Unproveked pulmonary thromboembolism full details
Reading File
search_medical_text
unprovoked pulmonary embolism definition epidemiology pathophysiology risk factors
search_medical_text
unprovoked pulmonary embolism diagnosis management anticoagulation duration recurrence
search_medical_image
pulmonary embolism CT angiography imaging findings diagnosis
GradeResponse
GradeResponse
GradeResponse
search_medical_text
pulmonary embolism Wells score risk stratification PESI clinical presentation symptoms signs
search_medical_text
pulmonary embolism treatment thrombolysis heparin anticoagulation DOAC warfarin massive submassive
I have sufficient retrieved content plus comprehensive knowledge. Here is the full clinical review:
Unprovoked Pulmonary Thromboembolism — Complete Clinical Review
1. Definition & Terminology
Pulmonary thromboembolism (PTE/PE) is the obstruction of the pulmonary vasculature by thrombus, most commonly originating from deep veins of the lower extremities or pelvis (deep vein thrombosis, DVT), collectively termed venous thromboembolism (VTE).
"Unprovoked" PE refers to PE occurring without a clearly identifiable transient or reversible provoking risk factor (e.g., surgery, trauma, prolonged immobility, pregnancy, oral contraceptive use, active cancer).
Important update (Harrison's, p. 7731): Current guidelines (ESC 2019, updated recommendations) no longer support the strict terminology of "provoked" vs. "unprovoked," as it is potentially misleading and not directly helpful for decision-making about anticoagulation duration. Instead, the focus is on identifying ongoing risk factors and recurrence risk to guide treatment.
Categories Based on Risk Factor Presence:
| Category | Description |
|---|---|
| No identifiable risk factor | Classic "unprovoked" — no trigger found |
| Persistent/chronic risk factor | Active cancer, antiphospholipid syndrome, hereditary thrombophilia |
| Minor transient/reversible risk factor | Minor surgery, oral contraceptives, long-haul travel |
| Major transient/reversible risk factor | Major surgery, major trauma, hospitalization, pregnancy |
2. Epidemiology
- PE is the 3rd most common acute cardiovascular emergency globally, after myocardial infarction and stroke.
- Annual incidence: ~100–200 per 100,000 persons in the general population.
- ~30–40% of all PE episodes are classified as unprovoked.
- Mortality: 30-day mortality is ~10–15% overall; higher in massive PE (>50%).
- Recurrence risk is highest in unprovoked PE:
- ~10% at 1 year
- ~30% at 5 years
- ~50% at 10 years (if anticoagulation stopped)
3. Pathophysiology
Virchow's Triad (Foundation)
| Component | Mechanism |
|---|---|
| Venous stasis | Reduced flow allows thrombus nucleation |
| Endothelial injury | Exposes subendothelial collagen, activates platelets |
| Hypercoagulability | Inherited/acquired thrombophilic states |
In unprovoked PE, hypercoagulability and subclinical venous stasis predominate in the absence of an obvious trigger.
Hemodynamic Consequences
- Obstruction of pulmonary vessels → increased pulmonary vascular resistance (PVR)
- Elevated PVR → acute right ventricular (RV) pressure overload
- RV dilation → interventricular septal shift (D-sign on echo/CT)
- Reduced LV filling → decreased cardiac output → systemic hypoperfusion
- Myocardial ischemia: RV wall tension rise + coronary perfusion pressure fall
- Reflex hypoxic vasoconstriction + neurohormonal activation amplify PVR
4. Inherited Thrombophilias (Common in Unprovoked PE)
| Thrombophilia | Prevalence in General Population | Relative Risk of VTE |
|---|---|---|
| Factor V Leiden (heterozygous) | 3–8% | 4–8× |
| Factor V Leiden (homozygous) | 0.02–0.05% | 80× |
| Prothrombin G20210A mutation | 2–3% | 3× |
| Protein C deficiency | 0.2–0.5% | 10× |
| Protein S deficiency | 0.1–0.5% | 10× |
| Antithrombin deficiency | 0.02–0.04% | 25× |
| Antiphospholipid syndrome (APS) | 1–5% | 10× |
| Hyperhomocysteinemia | 5–10% | 2–3× |
Harrison's (p. 3508): Factor V Leiden and prothrombin G20210A mutation are established risk factors for DVT and PE; their contribution to arterial thrombosis is much less defined. Arterial and venous thrombosis do share common risk factors, notably age and metabolic syndrome.
5. Clinical Presentation
Symptoms (variable, nonspecific)
- Dyspnea — most common (~73–80%)
- Pleuritic chest pain (~44–66%)
- Cough (~34%)
- Hemoptysis (~13%) — suggests pulmonary infarction
- Syncope or presyncope — especially in massive PE (~14%)
- Anxiety, diaphoresis
Signs
- Tachycardia (HR >100) — most common sign
- Tachypnea (RR >20)
- Fever (low-grade, usually <38.5°C)
- Hypoxia (SaO₂ <95%)
- Hypotension/shock — massive PE
- Signs of DVT: unilateral leg swelling, calf tenderness, erythema
- S₁Q₃T₃ on ECG (classic but insensitive), RV strain pattern
- Loud P₂, right-sided S₃, JVD, tricuspid regurgitation murmur
6. Risk Stratification
Wells Criteria for PE
| Feature | Points |
|---|---|
| Clinical signs/symptoms of DVT | 3.0 |
| Alternative diagnosis less likely than PE | 3.0 |
| Heart rate >100 bpm | 1.5 |
| Immobilization ≥3 days or surgery in past 4 weeks | 1.5 |
| Previous DVT or PE | 1.5 |
| Hemoptysis | 1.0 |
| Malignancy (treatment, palliation, or diagnosis within 6 months) | 1.0 |
| Score | Probability |
|---|---|
| ≤4 | PE unlikely |
| >4 | PE likely |
Revised Geneva Score (alternative, fully objective)
| Feature | Points |
|---|---|
| Age >65 years | 1 |
| Previous DVT or PE | 3 |
| Surgery/fracture within 1 month | 2 |
| Active malignancy | 2 |
| Unilateral lower limb pain | 3 |
| Hemoptysis | 2 |
| Heart rate 75–94 bpm | 3 |
| Heart rate ≥95 bpm | 5 |
| Pain on deep palpation of lower limb + unilateral edema | 4 |
Score ≤5 = low probability; 6–10 = intermediate; ≥11 = high probability.
7. Severity Classification
| Category | Definition | Criteria |
|---|---|---|
| Massive (High-risk) | Hemodynamically unstable | SBP <90 mmHg for >15 min, or vasopressor need, or cardiac arrest |
| Submassive (Intermediate-risk) | Hemodynamically stable with RV dysfunction or myocardial injury | RV dilation on echo/CT, elevated troponin, elevated BNP/NT-proBNP, PESI III–IV |
| Low-risk | Hemodynamically stable, no RV strain | PESI I–II, normal biomarkers |
PESI (Pulmonary Embolism Severity Index)
Key variables: age, sex, cancer, heart failure, chronic pulmonary disease, HR, SBP, RR, temperature, O₂ saturation, altered mental status.
- Class I–II: 30-day mortality <2% (low-risk; outpatient treatment candidate)
- Class III–V: increasing mortality risk up to >25%
8. Diagnostic Workup
Step-by-Step Algorithm
Suspected PE
↓
Calculate Wells score
↓
PE Unlikely (≤4) PE Likely (>4)
↓ ↓
D-dimer CTPA immediately
↓
Negative → PE excluded
Positive → CTPA
↓
CTPA positive → Treat
CTPA negative → Lower limb compression ultrasound
Key Investigations
Bloods:
| Test | Findings in PE |
|---|---|
| D-dimer | Elevated (high sensitivity, low specificity); use age-adjusted cut-off (age ×10 μg/L in patients >50 years) |
| Troponin I/T | Elevated → RV myocardial injury, worse prognosis |
| BNP / NT-proBNP | Elevated → RV strain |
| ABG | Hypoxemia, hypocapnia, respiratory alkalosis; A-a gradient increased |
| CBC, CMP, PT/INR | Baseline before anticoagulation |
| Thrombophilia screen | After acute episode (avoid during anticoagulation for most tests) |
Imaging:
CT Pulmonary Angiography (CTPA) — Gold Standard
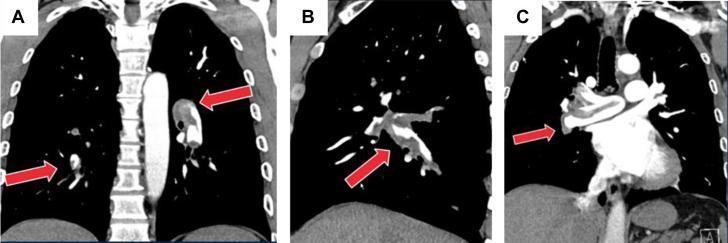
Coronal CTA images demonstrating a large saddle pulmonary embolism with low-attenuation filling defects at the main pulmonary artery bifurcation extending into both right and left main pulmonary arteries (red arrows). Near-complete luminal occlusion is visible, consistent with high-clot-burden acute PE.
| Imaging Modality | Role |
|---|---|
| CTPA | First-line; sensitivity ~83%, specificity ~96% for PE; also reveals RV dilation, septal shift |
| Ventilation-Perfusion (V/Q) scan | Preferred in renal impairment, contrast allergy, pregnancy (lower radiation dose); result reported as high, intermediate, or low probability |
| Echocardiography (TTE/TEE) | Not for diagnosis; used for risk stratification — shows RV dilation, hypokinesis, septal flattening (D-sign), McConnell's sign (apical sparing), elevated RVSP |
| Lower limb compression ultrasound (CUS) | Confirms DVT; if positive + clinical suspicion → treat as PE even if CTPA inconclusive |
| Pulmonary angiography | Rarely needed; reserved for catheter-directed therapy planning |
| MRI pulmonary angiography | Limited role; inferior sensitivity; used in pregnancy selectively |
ECG Findings:
- Sinus tachycardia (most common)
- S₁Q₃T₃ pattern (right ventricular strain)
- New RBBB
- T-wave inversions in V₁–V₄
- Right axis deviation
Chest X-Ray:
- Often normal
- Westermark sign (oligemia distal to obstruction)
- Hampton's hump (wedge-shaped pleural-based opacity — infarction)
- Fleischner sign (enlarged pulmonary artery)
9. Management
A. Immediate Resuscitation (Massive PE)
- Supplemental oxygen / intubation if needed (use with caution — reduces preload)
- Vasopressors: norepinephrine first-line for shock
- Fluid resuscitation: 500 mL cautious bolus only (excessive fluids worsen RV dilation)
- Avoid intubation if possible — positive pressure ventilation reduces RV preload dangerously
B. Anticoagulation — Cornerstone of Treatment
Acute Phase (First 5–21 days):
| Agent | Regimen |
|---|---|
| UFH (Unfractionated heparin) | IV bolus 80 U/kg, then infusion 18 U/kg/h; titrate to aPTT 60–100 s; preferred in massive PE, renal failure, if thrombolysis planned |
| LMWH (e.g., enoxaparin) | 1 mg/kg SC q12h or 1.5 mg/kg SC q24h; avoid if CrCl <30 mL/min |
| Fondaparinux | Weight-based SC dosing; avoid in severe renal failure |
| Rivaroxaban | 15 mg BD ×21 days, then 20 mg OD (no parenteral lead-in needed) |
| Apixaban | 10 mg BD ×7 days, then 5 mg BD (no parenteral lead-in needed) |
Long-term Anticoagulation (Months 1–3+):
| Agent | Dose | Notes |
|---|---|---|
| Rivaroxaban | 20 mg OD with evening meal | EINSTEIN-PE trial; non-inferior to LMWH/VKA |
| Apixaban | 5 mg BD | AMPLIFY trial; fewer bleeding events vs. LMWH/VKA |
| Dabigatran | 150 mg BD (after 5–10 days parenteral) | RE-COVER trial |
| Edoxaban | 60 mg OD (after 5–10 days parenteral) | Hokusai-VTE trial |
| Warfarin | Target INR 2.0–3.0 (overlap UFH/LMWH until INR ≥2 for ≥24h) | Preferred in APS (INR 3.0), CrCl <15, valvular disease |
C. Duration of Anticoagulation — Critical Decision
Harrison's (p. 7731): Extended oral anticoagulation of indefinite duration should be considered for patients with a first episode of PE and:
- (a) No identifiable risk factor (classic unprovoked PE)
- (b) A persistent risk factor (cancer, APS, hereditary thrombophilia)
- (c) A minor transient or reversible risk factor (e.g., OCPs, long travel)
| Clinical Scenario | Recommended Duration |
|---|---|
| Major transient/reversible risk factor (e.g., major surgery) | 3 months |
| Minor transient risk factor | 3–6 months, consider extended |
| Unprovoked PE (no identifiable risk factor) | Minimum 3 months, then reassess; extended/indefinite strongly recommended if bleeding risk acceptable |
| Cancer-associated PE | Indefinite (until cancer remission); prefer LMWH or DOACs (rivaroxaban/edoxaban) |
| Recurrent unprovoked PE | Indefinite |
| APS | Indefinite with warfarin (INR 2–3) |
Extended anticoagulation options (reduced dose after 6 months):
- Rivaroxaban 10 mg OD (EINSTEIN-CHOICE trial) — reduces recurrence, low bleeding
- Apixaban 2.5 mg BD (AMPLIFY-EXT trial) — superior to placebo, similar bleeding to placebo
- Aspirin — inferior to DOACs for recurrence prevention; only if anticoagulation refused
D. Thrombolysis (Systemic)
Indication: Massive PE (high-risk) with hemodynamic compromise
| Agent | Dose |
|---|---|
| Alteplase (tPA) | 100 mg IV over 2 hours (preferred) |
| Streptokinase | 250,000 U loading, then 100,000 U/h ×24h (less used) |
| Tenecteplase | Weight-based bolus |
Absolute contraindications: Prior hemorrhagic stroke, ischemic stroke <3 months, active bleeding, CNS neoplasm, major surgery/trauma <3 weeks, intracranial/spinal surgery <2 months
For submassive PE: Thrombolysis not routinely recommended unless clinical deterioration; individualized decision (PEITHO trial).
E. Catheter-Directed Therapy (CDT)
- Catheter-directed thrombolysis (CDT): lower dose tPA directly into thrombus via pulmonary artery catheter
- Catheter-directed mechanical thrombectomy: rheolytic, aspiration, or ultrasound-facilitated
- Indications: Massive/submassive PE when systemic thrombolysis contraindicated or failed
- EKOS system (ultrasound-accelerated CDT): reduces clot burden with lower bleeding risk
F. Surgical Embolectomy
- Reserved for: massive PE with failed/contraindicated thrombolysis; large central thrombus
- 30-day mortality: 20–30% in experienced centers
- May be performed with or without cardiopulmonary bypass
G. IVC Filter
- Indications:
- Absolute contraindication to anticoagulation
- Recurrent PE despite adequate anticoagulation
- Retrievable filters preferred — retrieve once anticoagulation can be resumed
- Do not routinely add IVC filter to anticoagulation (does not improve mortality; increases DVT risk — PREPIC trial)
10. Special Situations in Unprovoked PE
Thrombophilia Testing
- Test after completing acute anticoagulation (results unreliable during anticoagulation)
- Testing for antithrombin, protein C, protein S requires being off warfarin ≥2 weeks; being off DOACs ≥2 days
- Who to test: Young patients (<50), family history of VTE, recurrent unprovoked VTE, unusual site thrombosis (mesenteric, cerebral), APS suspicion
- Testing does not change initial management; influences long-term anticoagulation decisions
Occult Malignancy Screening
- ~4–10% of unprovoked PE patients harbor occult malignancy
- Recommended workup:
- Minimum: History, physical exam, standard bloodwork, chest X-ray, age-appropriate cancer screening (mammogram, Pap smear, colonoscopy, PSA)
- Limited CT scan of abdomen/pelvis if not already performed
- Full-body PET-CT: not routinely recommended, controversial cost-benefit
Antiphospholipid Syndrome (APS)
- Test with: anticardiolipin antibodies (IgG/IgM), anti-β₂-glycoprotein I (IgG/IgM), lupus anticoagulant
- Requires 2 positive tests ≥12 weeks apart for diagnosis
- Management: Warfarin (INR 2–3); DOACs are inferior in triple-positive APS (TRAPS trial) — avoid DOACs in confirmed APS
11. Prognosis & Recurrence
| Outcome | Data |
|---|---|
| 30-day all-cause mortality | ~10–15% (massive PE: >50%) |
| PE-specific mortality | ~2–5% in hemodynamically stable patients |
| Recurrence at 1 year (off anticoagulation) | ~10% |
| Recurrence at 5 years (off anticoagulation) | ~30% |
| Post-PE syndrome (functional limitation, dyspnea) | ~30–50% |
| Chronic thromboembolic pulmonary hypertension (CTEPH) | ~1–5% (higher in unprovoked) |
CTEPH: Failure of thrombus resolution → persistent mechanical obstruction + vascular remodeling → progressive pulmonary hypertension
- Evaluate at 3–6 months post-PE: echocardiogram, V/Q scan
- Treatment: pulmonary endarterectomy (PEA) (curative in operable disease), balloon pulmonary angioplasty (BPA), riociguat (medical therapy)
12. Predictors of Recurrence (to Guide Indefinite Anticoagulation)
| Factor Favoring Indefinite Anticoagulation | Factor Suggesting Limited Duration |
|---|---|
| Unprovoked PE (no identifiable trigger) | Major reversible trigger (surgery, trauma) |
| Male sex | Female sex (lower recurrence risk) |
| Residual thrombosis on imaging | No residual thrombus |
| Elevated D-dimer 1 month after stopping anticoagulation | D-dimer negative after stopping |
| Inherited thrombophilia (esp. homozygous, combined defects) | Low bleeding risk concern |
| Prior unprovoked VTE | First event with reversible cause |
| APS | — |
| Active cancer | — |
Clinical prediction scores for recurrence:
- HERDOO2 rule (for women): low recurrence risk if ≤1 of: hyperpigmentation/edema/redness (leg), D-dimer ≥250 μg/L, obesity (BMI ≥30), age ≥65 — these women may safely stop anticoagulation
- Vienna Prediction Model: uses sex, location (DVT vs. PE), D-dimer
- DASH score: D-dimer, Age, Sex, Hormonal therapy — ≤1 point = low risk of recurrence
13. Summary Algorithm for Unprovoked PE Management
Confirmed Unprovoked PE
↓
Acute anticoagulation (LMWH/DOAC/UFH)
↓
Risk stratify (PESI, echo, biomarkers)
↓
Massive PE ──→ Thrombolysis / Embolectomy / CDT
↓
Stable → Continue anticoagulation ≥3 months
↓
Assess: bleeding risk vs. recurrence risk
Screen for occult malignancy
Thrombophilia testing (after acute treatment)
↓
Extended/Indefinite anticoagulation (strongly preferred in unprovoked PE)
Use reduced-dose DOAC after 6 months if appropriate
↓
Follow-up echo/V-Q at 3–6 months to rule out CTEPH
Key References
- Harrison's Principles of Internal Medicine, 21st Edition (pp. 3508, 7731)
- ESC Guidelines on Pulmonary Embolism (2019, updated 2022)
- EINSTEIN-PE, AMPLIFY, AMPLIFY-EXT, PEITHO, EINSTEIN-CHOICE, Hokusai-VTE trials
- PREPIC trial (IVC filters)
- TRAPS trial (DOACs in APS)
- HERDOO2 rule, Vienna Prediction Model, DASH score
This is a shared conversation. Sign in to Orris to start your own chat.