Rejection of lung post transplantation full details
Reading File
search_medical_text
lung transplant rejection types hyperacute acute chronic pathophysiology
search_medical_text
lung transplant rejection diagnosis treatment management bronchiolitis obliterans
search_medical_image
lung transplant rejection histology pathology imaging
GradeResponse
GradeResponse
GradeResponse
Here is a comprehensive overview of lung transplant rejection:
Lung Transplant Rejection — Full Details
Overview
Lung transplant rejection is the immune-mediated attack on the allograft by the recipient's immune system. It is the leading cause of morbidity and mortality after lung transplantation. Unlike other solid organ transplants, the lung is uniquely susceptible due to its constant exposure to the external environment (pathogens, antigens), its large lymphatic network, and its rich alveolar immune cell population.
Rejection is broadly classified into:
| Type | Timing | Mechanism |
|---|---|---|
| Hyperacute rejection | Minutes to hours | Pre-formed antibodies (humoral) |
| Acute cellular rejection (ACR) | Days to months (peak: first year) | T-cell mediated |
| Antibody-mediated rejection (AMR) | Variable | Donor-specific antibodies (DSA) |
| Chronic lung allograft dysfunction (CLAD) | Months to years | Mixed cellular + humoral |
1. Hyperacute Rejection
Pathophysiology
- Caused by pre-formed recipient antibodies against donor HLA antigens or ABO blood group antigens.
- Antibodies bind to donor endothelium → complement activation → neutrophil recruitment → endothelial injury → vascular occlusion → ischemic necrosis within minutes to hours.
- Rare due to pre-transplant crossmatching and ABO matching.
Clinical Features
- Sudden onset graft failure immediately post-reperfusion.
- Severe hypoxemia, pulmonary edema, hemodynamic instability.
- Often indistinguishable clinically from severe primary graft dysfunction (PGD).
Management
- Largely preventable (crossmatch/ABO compatibility).
- Treatment: plasmapheresis, IVIG, anti-CD20 (rituximab), eculizumab.
2. Acute Cellular Rejection (ACR)
Epidemiology
- Most common in the first post-transplant year, though can occur at any time (Harrison's, p. 8150).
- Frequency decreases after year one but never disappears.
- Infections (especially CMV) can stimulate and precipitate ACR.
Pathophysiology
- T-lymphocyte mediated response to donor MHC antigens.
- Direct pathway: recipient T cells recognize intact donor MHC molecules on donor APCs.
- Indirect pathway: recipient APCs present processed donor peptides to host T cells.
- Leads to lymphocytic infiltration around blood vessels (perivascular) and airways.
Histopathology (ISHLT Grading — A and B grades)
Vascular (Grade A) — Perivascular lymphocytic infiltrates:
| Grade | Description |
|---|---|
| A0 | No rejection |
| A1 (Minimal) | Scattered perivascular mononuclear cells, ≥2 vessels affected |
| A2 (Mild) | More frequent perivascular cuffing, 2-3 cell layers thick |
| A3 (Moderate) | Dense perivascular cuffing + extension into alveolar septa, eosinophils/neutrophils |
| A4 (Severe) | Diffuse perivascular, interstitial, air-space infiltration; alveolar damage, necrosis |
Airway (Grade B) — Lymphocytic bronchiolitis:
| Grade | Description |
|---|---|
| B0 | No airway inflammation |
| B1R (Low-grade) | Rare scattered mononuclear cells in bronchiolar submucosa |
| B2R (High-grade) | Larger infiltrates with eosinophils, epithelial injury |
| BX | Ungradeable |
Clinical Features
- May be asymptomatic (detected on surveillance bronchoscopy).
- Symptoms when present: dyspnea, dry cough, low-grade fever, hypoxemia, reduced exercise tolerance.
- Decline in FEV₁/FVC on spirometry.
- CXR/CT: may be normal or show subtle infiltrates, interstitial thickening.
Diagnosis
- Transbronchial biopsy (TBB) via bronchoscopy — gold standard.
- BAL to exclude infection concurrently.
- Routine surveillance bronchoscopy programs are widely used at 1, 3, 6, 12 months post-transplant.
Treatment
- High-dose IV methylprednisolone (500–1000 mg/day × 3 days) — first-line.
- Refractory cases: antithymocyte globulin (ATG), alemtuzumab.
- Optimization of baseline immunosuppression (tacrolimus levels, MMF dosing).
- ACR is a major risk factor for CLAD (Harrison's, p. 8150).
3. Antibody-Mediated Rejection (AMR)
Pathophysiology
- Caused by donor-specific antibodies (DSA) — usually anti-HLA class I or II antibodies.
- DSA bind to donor vascular endothelium → complement activation (C1q → C4d deposition) → neutrophilic vasculitis → capillary injury.
- Increasingly recognized as a distinct and important form of rejection (Harrison's, p. 8150).
Histopathology
- Neutrophilic capillaritis involving small vessels and capillaries.
- C4d deposition on immunofluorescence (marker of complement activation).
- May be acute or chronic.
Clinical Features
- Often presents as acute deterioration in lung function.
- May mimic ACR or infectious pneumonia.
- Associated with circulating DSA on laboratory testing.
Diagnosis
- Triad:
- Clinical/functional decline
- Circulating DSA (Luminex bead assay)
- Histological evidence (neutrophilic capillaritis + C4d positivity)
Treatment
- Plasmapheresis (remove circulating antibodies)
- IVIG (immune modulation)
- Rituximab (anti-CD20, deplete B cells)
- Bortezomib (proteasome inhibitor — targets plasma cells)
- Eculizumab (anti-C5, blocks complement) in refractory cases
- Optimize calcineurin inhibitor levels
4. Chronic Lung Allograft Dysfunction (CLAD)
Overview
- The most important long-term complication of lung transplantation.
- Defined as a sustained (≥3 weeks) decline in FEV₁ to <80% of best post-transplant value.
- Affects ~50% of recipients by 5 years; the primary cause of death beyond year 1.
- Has two main phenotypes: BOS and RAS.
4a. Bronchiolitis Obliterans Syndrome (BOS)
The dominant phenotype of CLAD (~70% of cases).
Pathophysiology:
- Obliterative bronchiolitis (OB): fibroproliferative process affecting small airways.
- Subepithelial fibrosis → luminal obliteration of bronchioles → obstructive ventilatory defect.
- Triggered by repeated alloimmune injury (ACR episodes, AMR) and non-alloimmune injury (infection, GERD, aspiration).
BOS Grading (ISHLT):
| Stage | FEV₁ (% of baseline) |
|---|---|
| BOS 0 | >90% |
| BOS 0-p (potential) | 81–90% and/or FEF25–75% ≤75% |
| BOS 1 | 66–80% |
| BOS 2 | 51–65% |
| BOS 3 | ≤50% |
Clinical Features:
- Insidious progressive dyspnea and cough.
- Obstructive pattern on PFTs.
- CT: mosaic attenuation, air trapping on expiration, bronchiectasis.
- TBB often non-diagnostic (patchy lesions missed); diagnosis is clinical/functional.
Treatment:
- No proven disease-modifying therapy.
- Azithromycin — anti-inflammatory properties; shown to improve FEV₁ in some patients.
- Inhaled cyclosporine — some benefit in selected patients.
- Augmented systemic immunosuppression (tacrolimus, MMF adjustment).
- Extracorporeal photopheresis (ECP) — in refractory BOS.
- Manage treatable triggers: anti-reflux surgery (Nissen fundoplication) for GERD/aspiration, treat infections.
- Re-transplantation in selected patients with end-stage BOS.
4b. Restrictive Allograft Syndrome (RAS)
Less common (~30%) but more aggressive phenotype.
Pathophysiology:
- Pleuroparenchymal fibroelastosis + peripheral fibrosis.
- Restrictive ventilatory defect (reduced TLC).
- Upper-lobe predominant fibrosis on HRCT.
Clinical Features:
- Rapid progressive dyspnea.
- Restrictive PFT pattern.
- CT: upper-lobe pleural and subpleural fibrosis.
Prognosis:
- Median survival after RAS diagnosis: ~6–18 months (much worse than BOS).
- Poor response to current therapies.
Summary Comparison Table
| Feature | ACR | AMR | BOS (CLAD) | RAS (CLAD) |
|---|---|---|---|---|
| Timing | First year (peak) | Variable | >6–12 months | >12 months |
| Mechanism | T-cell mediated | Donor-specific antibodies | Fibroproliferative/alloimmune | Fibroproliferative |
| Histology | Perivascular lymphocytes | Neutrophilic capillaritis, C4d+ | Obliterative bronchiolitis | Pleuroparenchymal fibroelastosis |
| PFTs | ↓ FEV₁/FVC | ↓ FEV₁ | Obstructive (↓ FEV₁) | Restrictive (↓ TLC) |
| Key test | TBB | DSA + TBB + C4d | Clinical/functional + CT | Clinical/functional + CT |
| Treatment | IV methylprednisolone | Plasmapheresis/IVIG/Rituximab | Azithromycin/ECP | Limited; re-transplant |
Risk Factors for Rejection
- HLA mismatch (especially HLA-DR)
- Prior episodes of ACR
- CMV infection/mismatch (donor+/recipient−)
- Non-adherence to immunosuppression
- GERD and chronic aspiration
- Community-acquired respiratory infections
- De novo DSA development
- Prolonged ischemic time
Immunosuppression Maintenance (Standard Triple Therapy)
To prevent rejection, all lung transplant recipients receive:
- Calcineurin inhibitor: Tacrolimus (preferred) or cyclosporine
- Antiproliferative agent: Mycophenolate mofetil (MMF) or azathioprine
- Corticosteroid: Prednisone (tapering dose)
Induction therapy (perioperative): Basiliximab (IL-2R antagonist) or ATG.
Imaging in Lung Transplant Rejection
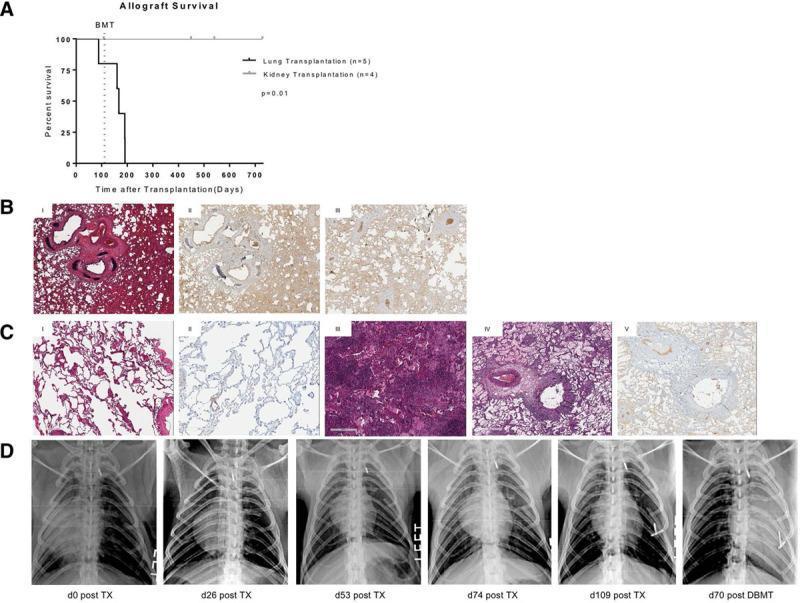
This composite image illustrates the progression of lung allograft rejection: (A) Kaplan-Meier survival curve showing inferior lung vs. kidney allograft survival; (B-C) H&E staining showing dense lymphocytic infiltration (A4 grade ACR) and C4d immunohistochemistry confirming AMR; (D) Serial chest radiographs showing progression from a well-aerated graft to diffuse opacification reflecting severe rejection.
Prognosis
- 5-year survival post-lung transplant: ~55% (ISHLT Registry).
- CLAD develops in ~50% by year 5 and accounts for ~30–40% of all post-transplant deaths.
- RAS carries a median post-diagnosis survival of <2 years.
- Re-transplantation is the only definitive treatment for end-stage CLAD but outcomes are inferior to primary transplant.
Sources: Harrison's Principles of Internal Medicine, 21st Edition (p. 8150); ISHLT Grading System for Lung Allograft Rejection; supplemented with ISHLT consensus guidelines on CLAD and AMR.
This is a shared conversation. Sign in to Orris to start your own chat.