ascribed to sympathetic overactivity are now thought to result from catecholamine hypersensitivity at peripheral receptors
b) Central Sensitization
- In Miller's framework of chronic pain neurophysiology, central sensitization — amplification of nociceptive signals in the dorsal horn — plays a critical role in maintaining and spreading CRPS pain beyond the original nerve distribution
- NMDA receptor upregulation, disinhibition, and cortical reorganization perpetuate pain
c) Peripheral Nerve Injury (Specific to CRPS-II)
- Unlike CRPS-I, CRPS-II requires a definable nerve lesion
- Axonal injury triggers ectopic discharge, upregulation of sodium channels, and abnormal sympatho-afferent coupling — injured afferents develop adrenoceptor sensitivity, so circulating catecholamines directly excite nociceptors
d) Psychosocial Factors
- Significant emotional disturbance at onset is present in many patients
- Stress may be a precipitating factor
- Miller's biopsychosocial model of chronic pain applies: biological injury, psychological (catastrophizing, fear-avoidance), and social reinforcement all perpetuate the syndrome — Miller's Anesthesia, 10th ed., p. 6835–6836
4. Clinical Features
Pain is the cardinal feature — described as burning, aching, pricking, or shooting, typically disproportionate to the initiating injury and extending beyond the nerve territory.
Pain Characteristics
- Hyperalgesia: exaggerated pain response to a normally painful stimulus (e.g., pin prick)
- Mechanical allodynia: light touch perceived as severe, long-lasting pain
- Thermal allodynia: cooling stimuli (e.g., drop of alcohol) perceived as painful
- Pain severity is not proportional to the initial injury
Trophic and Autonomic Changes
Skin color/temperature:
- The affected area may be reddish, then turn blue, purple, or pale within minutes to hours
- Livedo reticularis (non-blanchable reticulated skin pattern) is common
- Temperature asymmetry between affected and unaffected sides may exceed 1°C
Sudomotor:
- ~60% of patients report excessive sweating in the affected limb
Edema:
- Present in the majority; ranges from mild cellulitis-like swelling in early stages to severe edema requiring Doppler to exclude DVT in late stages
Motor dysfunction:
- Not required for diagnosis, but weakness, tremor, or dystonia may be present
Trophic changes (late):
- Waxy, shiny skin; brittle, ridged nails; muscle wasting; joint contractures
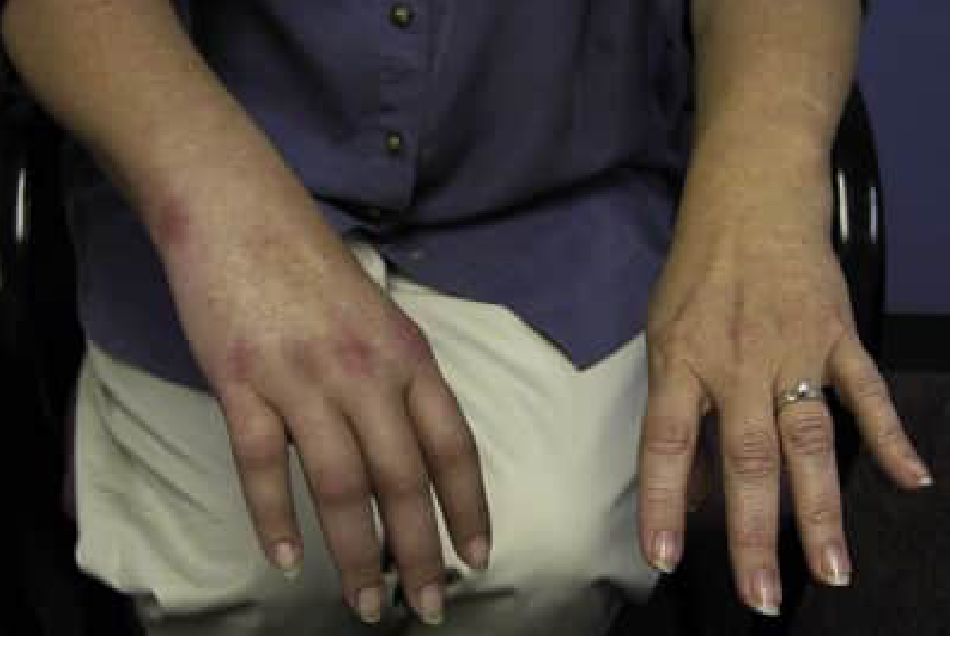
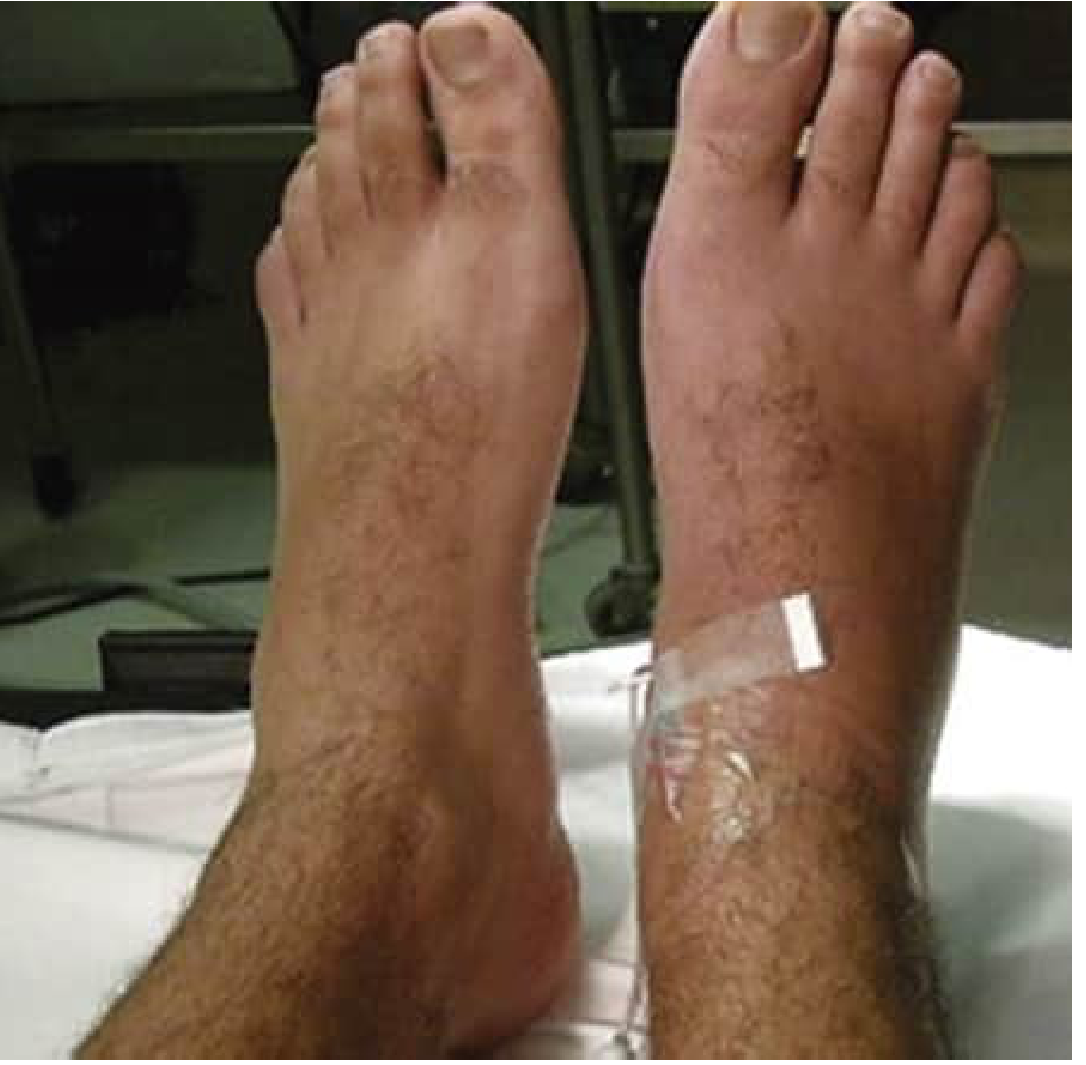
5. Clinical Staging
Three stages of progression have been described (Schwartzman and Maleki):
| Stage | Features | Duration |
|---|---|---|
| Stage I (Early) | Burning/throbbing pain, allodynia, localized edema, increased skin temperature, vasomotor changes (color, temperature) | Weeks to months |
| Stage II | Progressive soft tissue edema, skin thickening, articular soft tissue changes, muscle wasting | 3–6 months |
| Stage III (Late) | Joint contractures, frozen shoulder, digital contractures, waxy trophic skin, brittle nails, severe radiographic demineralization | Ongoing |
- CRPS can spread from one limb to others and, in severe cases, involve all four extremities with severe pain, edema, cold/cyanotic limbs, contractures, and atrophy
6. Diagnosis
Diagnosis is essentially clinical. Key features:
- Diffuse, severe, nonsegmental pain
- Cyanosis or mottling, increased sweating, shiny skin
- Swollen non-articular tissue
- Coldness to touch
- History of a definable nerve injury (differentiating CRPS-II from CRPS-I)
Investigations
- Autonomic testing: Resting sweat output + quantitative sudomotor axon reflex test (QSART) together are 94% sensitive and 98% specific — excellent predictors of response to sympathetic block
- Bone scintigraphy: Most sensitive in Stage I; less useful in later stages
- Plain radiographs: Show severe bone demineralization in Stage III
- Sympathetic block: Serves both a diagnostic and therapeutic role — a positive response to stellate ganglion or lumbar sympathetic block supports the diagnosis
7. Management — Anesthetic Perspective
Miller's Anesthesia places CRPS management firmly within the framework of multimodal, interdisciplinary chronic pain management.
a) Pharmacological Treatment
| Drug Class | Role |
|---|---|
| Antineuropathics (gabapentin, pregabalin) | First-line for neuropathic pain; also used perioperatively |
| TCAs (amitriptyline) | Neuropathic pain and comorbid depression |
| α₂-agonists (clonidine) | Analgesia; perioperative adjunct |
| Bisphosphonates | Prevent bone resorption; also analgesic |
| Prazosin, propranolol, nifedipine/verapamil, guanethidine, phenoxybenzamine | Occasionally effective via sympatholytic/adrenoceptor mechanisms |
| Ketamine (S-ketamine) | NMDA antagonism at subanesthetic doses — provides effective and long-term pain relief in CRPS-I patients; also used in CRPS-II |
b) Interventional/Regional Anesthetic Techniques
Regional anesthesia occupies a central role in CRPS management in Miller's. — Miller's Anesthesia, 10th ed., p. 10961
Stellate Ganglion Block
- Indicated for upper extremity CRPS (including CRPS-II)
- Sympathetically maintained pain may respond well — "sympathetically maintained pain syndrome" is a key indication
- Early stellate ganglion blocks may significantly decrease pain and hasten clinical recovery
- May also prevent recurrence of CRPS after reoperation of the affected extremity
- A Horner syndrome confirms correct placement; ultrasound guidance is preferred
Lumbar Sympathetic Block
- For lower extremity CRPS
Intravenous Regional Block (IVRB / Bier Block)
- IVRB with bretylium provided significantly longer analgesia than lidocaine in a double-blind study
Epidural Techniques
- Epidural clonidine and ketamine — good pain relief reported
- Epidural anesthesia facilitates physiotherapy and functional rehabilitation
Continuous Peripheral Nerve Blocks
- Perineural catheters reduce pain, facilitate physiotherapy, and are used for functional rehabilitation
- Reported effective in children with CRPS as well — Miller's Anesthesia, 10th ed., p. 10961
Intrathecal Drug Delivery
- Intrathecal baclofen and morphine — good pain relief reported in refractory cases
c) Spinal Cord Stimulation (SCS)
- SCS has gained interest; unblinded studies suggest selected CRPS patients may benefit
- An early study with 2-year follow-up reported long-term pain reduction and improved health-related quality of life with SCS
- However, a more recent RCT (Kemler et al., 2008) with 5-year follow-up found no extra benefit for SCS + physical therapy vs. physical therapy alone in terms of pain relief
- Miller's notes: SCS "may have difficulty stopping the spread of CRPS, and once CRPS spreads out of the area initially covered, pain is no longer under control" — Miller's Anesthesia, 10th ed., p. 6867
d) Surgical Sympathectomy
- Used for progressive disease in patients who have previously responded to sympathetic block
- Not first-line; evidence is limited
e) Psychological and Rehabilitative Approaches
- Miller's emphasizes the biopsychosocial model — monomodal therapy is insufficient
- Multimodal interdisciplinary programs simultaneously address physical, psychological, and social factors
- Functional rehabilitation is the primary goal of all interventions
- TENS and acupuncture may provide adjunctive benefit
8. CRPS-II vs. CRPS-I — Key Differences
| Feature | CRPS-I | CRPS-II |
|---|---|---|
| Former name | Reflex Sympathetic Dystrophy | Causalgia |
| Nerve lesion | None identifiable | Definable major nerve injury |
| Typical triggers | Fracture, soft tissue injury, MI | High-velocity injury, penetrating trauma, nerve surgery |
| Pain distribution | Nonsegmental, regional | May initially follow nerve territory, then spread |
| Sympathetically maintained pain | Common | Common (but also independent mechanisms) |
| Treatment | Same multimodal approach | Same + attention to the nerve injury itself |
9. Perioperative Considerations for CRPS Patients (Miller's)
When a CRPS patient requires surgery, Miller's offers specific guidance:
- Central sensitization may cause increased and prolonged postoperative pain
- Higher analgesic requirements expected — titrate opioids starting at 2–4× the usual dose for opioid-naïve patients
- Maintain preoperative medications perioperatively (gabapentinoids, antidepressants, etc.) — abrupt discontinuation risks rebound hyperalgesia or seizures
- Regional anesthesia preferred where possible — prevents triggering/worsening CRPS
- Ketamine as adjunct: NMDA antagonism reduces central sensitization and opioid requirements
- Monitor for withdrawal in patients on chronic opioids: unexplained tachycardia, restlessness, sweating, hypertension
- Pain service consultation is recommended at discharge — Miller's Anesthesia, 10th ed., pp. 6867–6884
Summary
CRPS-II (causalgia) is a neuropathic pain syndrome defined by the presence of a definable peripheral nerve lesion, distinguishing it from CRPS-I. It presents with disproportionate regional pain, allodynia, hyperalgesia, autonomic dysfunction, edema, and trophic changes that evolve through three clinical stages. Pathophysiology involves peripheral nerve injury–driven ectopic discharge, catecholamine hypersensitivity, and central sensitization. Management is multimodal — pharmacological (antineuropathics, ketamine, bisphosphonates), interventional (stellate/lumbar sympathetic block, epidural analgesia, IVRB, intrathecal therapy), neuromodulatory (SCS), and rehabilitative/psychological. From the anesthesiologist's perspective, regional anesthetic techniques play a central therapeutic and preventive role, and perioperative management of these patients demands careful continuation of existing analgesic regimens and aggressive multimodal analgesia.
Primary source: Miller's Anesthesia, 2-Volume Set, 10th Edition (ISBN 9780323935920), pp. 6835–6867, 10961
Supplemental: Bradley and Daroff's Neurology in Clinical Practice (ISBN 9780323642613)
Using millers anaesthesiology book discuss stellate ganglion block, gasserian ganglion block, intercostal block and celiac plexus block
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is the complete discussion of all four blocks as covered in Miller's Anesthesia 10e (with cross-reference to Barash for the Gasserian ganglion technique, which Miller's itself acknowledges is more a chronic pain subspecialty procedure):
Autonomic and Peripheral Nerve Blocks
Miller's Anesthesia, 2-Volume Set, 10th Edition
Overarching Framework
Miller's situates all nerve block techniques within its biopsychosocial model of pain management. Contemporary practice reflects a more judicial approach:
"Nerve block therapy alone is usually not curative, but it can facilitate participation in rehabilitation and therefore may have a role in the management of chronic pain. Regardless which procedure is considered, a consensus decision on its use must be reached within the interdisciplinary team." — Miller's Anesthesia, 10e, p. 6864
1. Cervicothoracic (Stellate) Ganglion Block
Anatomy
The stellate ganglion is formed by fusion of the inferior cervical and first thoracic sympathetic ganglia at the level of C7–T1. It lies:
- Anterolateral to the C7–T1 vertebral bodies
- Medial to the carotid sheath
- Anterior to the transverse processes and longus colli muscle
- Posterolateral to the trachea and esophagus
Technique
- Patient supine with neck slightly extended
- Longus colli muscle identified ultrasonographically — the key sonographic landmark
- Local anesthetic injected superior to the longus colli muscle
- Ultrasound guidance is strongly recommended — this is described as the contemporary standard in Miller's
- Horner syndrome (ptosis, miosis, anhidrosis, enophthalmos) = confirmation of correct sympathetic block
Indications (per Miller's 10e)
| Indication | Notes |
|---|---|
| Ventricular tachyarrhythmia (congenital long-QT syndrome) | Left stellate block specifically recommended |
| Severe ipsilateral circulatory disorders of the upper extremity | |
| Herpes zoster ophthalmicus | Acute pain syndrome |
| CRPS / Sympathetically maintained pain syndrome | Early stellate blocks may significantly decrease pain and hasten recovery; may prevent CRPS recurrence after reoperation |
| Phantom limb pain | Facilitates physiotherapy and functional rehabilitation |
"Stellate ganglion block is a rather dangerous procedure with very few but very specific indications." — Miller's Anesthesia, 10e, Ch. 74 (p. 11047)
Complications
- Intravascular injection (vertebral artery, carotid artery) → seizures, cardiovascular collapse
- Pneumothorax (apex of lung is adjacent)
- Phrenic nerve block → unilateral diaphragm paralysis (bilateral stellate block is absolutely contraindicated)
- Recurrent laryngeal nerve block → hoarseness, dysphagia
- Brachial plexus block (unintended spread)
- Epidural or intrathecal injection with spinal spread
- Horner syndrome per se is an expected endpoint, not a complication
2. Semilunar (Gasserian) Ganglion Block
Miller's 10e references the Gasserian ganglion anatomically in the context of the oculocardiac reflex (Ch. 58) and cites percutaneous gasserian procedures (microcompression, balloon compression) in its neurosurgical references. The technique itself is acknowledged as a chronic pain subspecialty procedure, cross-referenced to dedicated regional anesthesia texts.
Anatomy
The Gasserian (semilunar) ganglion is the sensory ganglion of CN V (trigeminal nerve), located in Meckel's cave — a dural recess at the apex of the petrous temporal bone. It gives rise to three divisions:
| Division | Exit | Territory |
|---|---|---|
| V₁ (ophthalmic) | Superior orbital fissure | Forehead, eye, upper nose |
| V₂ (maxillary) | Foramen rotundum | Midface, upper teeth |
| V₃ (mandibular) | Foramen ovale | Lower face, jaw, anterior tongue |
Needle access to the ganglion is via foramen ovale (V₃ root).
Miller's Context — Oculocardiac Reflex
"The pain, pressure or traction impulses are conducted by the ciliary nerves to the ciliary ganglion and then via the ophthalmic division of the trigeminal nerve to the Gasserian ganglion and the sensory nucleus of the trigeminal nerve." — Miller's Anesthesia, 10e, p. 4063
This forms the afferent limb of the oculocardiac reflex: ocular traction → Gasserian ganglion → trigeminal sensory nucleus → Edinger-Westphal nucleus → vagal efferents → bradycardia/asystole.
Indications
- Disabling trigeminal neuralgia (tic douloureux) — primary indication
- Head and neck cancer pain (neurolytic)
- When medical management (carbamazepine) has failed
Technique (Fluoroscopy-Guided — Hartel Approach)
"The most comprehensive block of the trigeminal nerve targets the central ganglion. This block is usually performed under fluoroscopic guidance to treat disabling trigeminal neuralgia. Few operating room anesthesiologists perform this technically challenging block." — Barash, Cullen and Stoelting, 9e, Ch. 36 (cross-referenced by Miller's)
- Patient supine, mouth open
- Skin entry point: lateral to the corner of the mouth, 2.5–3 cm
- 20-gauge spinal needle directed toward the foramen ovale under AP and lateral fluoroscopy
- Traverses infratemporal fossa → enters Meckel's cave through foramen ovale
- Entry confirmed by CSF flow from the dural sleeve surrounding the ganglion
- Inject local anesthetic / glycerol / or apply RF energy
Neurolytic Options at the Gasserian Ganglion (cited in Miller's)
| Technique | Method |
|---|---|
| Percutaneous balloon microcompression | Mullan technique; balloon inflated at ganglion |
| Glycerol rhizolysis | Hakansson technique; preserves touch sensation better |
| Radiofrequency thermocoagulation | Most widely used; selective V₂/V₃ lesioning possible |
Miller's 10e specifically cites Fraioli et al. (percutaneous microcompression, J Neurosurg 1989) and Skirving & Dan's 20-year review of balloon compression (J Neurosurg 2001). — Miller's Anesthesia, 10e, p. 4231
Complications
- Corneal anesthesia → keratitis, corneal ulceration (most feared; V₁ injury)
- Masseter weakness (motor root at V₃)
- Anesthesia dolorosa — painful deafferentation state, more common with neurolytic agents
- Intracranial hemorrhage
- Carotid artery puncture
- CSF leak / meningitis
- Ipsilateral Horner syndrome (sympathetic fibres near ganglion)
3. Intercostal Nerve Block
Anatomy
Miller's provides a precise triangular space description:
"Intercostal nerves run along the lower border of each rib, within the intercostal space, a triangular space, with: (1) a medial border formed by the posterior intercostal and innermost intercostal muscles, endothoracic fascia, and parietal pleura; (2) a lateral border formed by the internal and external intercostal muscles and intercostal membrane; (3) a base formed by the lower rib." — Miller's Anesthesia, 10e, p. 11085
Within the costal groove, the neurovascular bundle runs in the order (superior → inferior): Vein – Artery – Nerve (VAN).
Technique
- Position: semi-prone at the midaxillary line — described as the safest approach in Miller's
- Needle: 22- or 20-gauge Tuohy needle (intradermal needles are explicitly stated as inappropriate)
- Guidance: In-plane ultrasound strongly preferred
Two-step needle manoeuvre:
- Insert needle in-plane to contact rib
- Redirect needle and walk off the rib caudally into the intercostal space
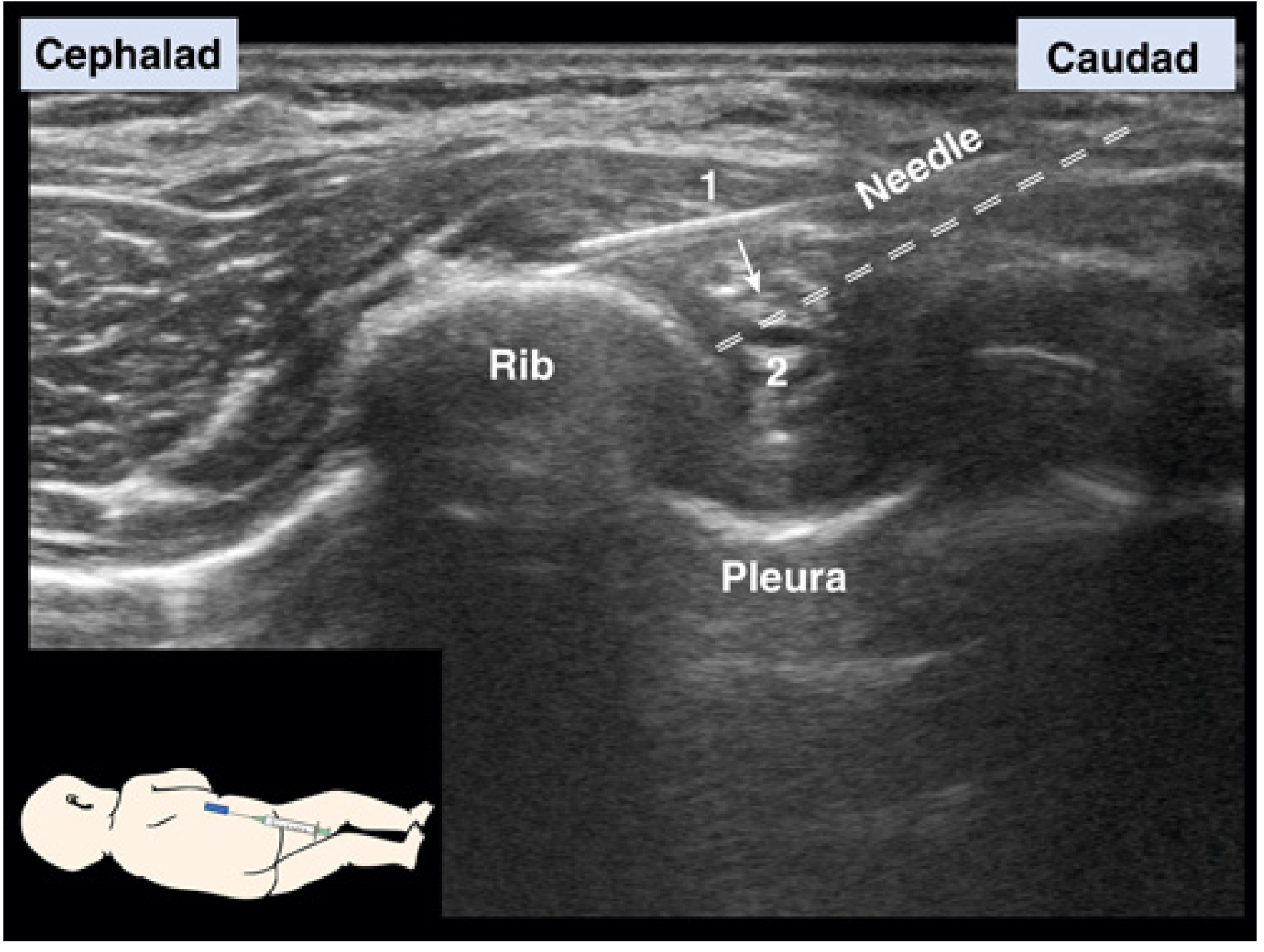
Catheter Technique
- A catheter may be placed for repeated reinjections
- Can also be inserted intraoperatively under direct surgical vision
- Large-volume injection can spread to distant intercostal spaces via the paravertebral space (even contralateral), and to the epidural space
"The safety of continuous techniques is questionable because of the high systemic uptake of local anesthetic." — Miller's Anesthesia, 10e, p. 11086
Indications
| Indication |
|---|
| Rib fractures (multiple) — pain management and respiratory splinting |
| Thoracotomy (multiple adjacent spaces) |
| Upper abdominal surgery (hepatobiliary) |
| Liver transplantation |
| Pleural drainage / chest tube insertion |
| Intraoperative and postoperative thoracic analgesia |
Safety Requirements — Explicit in Miller's
- Contraindicated in patients with impaired oxygenation or gas exchange
- Not suitable for outpatient surgery
- All patients must be kept under intensive medical observation — risk of clinically delayed pneumothorax
- Patients with catheters or large-volume injections should be admitted to ICU for monitoring of respiratory function and delayed pneumothorax
Complications
| Complication | Notes |
|---|---|
| Pneumothorax | Most feared; may be clinically delayed — all patients need monitoring |
| Systemic local anesthetic toxicity | Intercostal space has the highest absorption rate of any regional block site due to rich vascularity |
| Epidural/spinal spread | Via paravertebral space |
| Hemothorax | Intercostal vessel injury |
| Bilateral respiratory depression | With bilateral blocks |
4. Celiac Plexus Block
Anatomy
The celiac plexus is the largest prevertebral autonomic plexus, lying retroperitoneally at T12–L1 surrounding the celiac artery origin. It receives:
- Greater splanchnic nerves (T5–T9) — preganglionic sympathetic
- Lesser splanchnic nerves (T10–T11)
- Least splanchnic nerve (T12)
- Parasympathetic fibres via the vagus nerve
- Visceral afferent pain fibres from upper abdominal viscera
Miller's specifically describes how renal sympathetic innervation converges here:
"Sympathetic nerves to the kidney originate as preganglionic fibers from the eighth thoracic through the first lumbar segments and converge at the celiac plexus and aorticorenal ganglia. Postganglionic fibers to the kidney arise mainly from the celiac..." — Miller's Anesthesia, 10e, p. 984
Visceral Pain Coverage
| Organ | Covered? |
|---|---|
| Pancreas | ✓ (most clinically important) |
| Liver, gallbladder, bile ducts | ✓ |
| Stomach, duodenum | ✓ |
| Small intestine to mid-transverse colon | ✓ |
| Kidneys, adrenal glands, spleen | ✓ |
| Pelvic organs | ✗ (inferior hypogastric plexus) |
Indications
| Indication | Evidence |
|---|---|
| Pancreatic cancer pain | Strongest evidence — Cochrane SR cited in Miller's 10e |
| Upper abdominal malignancy pain | Significant opioid-sparing effect |
| Chronic pancreatitis pain (benign) | Benefit less durable |
| Post-liver transplant pain (children) | Miller's cites regional techniques including celiac block |
Miller's cites the Cochrane Database Systematic Review on "Celiac plexus block for pancreatic cancer pain in adults" (2011) as the key evidence base. — Miller's Anesthesia, 10e, p. 6866
Techniques
Posterior Retrocrural Approach (Classic):
- Patient prone
- Bilateral 20-gauge needles (15–20 cm) inserted at L1 level, angled toward the anterior surface of the L1 vertebral body
- Fluoroscopic or CT guidance; contrast injection to confirm pre-aortic spread
- Drug deposited anterior to the aorta / around the celiac axis
Anterior Transabdominal Approach:
- Patient supine
- CT or ultrasound guided
- Single needle, transgastric or transhepatic path
- Preferred in patients who cannot lie prone or when posterior anatomy is distorted
Intraoperative (Surgical) Approach:
- Direct injection at celiac axis at time of laparotomy or laparoscopy
- Increasingly used during pancreatic cancer resection
Drug Selection
| Purpose | Agent | Volume |
|---|---|---|
| Diagnostic / therapeutic | Bupivacaine 0.25–0.5% | 20–25 mL per side (bilateral) |
| Neurolysis (cancer pain) | Absolute alcohol (50–100%) | 20–25 mL per side |
| Alternative neurolysis | 6% phenol | 10–15 mL per side |
Protocol: Always perform a diagnostic local anesthetic block first to confirm adequate pain relief before proceeding with neurolysis.
Efficacy
- Pancreatic cancer: superior to systemic opioids alone; reduces opioid consumption and side effects; pain relief lasting weeks to months
- Miller's acknowledges celiac plexus block as one of the few interventional pain techniques with meaningful cancer pain evidence — p. 6866
Complications
| Complication | Notes |
|---|---|
| Orthostatic hypotension | Most common (sympatholysis → splanchnic vasodilation → venous pooling); treat with IV fluids |
| Diarrhea | Parasympathetic predominance; usually transient |
| Paraplegia | Rare (0.15%) but catastrophic — injection into or spasm of artery of Adamkiewicz (anterior spinal artery supply) |
| Retroperitoneal hematoma | Posterior approach; vessel injury |
| Pneumothorax | Posterior approach |
| Renal puncture | Posterior approach |
| Aortic/IVC puncture | |
| Retroperitoneal infection/abscess | |
| Intravascular injection of neurolytic agent |
Comparative Overview
| Feature | Stellate Ganglion | Gasserian Ganglion | Intercostal | Celiac Plexus |
|---|---|---|---|---|
| System | Sympathetic | CN V sensory | Somatic | Sympathetic (visceral) |
| Level | C7-T1 | Base of skull | Any rib | T12-L1 |
| Guidance | Ultrasound (preferred) | Fluoroscopy (mandatory) | Ultrasound (preferred) | CT/fluoro/US |
| Success marker | Horner syndrome | Facial numbness V1–V3 | Dermatomal anesthesia | Pain relief + diarrhea |
| Key indication | CRPS, long-QT arrhythmia | Trigeminal neuralgia | Rib fractures, thoracotomy | Pancreatic cancer pain |
| Most feared complication | Intravascular injection / bilateral block | Corneal anesthesia / anesthesia dolorosa | Pneumothorax / LA toxicity | Paraplegia / hypotension |
| Neurolytic option | Rarely (surgical sympathectomy) | Yes (alcohol, glycerol, RF) | Not standard | Yes (alcohol/phenol, cancer) |
| Outpatient appropriate? | With caution | No | No | No |
Primary source: Miller's Anesthesia, 2-Volume Set, 10th Edition (ISBN 9780323935920), pp. 984, 2379, 4063, 4231, 6864–6866, 10961, 11085–11086
Supplemental (Gasserian technique): Barash, Cullen and Stoelting's Clinical Anesthesia, 9e (ISBN 9781975199074), p. 2931
Using OH's ICU book discuss in detail about shock, Hemorrhagic shock, septic shock, cardiogenic shock and role of inotropes and vasopressors in management of shock.
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
This is a shared conversation. Sign in to Orris to start your own chat.